A convulsion is a medical condition where the body muscles contract and relax rapidly and repeatedly, resulting in uncontrolled shaking. Because epileptic seizures typically include convulsions, the term convulsion is often used as a synonym for seizure. However, not all epileptic seizures result in convulsions, and not all convulsions are caused by epileptic seizures. Non-epileptic convulsions have no relation with epilepsy, and are caused by non-epileptic seizures.
Familial hemiplegic migraine (FHM) is an autosomal dominant type of hemiplegic migraine that typically includes weakness of half the body which can last for hours, days, or weeks. It can be accompanied by other symptoms, such as ataxia, coma, and paralysis. Migraine attacks may be provoked by minor head trauma. Some cases of minor head trauma in patients with hemiplegic migraine can develop into delayed cerebral edema, a life-threatening medical emergency. Clinical overlap occurs in some FHM patients with episodic ataxia type 2 and spinocerebellar ataxia type 6, benign familial infantile epilepsy, and alternating hemiplegia of childhood.
Sudden unexpected death in epilepsy (SUDEP) is a fatal complication of epilepsy. It is defined as the sudden and unexpected, non-traumatic and non-drowning death of a person with epilepsy, without a toxicological or anatomical cause of death detected during the post-mortem examination.
Episodic ataxia (EA) is an autosomal dominant disorder characterized by sporadic bouts of ataxia with or without myokymia. There are seven types recognized but the majority are due to two recognized entities. Ataxia can be provoked by psychological stress or startle, or heavy exertion, including exercise. Symptoms can first appear in infancy. There are at least six loci for EA, of which 4 are known genes. Some patients with EA also have migraine or progressive cerebellar degenerative disorders, symptomatic of either familial hemiplegic migraine or spinocerebellar ataxia. Some patients respond to acetazolamide though others do not.

CDKL5 is a gene that provides instructions for making a protein called cyclin-dependent kinase-like 5 also known as serine/threonine kinase 9 (STK9) that is essential for normal brain development. Mutations in the gene can cause deficiencies in the protein. The gene regulates neuronal morphology through cytoplasmic signaling and controlling gene expression. The CDKL5 protein acts as a kinase, which is an enzyme that changes the activity of other proteins by adding a cluster of oxygen and phosphorus atoms at specific positions. Researchers are currently working to determine which proteins are targeted by the CDKL5 protein.

Kv7.2 (KvLQT2) is a voltage- and lipid-gated potassium channel protein coded for by the gene KCNQ2.

Kv7.3 (KvLQT3) is a potassium channel protein coded for by the gene KCNQ3.

Sodium channel protein type 1 subunit alpha (SCN1A), is a protein which in humans is encoded by the SCN1A gene.

Sodium channel protein type 2 subunit alpha, is a protein that in humans is encoded by the SCN2A gene. Functional sodium channels contain an ion conductive alpha subunit and one or more regulatory beta subunits. Sodium channels which contain sodium channel protein type 2 subunit alpha are sometimes called Nav1.2 channels.

Leucine-rich, glioma inactivated 1, also known as LGI1, is a protein which in humans is encoded by the LGI1 gene. It may be a metastasis suppressor.

ATP-sensitive inward rectifier potassium channel 10 is a protein that in humans is encoded by the KCNJ10 gene.

Potassium voltage-gated channel subfamily KQT member 4, also known as voltage-gated potassium channel subunit Kv7.4, is a protein that in humans is encoded by the KCNQ4 gene.
A potassium channel opener is a type of drug which facilitates ion transmission through potassium channels.
M current is a type of noninactivating potassium current first discovered in bullfrog sympathetic ganglion cells.
In molecular biology, the ankyrin-G binding motif of KCNQ2-3 is a protein motif found in the potassium channels KCNQ2 and KCNQ3.
Benign familial infantile epilepsy (BFIE) is an epilepsy syndrome. Affected children, who have no other health or developmental problems, develop seizures during infancy. These seizures have focal origin within the brain but may then spread to become generalised seizures. The seizures may occur several times a day, often grouped in clusters over one to three days followed by a gap of one to three months. Treatment with anticonvulsant drugs is not necessary but they are often prescribed and are effective at controlling the seizures. This form of epilepsy resolves after one or two years, and appears to be completely benign. The EEG of these children, between seizures, is normal. The brain appears normal on MRI scan.
An epilepsy syndrome is defined as "a characteristic cluster of clinical and EEG features, often supported by specific etiological findings ."
A neonatal seizure is a seizure in a baby younger than age 4-weeks that is identifiable by an electrical recording of the brain. It is an occurrence of abnormal, paroxysmal, and persistent ictal rhythm with an amplitude of 2 microvolts in the electroencephalogram,. These may be manifested in form of stiffening or jerking of limbs or trunk. Sometimes random eye movements, cycling movements of legs, tonic eyeball movements, and lip-smacking movements may be observed. Alteration in heart rate, blood pressure, respiration, salivation, pupillary dilation, and other associated paroxysmal changes in the autonomic nervous system of infants may be caused due to these seizures. Often these changes are observed along with the observance of other clinical symptoms. A neonatal seizure may or may not be epileptic. Some of them may be provoked. Most neonatal seizures are due to secondary causes. With hypoxic ischemic encephalopathy being the most common cause in full term infants and intraventricular hemorrhage as the most common cause in preterm infants.
KCNQ2 encephalopathy typically presents with tonic seizures from the first week of life. The seizures can be frequent and often difficult to treat. Seizures can resolve within months or years but can impair the development of several domains such as motor, social, cognitive and language.
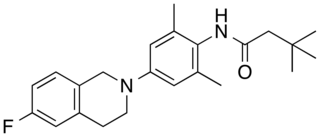
XEN1101 (encukalner) is an experimental small molecule anticonvulsant and selective Kv7.2/Kv7.3 potassium channel opener being investigated as a treatment for refractory focal onset seizures and major depressive disorder.







