Pyrrole is a heterocyclic, aromatic, organic compound, a five-membered ring with the formula C4H4NH. It is a colorless volatile liquid that darkens readily upon exposure to air. Substituted derivatives are also called pyrroles, e.g., N-methylpyrrole, C4H4NCH3. Porphobilinogen, a trisubstituted pyrrole, is the biosynthetic precursor to many natural products such as heme.

Terpenes are a class of natural products consisting of compounds with the formula (C5H8)n for n ≥ 2. Comprising more than 30,000 compounds, these unsaturated hydrocarbons are produced predominantly by plants, particularly conifers. Terpenes are further classified by the number of carbons: monoterpenes (C10), sesquiterpenes (C15), diterpenes (C20), as examples. The terpene alpha-pinene is a major component of the common solvent, turpentine.
Furan is a heterocyclic organic compound, consisting of a five-membered aromatic ring with four carbon atoms and one oxygen atom. Chemical compounds containing such rings are also referred to as furans.

Cinnamic acid is an organic compound with the formula C6H5-CH=CH-COOH. It is a white crystalline compound that is slightly soluble in water, and freely soluble in many organic solvents. Classified as an unsaturated carboxylic acid, it occurs naturally in a number of plants. It exists as both a cis and a trans isomer, although the latter is more common.
The Paternò–Büchi reaction, named after Emanuele Paternò and George Büchi, who established its basic utility and form, is a photochemical reaction, specifically a 2+2 photocycloaddition, which forms four-membered oxetane rings from an excited carbonyl and reacting with an alkene.
The Larock indole synthesis is a heteroannulation reaction that uses palladium as a catalyst to synthesize indoles from an ortho-iodoaniline and a disubstituted alkyne. It is also known as Larock heteroannulation. The reaction is extremely versatile and can be used to produce varying types of indoles. Larock indole synthesis was first proposed by Richard C. Larock in 1991 at Iowa State University.
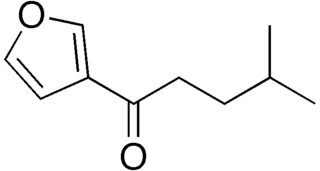
Perilla ketone is a natural terpenoid that consists of a furan ring with a six-carbon side chain containing a ketone functional group. It is a colorless oil that is sensitive to oxygen, becoming colored upon standing. The ketone was identified in 1943 by Sebe as the main component of the essential oil of Perilla frutescens. Perilla ketone is present in the leaves and seeds of purple mint, which is toxic to some animals. When cattle and horses consume purple mint when grazing in fields in which it grows, the perilla ketone causes pulmonary edema leading to a condition sometimes called perilla mint toxicosis.
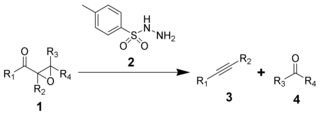
The Eschenmoser fragmentation, first published in 1967, is the chemical reaction of α,β-epoxyketones (1) with aryl sulfonylhydrazines (2) to give alkynes (3) and carbonyl compounds (4). The reaction is named after the Swiss chemist Albert Eschenmoser, who devised it in collaboration with an industrial research group of Günther Ohloff, and applied it to the production of muscone and related macrocyclic musks. The reaction is also sometimes known as the Eschenmoser–Ohloff fragmentation or the Eschenmoser–Tanabe fragmentation as Masato Tanabe independently published an article on the reaction the same year. The general formula of the fragmentation using p-toluenesulfonylhydrazide is:

Benzylideneacetone is the organic compound described by the formula C6H5CH=CHC(O)CH3. Although both cis- and trans-isomers are possible for the α,β-unsaturated ketone, only the trans isomer is observed. Its original preparation demonstrated the scope of condensation reactions to construct new, complex organic compounds. Benzylideneacetone is used as a flavouring ingredient in food and perfumes.

The Fukuyama indole synthesis is a versatile tin mediated chemical reaction that results in the formation of 2,3-disubstituted indoles. A practical one-pot reaction that can be useful for the creation of disubstituted indoles. Most commonly tributyltin hydride is utilized as the reducing agent, with azobisisobutyronitrile (AIBN) as a radical initiator. Triethylborane can also be used as a radical initiator. The reaction can begin with either an ortho-isocyanostyrene or a 2-alkenylthioanilide derivative, both forming the indole through Radical cyclization via an α-stannoimidoyl radical. The R group can be a range of both basic and acidic sensitive functional groups such as esters, THP ethers, and β-lactams. In addition the reaction is not stereospecific, in that both the cis and trans isoform can be used to obtain the desired product.

Pancratistatin (PST) is a natural compound initially extracted from spider lily, a Hawaiian native plant of the family Amaryllidaceae (AMD).
2-Methylundecanal is an organic compound that is found naturally in kumquat peel oil. This compound smells herbaceous, orange, and ambergris-like. At high dilution it has a flavor similar to honey and nuts. It is a colorless or pale yellow liquid that is soluble in organic solvents such as ether and ethanol. It is used as a fragrance component in soaps, detergents, and perfumes.

Ambroxide, widely known by the brand name Ambroxan, is a naturally occurring terpenoid and one of the key constituents responsible for the odor of ambergris. It is an autoxidation product of ambrein. Ambroxide is used in perfumery for creating ambergris notes and as a fixative. Small amounts are used as a flavoring in food.

Philip Kraft is a German organic chemist. Since 1996 he has been employed by Givaudan, a leading Flavor and Fragrance company, where he designs captive odorants for use in perfumes. He has lectured at the University of Bern, the University of Zurich, and the ETH Zurich.

3-Bromofuran is a colorless, organic compound with the molecular formula C4H3BrO. A versatile intermediate product for synthesizing more complex compounds, it used in the syntheses of a variety of economically important drugs.
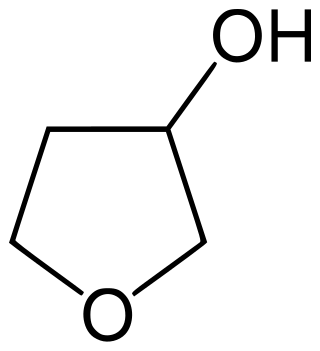
3-Hydroxytetrahydrofuran is a colorless liquid with a normal boiling point of 179 °C and boiling at 88−89 °C at 17 mmHg, with density. 3-OH THF is a useful pharmaceutical intermediate. The chiral version of this compound is an intermediate to launched retroviral drugs.

Pomarose is a high-impact captive odorant patented by Givaudan. It is a double-unsaturated ketone that does not occur in nature. Pomarose has a powerful fruity rose odor with nuances of apples, plums and raisins, which is almost entirely due to the (2E,5Z)-stereoisomer, while its (2E,5E)-isomer is barely detectable for most people. Catalyzed by traces of acids, both isomers equilibrate however quickly upon standing in glass containers.

Isovaleraldehyde organic compound, also known as 3-methylbutanal, with the formula (CH3)2CHCH2CHO. It is an aldehyde, a colorless liquid at STP, and found in low concentrations in many types of food. It can be produced commercially and is used as a reagent for the production of pharmaceuticals and pesticides.
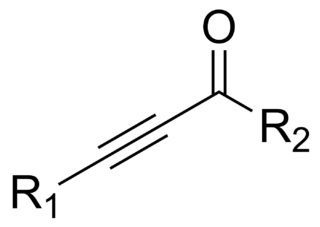
In organic chemistry, an ynone is an organic compound containing a ketone functional group and a C≡C triple bond. Many ynones are α,β-ynones, where the carbonyl and alkyne groups are conjugated. Capillin is a naturally occurring example. Some ynones are not conjugated.
Isophytol is a terpenoid alcohol that is used as a fragrance and as an intermediate in the production of vitamin E and K1.














