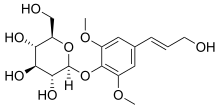
A glycosidic bond or glycosidic linkage is a type of ether bond that joins a carbohydrate (sugar) molecule to another group, which may or may not be another carbohydrate.
A glucoside is a glycoside that is chemically derived from glucose. Glucosides are common in plants, but rare in animals. Glucose is produced when a glucoside is hydrolysed by purely chemical means, or decomposed by fermentation or enzymes.

In chemistry, a glycoside is a molecule in which a sugar is bound to another functional group via a glycosidic bond. Glycosides play numerous important roles in living organisms. Many plants store chemicals in the form of inactive glycosides. These can be activated by enzyme hydrolysis, which causes the sugar part to be broken off, making the chemical available for use. Many such plant glycosides are used as medications. Several species of Heliconius butterfly are capable of incorporating these plant compounds as a form of chemical defense against predators. In animals and humans, poisons are often bound to sugar molecules as part of their elimination from the body.

Stilbenoids are hydroxylated derivatives of stilbene. They have a C6–C2–C6 structure. In biochemical terms, they belong to the family of phenylpropanoids and share most of their biosynthesis pathway with chalcones. Most stilbenoids are produced by plants, and the only known exception is the antihelminthic and antimicrobial stilbenoid, 2-isopropyl-5-[(E)-2-phenylvinyl]benzene-1,3-diol, biosynthesized by the Gram-negative bacterium Photorhabdus luminescens.

Apigenin (4′,5,7-trihydroxyflavone), found in many plants, is a natural product belonging to the flavone class that is the aglycone of several naturally occurring glycosides. It is a yellow crystalline solid that has been used to dye wool.

Lamium album, commonly called white nettle or white dead-nettle, is a flowering plant in the family Lamiaceae. It is native throughout Europe and Asia, growing in a variety of habitats from open grassland to woodland, generally on moist, fertile soils.

Phellodendron amurense is a species of tree in the family Rutaceae, commonly called the Amur cork tree. It is a major source of huáng bò, one of the 50 fundamental herbs used in traditional Chinese medicine. The Ainu people used this plant, called shikerebe-ni, as a painkiller. It is known as hwangbyeok in Korean and (キハダ) kihada in Japanese.
Neoflavonoids are a class of polyphenolic compounds. While flavonoids have the 2-phenylchromen-4-one backbone, neoflavonoids have the 4-phenylchromen backbone with no hydroxyl group substitution at position 2.

Eleutherosides are a diverse group of chemical compounds that were isolated from roots of the herb Eleutherococcus senticosus which is commercially offered mostly as extracts. Eleutheroside A is a saponin and sterol glycoside while other eleutherosides, such as eleutheroside B (syringin), are phenyl propanoid glycosides. They serve as marker compounds for the Thin layer chromatography identification of Eleutherococcus senticosus herbal preparations and dietary supplements.
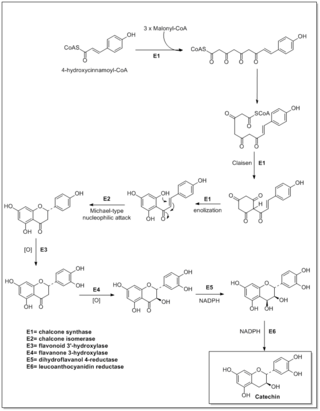
Flavonoids are synthesized by the phenylpropanoid metabolic pathway in which the amino acid phenylalanine is used to produce 4-coumaroyl-CoA. This can be combined with malonyl-CoA to yield the true backbone of flavonoids, a group of compounds called chalcones, which contain two phenyl rings. Conjugate ring-closure of chalcones results in the familiar form of flavonoids, the three-ringed structure of a flavone. The metabolic pathway continues through a series of enzymatic modifications to yield flavanones → dihydroflavonols → anthocyanins. Along this pathway, many products can be formed, including the flavonols, flavan-3-ols, proanthocyanidins (tannins) and a host of other various polyphenolics.

In enzymology, a flavonol 3-O-glucosyltransferase is an enzyme that catalyzes the chemical reaction

Dandelion 'coffee' is a tisane made from the root of the dandelion plant. The roasted dandelion root pieces and the beverage have some resemblance to coffee in appearance and taste, and it is thus commonly considered a coffee substitute. Dandelion root is used for both medicinal and culinary purposes and is thought to be a detoxifying herb.

Petunidin (Pt), like Europinidin and Malvidin, is derived from Delphinidin and is an O-methylated anthocyanidin of the 3-hydroxy type. It is a natural organic compound, a dark-red or purple water-soluble pigment found in many redberries including chokeberries, Saskatoon berries or different species of grape, and also part of the pigments responsible for the petal colors in many flowers. This pigment gives the Indigo Rose tomatoes the majority of their deep purple color when the fruits are exposed to sunlight. The name of the molecule itself is derived from the word Petunia.

Cucurbitacin is a class of biochemical compounds that some plants – notably members of the pumpkin and gourd family, Cucurbitaceae – produce and which function as a defence against herbivores. Cucurbitacins are chemically classified as triterpenes, formally derived from cucurbitane, a triterpene hydrocarbon – specifically, from the unsaturated variant cucurbit-5-ene, or 19(10→9β)-abeo-10α-lanost-5-ene. They often occur as glycosides.

Tricin is a chemical compound. It is an O-methylated flavone, a type of flavonoid. It can be found in rice bran and sugarcane.

The flavanonols are a class of flavonoids that use the 3-hydroxy-2,3-dihydro-2-phenylchromen-4-one backbone.
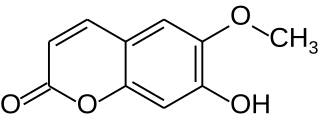
Scopoletin is a coumarin found in the root of plants in the genus Scopolia such as Scopolia carniolica and Scopolia japonica, in chicory, in Artemisia scoparia, in the roots and leaves of stinging nettle, in the passion flower, in Brunfelsia, in Viburnum prunifolium, in Solanum nigrum, in Datura metel, in Mallotus resinosus, or and in Kleinhovia hospita. It can also be found in fenugreek, vinegar, some whiskies or in dandelion coffee. A similar coumarin is scoparone. Scopoletin is highly fluorescent when dissolved in DMSO or water and is regularly used as a fluorimetric assay for the detection of hydrogen peroxide in conjunction with horseradish peroxidase. When oxidized, its fluorescence is strongly suppressed.
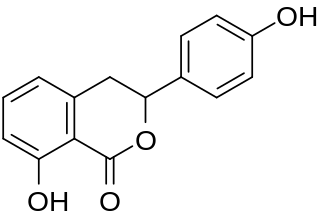
Hydrangenol is a dihydroisocoumarin. It can be found in Hydrangea macrophylla, as well as its 8-O-glucoside. (−)-Hydrangenol 4′-O-glucoside and (+)-hydrangenol 4′-O-glucoside can be found in Hydrangeae Dulcis Folium, the processed leaves of H. macrophylla var. thunbergii.

Catechin-7-O-glucoside is a flavan-3-ol glycoside formed from catechin.

Phlomoides tuberosa, the sage-leaf mullein, is a perennial herbaceous flowering plant in the family Lamiaceae, native to China, Kazakhstan, Kyrgyzstan, Mongolia, Russia; SW Asia and Europe. Enlarged, tuberous roots give rise to erect stems to 150 cm bearing purple-red flowers.