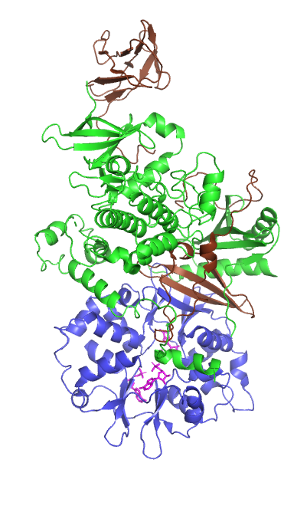
5' AMP-activated protein kinase or AMPK or 5' adenosine monophosphate-activated protein kinase is an enzyme that plays a role in cellular energy homeostasis, largely to activate glucose and fatty acid uptake and oxidation when cellular energy is low. It belongs to a highly conserved eukaryotic protein family and its orthologues are SNF1 in yeast, and SnRK1 in plants. It consists of three proteins (subunits) that together make a functional enzyme, conserved from yeast to humans. It is expressed in a number of tissues, including the liver, brain, and skeletal muscle. In response to binding AMP and ADP, the net effect of AMPK activation is stimulation of hepatic fatty acid oxidation, ketogenesis, stimulation of skeletal muscle fatty acid oxidation and glucose uptake, inhibition of cholesterol synthesis, lipogenesis, and triglyceride synthesis, inhibition of adipocyte lipogenesis, inhibition of adipocyte lipolysis, and modulation of insulin secretion by pancreatic β-cells.

MT-ATP8 is a mitochondrial gene with the full name 'mitochondrially encoded ATP synthase membrane subunit 8' that encodes a subunit of mitochondrial ATP synthase, ATP synthase Fo subunit 8. This subunit belongs to the Fo complex of the large, transmembrane F-type ATP synthase. This enzyme, which is also known as complex V, is responsible for the final step of oxidative phosphorylation in the electron transport chain. Specifically, one segment of ATP synthase allows positively charged ions, called protons, to flow across a specialized membrane inside mitochondria. Another segment of the enzyme uses the energy created by this proton flow to convert a molecule called adenosine diphosphate (ADP) to ATP. Subunit 8 differs in sequence between Metazoa, plants and Fungi.
MT-ATP6 is a mitochondrial gene with the full name 'mitochondrially encoded ATP synthase membrane subunit 6' that encodes the ATP synthase Fo subunit 6. This subunit belongs to the Fo complex of the large, transmembrane F-type ATP synthase. This enzyme, which is also known as complex V, is responsible for the final step of oxidative phosphorylation in the electron transport chain. Specifically, one segment of ATP synthase allows positively charged ions, called protons, to flow across a specialized membrane inside mitochondria. Another segment of the enzyme uses the energy created by this proton flow to convert a molecule called adenosine diphosphate (ADP) to ATP. Mutations in the MT-ATP6 gene have been found in approximately 10 to 20 percent of people with Leigh syndrome.

ATP synthase F1 subunit beta, mitochondrial is an enzyme that in humans is encoded by the ATP5F1B gene.

ATP synthase F1 subunit alpha, mitochondrial is an enzyme that in humans is encoded by the ATP5F1A gene.

ATP synthase-coupling factor 6, mitochondrial is an enzyme subunit that in humans is encoded by the ATP5PF gene.

The human ATP5F1C gene encodes the gamma subunit of an enzyme called mitochondrial ATP synthase.

ATP synthase subunit b, mitochondrial is an enzyme that in humans is encoded by the ATP5PB gene.

ATP synthase subunit s, mitochondrial is an enzyme that in humans is encoded by the ATP5S gene.

Succinyl-CoA ligase [ADP-forming] subunit beta, mitochondrial (SUCLA2), also known as ADP-forming succinyl-CoA synthetase (SCS-A), is an enzyme that in humans is encoded by the SUCLA2 gene on chromosome 13.

The human gene ATP5PD encodes subunit d of the peripheral stalk part of the enzyme mitochondrial ATP synthase.

ATP synthase subunit delta, mitochondrial, also known as ATP synthase F1 subunit delta or F-ATPase delta subunit is an enzyme that in humans is encoded by the ATP5F1D gene. This gene encodes a subunit of mitochondrial ATP synthase. Mitochondrial ATP synthase catalyzes ATP synthesis, utilizing an electrochemical gradient of protons across the inner membrane during oxidative phosphorylation.

Cytochrome c oxidase subunit 6A1, mitochondrial is a protein that in humans is encoded by the COX6A1 gene. Cytochrome c oxidase 6A1 is a subunit of the cytochrome c oxidase complex, also known as Complex IV, the last enzyme in the mitochondrial electron transport chain. A mutation of the COX6A1 gene is associated with a recessive axonal or mixed form of Charcot-Marie-Tooth disease.

ATP synthase F1 subunit epsilon, mitochondrial is an enzyme that in humans is encoded by the ATP5F1E gene. The protein encoded by ATP5F1E is a subunit of ATP synthase, also known as Complex V. Variations of this gene have been associated with mitochondrial complex V deficiency, nuclear 3 (MC5DN3) and Papillary Thyroid Cancer.

The ATP5MC3 gene is one of three human paralogs that encode membrane subunit c of the mitochondrial ATP synthase.

FAD-dependent oxidoreductase domain-containing protein 1 (FOXRED1), also known as H17, or FP634 is an enzyme that in humans is encoded by the FOXRED1 gene. FOXRED1 is an oxidoreductase and complex I-specific molecular chaperone involved in the assembly and stabilization of NADH dehydrogenase (ubiquinone) also known as complex I, which is located in the mitochondrial inner membrane and is the largest of the five complexes of the electron transport chain. Mutations in FOXRED1 have been associated with Leigh syndrome and infantile-onset mitochondrial encephalopathy.

Cytochrome b-c1 complex subunit 2, mitochondrial (UQCRC2), also known as QCR2, UQCR2, or MC3DN5 is a protein that in humans is encoded by the UQCRC2 gene. The product of UQCRC2 is a subunit of the respiratory chain protein Ubiquinol Cytochrome c Reductase, which consists of the products of one mitochondrially encoded gene, MTCYTB and ten nuclear genes: UQCRC1, UQCRC2, Cytochrome c1, UQCRFS1, UQCRB, "11kDa protein", UQCRH, Rieske Protein presequence, "cyt. c1 associated protein", and "Rieske-associated protein." Defects in UQCRC2 are associated with mitochondrial complex III deficiency, nuclear, type 5.

ATP synthase mitochondrial F1 complex assembly factor 2 is an enzyme that in humans is encoded by the ATPAF2 gene.

Cytochrome c oxidase assembly protein COX15 homolog (COX15), also known as heme A synthase, is a protein that in humans is encoded by the COX15 gene. This protein localizes to the inner mitochondrial membrane and involved in heme A biosynthesis. COX15 is also part of a three-component mono-oxygenase that catalyses the hydroxylation of the methyl group at position eight of the protoheme molecule. Mutations in this gene has been reported in patients with hypertrophic cardiomyopathy as well as Leigh syndrome, and characterized by delayed onset of symptoms, hypotonia, feeding difficulties, failure to thrive, motor regression, and brain stem signs.

Transmembrane protein 70 is a protein that in humans is encoded by the TMEM70 gene. It is a transmembrane protein located in the mitochondrial inner membrane involved in the assembly of the F1 and Fo structural subunits of ATP synthase. Mutations in this gene have been associated with neonatal mitochondrial encephalo-cardiomyopathy due to ATP synthase deficiency, causing a wide variety of symptoms including 3-methylglutaconic aciduria, lactic acidosis, mitochondrial myopathy, and cardiomyopathy.






















