Structure and classification
Terpenoids are modified terpenes, [7] wherein methyl groups have been moved or removed, or oxygen atoms added. Some authors use the term "terpene" more broadly, to include the terpenoids. Just like terpenes, the terpenoids can be classified according to the number of isoprene units that comprise the parent terpene:
| Terpenoids | Analogue terpenes | Number of isoprene units | Number of carbon atoms | General formula | Examples [8] |
|---|---|---|---|---|---|
| Hemiterpenoids | Isoprene | 1 | 5 | C5H8 | DMAPP, isopentenyl pyrophosphate, isoprenol, isovaleramide, isovaleric acid, HMBPP, prenol |
| Monoterpenoids | Monoterpenes | 2 | 10 | C10H16 | Bornyl acetate, camphor, carvone, citral, citronellal, citronellol, geraniol, eucalyptol, hinokitiol, iridoids, linalool, menthol, thymol |
| Sesquiterpenoids | Sesquiterpenes | 3 | 15 | C15H24 | Farnesol, geosmin, humulone |
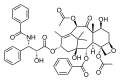
| Diterpenoids | Diterpenes | 4 | 20 | C20H32 | Abietic acid, ginkgolides, paclitaxel, retinol, salvinorin A, sclareol, steviol |
| Sesterterpenoids | Sesterterpenes | 5 | 25 | C25H40 | Andrastin A, manoalide |
| Triterpenoids | Triterpenes | 6 | 30 | C30H48 | Amyrin, betulinic acid, limonoids, oleanolic acid, sterols, squalene, ursolic acid |
| Tetraterpenoids | Tetraterpenes | 8 | 40 | C40H64 | Carotenoids |
| Polyterpenoid | Polyterpenes | >8 | >40 | (C5H8)n | Gutta-percha, natural rubber |
Terpenoids can also be classified according to the type and number of cyclic structures they contain: linear, acyclic, monocyclic, bicyclic, tricyclic, tetracyclic, pentacyclic, or macrocyclic. [8] The Salkowski test can be used to identify the presence of terpenoids. [9]
- Selected terpenoids
- Paclitaxel is a diterpenoid anticancer drug.
- Terpineols are monoterpenoids.
- Humulones are classified as sesquiterpenoids.
- Retinol is a diterpenoid.
- Hinokitiol is a monoterpenoid, a tropolone derivative.
- Geosmin is a sesquiterpenoid.