
Proton-pump inhibitors (PPIs) are a class of medications that cause a profound and prolonged reduction of stomach acid production. They do so by irreversibly inhibiting the stomach's H+/K+ ATPase proton pump.
Peptic ulcer disease (PUD) is a break in the inner lining of the stomach, the first part of the small intestine, or sometimes the lower esophagus. An ulcer in the stomach is called a gastric ulcer, while one in the first part of the intestines is a duodenal ulcer. The most common symptoms of a duodenal ulcer are waking at night with upper abdominal pain, and upper abdominal pain that improves with eating. With a gastric ulcer, the pain may worsen with eating. The pain is often described as a burning or dull ache. Other symptoms include belching, vomiting, weight loss, or poor appetite. About a third of older people have no symptoms. Complications may include bleeding, perforation, and blockage of the stomach. Bleeding occurs in as many as 15% of cases.
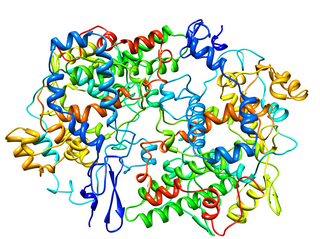
Cyclooxygenase (COX), officially known as prostaglandin-endoperoxide synthase (PTGS), is an enzyme that is responsible for biosynthesis of prostanoids, including thromboxane and prostaglandins such as prostacyclin, from arachidonic acid. A member of the animal-type heme peroxidase family, it is also known as prostaglandin G/H synthase. The specific reaction catalyzed is the conversion from arachidonic acid to prostaglandin H2 via a short-living prostaglandin G2 intermediate.

Zollinger–Ellison syndrome is a disease in which tumors cause the stomach to produce too much acid, resulting in peptic ulcers. Symptoms include abdominal pain and diarrhea.

Omeprazole, sold under the brand names Prilosec and Losec, among others, is a medication used in the treatment of gastroesophageal reflux disease (GERD), peptic ulcer disease, and Zollinger–Ellison syndrome. It is also used to prevent upper gastrointestinal bleeding in people who are at high risk. Omeprazole is a proton-pump inhibitor (PPI) and its effectiveness is similar to that of other PPIs. It can be taken by mouth or by injection into a vein. It is also available in the fixed-dose combination medication omeprazole/sodium bicarbonate as Zegerid and as Konvomep.

Gastric acid, gastric juice, or stomach acid is a digestive fluid formed within the stomach lining. With a pH between 1 and 3, gastric acid plays a key role in digestion of proteins by activating digestive enzymes, which together break down the long chains of amino acids of proteins. Gastric acid is regulated in feedback systems to increase production when needed, such as after a meal. Other cells in the stomach produce bicarbonate, a base, to buffer the fluid, ensuring a regulated pH. These cells also produce mucus – a viscous barrier to prevent gastric acid from damaging the stomach. The pancreas further produces large amounts of bicarbonate and secretes bicarbonate through the pancreatic duct to the duodenum to neutralize gastric acid passing into the digestive tract.
Parietal cells (also known as oxyntic cells) are epithelial cells in the stomach that secrete hydrochloric acid (HCl) and intrinsic factor. These cells are located in the gastric glands found in the lining of the fundus and body regions of the stomach. They contain an extensive secretory network of canaliculi from which the HCl is secreted by active transport into the stomach. The enzyme hydrogen potassium ATPase (H+/K+ ATPase) is unique to the parietal cells and transports the H+ against a concentration gradient of about 3 million to 1, which is the steepest ion gradient formed in the human body. Parietal cells are primarily regulated via histamine, acetylcholine and gastrin signalling from both central and local modulators.

Rabeprazole, sold under the brand name Aciphex, among others, is a medication that decreases stomach acid. It is used to treat peptic ulcer disease, gastroesophageal reflux disease, and excess stomach acid production such as in Zollinger–Ellison syndrome. It may also be used in combination with other medications to treat Helicobacter pylori. Effectiveness is similar to other proton pump inhibitors (PPIs). It is taken by mouth.

Achlorhydria and hypochlorhydria refer to states where the production of hydrochloric acid in gastric secretions of the stomach and other digestive organs is absent or low, respectively. It is associated with various other medical problems.
A Cushing ulcer, named after Harvey Cushing, is a gastric ulcer associated with elevated intracranial pressure. It is also called von Rokitansky–Cushing syndrome. Apart from the stomach, ulcers may also develop in the proximal duodenum and distal esophagus.

Glucose-dependent insulinotropic polypeptide, abbreviated as GIP, is an inhibiting hormone of the secretin family of hormones. While it is a weak inhibitor of gastric acid secretion, its main role, being an incretin, is to stimulate insulin secretion.
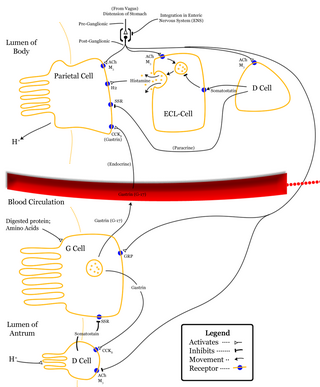
Enterochromaffin-like cells or ECL cells are a type of neuroendocrine cell found in the gastric glands of the gastric mucosa beneath the epithelium, in particular in the vicinity of parietal cells, that aid in the production of gastric acid via the release of histamine. They are also considered a type of enteroendocrine cell.
Gastric hydrogen potassium ATPase, also known as H+/K+ ATPase, is an enzyme which functions to acidify the stomach. It is a member of the P-type ATPases, also known as E1-E2 ATPases due to its two states.
Lafutidine (INN) is a second generation histamine H2 receptor antagonist having multimodal mechanism of action and used to treat gastrointestinal disorders. It is marketed in South Korea, Japan and India.

Tandamine is a selective norepinephrine reuptake inhibitor with a tricyclic structure. It was developed in the 1970s as an antidepressant but was never commercialized. Tandamine is analogous to pirandamine, which, instead, acts as a selective serotonin reuptake inhibitor (SSRI).
The nervous system, and endocrine system collaborate in the digestive system to control gastric secretions, and motility associated with the movement of food throughout the gastrointestinal tract, including peristalsis, and segmentation contractions.
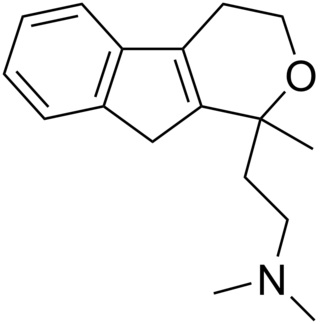
Pirandamine (AY-23,713) is a tricyclic derivative which acts as a selective serotonin reuptake inhibitor (SSRI). It was investigated in the 1970s as a potential antidepressant but clinical development was not commenced and it was never marketed. Pirandamine is structurally related to tandamine, which, in contrast, is a selective norepinephrine reuptake inhibitor.
Lupitidine (INN; lupitidine hydrochloride (USAN); development code SKF-93479) is a long-acting H2 receptor antagonist developed by Smith, Kline & French and described as an antiulcerogenic that was never marketed. It was shown to inhibit nocturnal gastric acid secretion and, in experiments on rodents, produced diffuse neuroendocrine cell hyperplasia and an increase in multifocal glandular hyperplasia due to hypergastrinemia resulting from the pharmacological suppression of gastric acid secretion.
Proton pump inhibitors (PPIs) block the gastric hydrogen potassium ATPase (H+/K+ ATPase) and inhibit gastric acid secretion. These drugs have emerged as the treatment of choice for acid-related diseases, including gastroesophageal reflux disease (GERD) and peptic ulcer disease. PPIs also can bind to other types of proton pumps such as those that occur in cancer cells and are finding applications in the reduction of cancer cell acid efflux and reduction of chemotherapy drug resistance.

Lipase inhibitors belong to a drug class that is used as an antiobesity agent. Their mode of action is to inhibit gastric and pancreatic lipases, enzymes that play an important role in the digestion of dietary fat. Lipase inhibitors are classified in the ATC-classification system as A08AB . Numerous compounds have been either isolated from nature, semi-synthesized, or fully synthesized and then screened for their lipase inhibitory activity but the only lipase inhibitor on the market is orlistat . Lipase inhibitors have also shown anticancer activity, by inhibiting fatty acid synthase.













