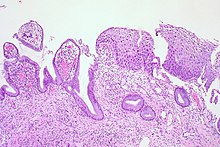
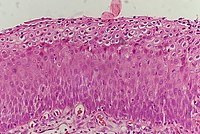
The cervix or cervix uteri is the lower part of the uterus (womb) in the human female reproductive system. The cervix is usually 2 to 3 cm long and roughly cylindrical in shape, which changes during pregnancy. The narrow, central cervical canal runs along its entire length, connecting the uterine cavity and the lumen of the vagina. The opening into the uterus is called the internal os, and the opening into the vagina is called the external os. The lower part of the cervix, known as the vaginal portion of the cervix, bulges into the top of the vagina. The cervix has been documented anatomically since at least the time of Hippocrates, over 2,000 years ago.

The Papanicolaou test is a method of cervical screening used to detect potentially precancerous and cancerous processes in the cervix or colon. Abnormal findings are often followed up by more sensitive diagnostic procedures and, if warranted, interventions that aim to prevent progression to cervical cancer. The test was independently invented in the 1920s by the Greek physician Georgios Papanikolaou and named after him. A simplified version of the test was introduced by the Canadian obstetrician Anna Marion Hilliard in 1957.

Cervical cancer is a cancer arising from the cervix. It is due to the abnormal growth of cells that have the ability to invade or spread to other parts of the body. Early on, typically no symptoms are seen. Later symptoms may include abnormal vaginal bleeding, pelvic pain or pain during sexual intercourse. While bleeding after sex may not be serious, it may also indicate the presence of cervical cancer.
Human papillomavirus infection is caused by a DNA virus from the Papillomaviridae family. Many HPV infections cause no symptoms and 90% resolve spontaneously within two years. In some cases, an HPV infection persists and results in either warts or precancerous lesions. These lesions, depending on the site affected, increase the risk of cancer of the cervix, vulva, vagina, penis, anus, mouth, tonsils, or throat. Nearly all cervical cancer is due to HPV and two strains – HPV16 and HPV18 – which account for 70% of cases. HPV16 is responsible for almost 90% of HPV-positive oropharyngeal cancers. Between 60% and 90% of the other cancers listed above are also linked to HPV. HPV6 and HPV11 are common causes of genital warts and laryngeal papillomatosis.

Colposcopy is a medical diagnostic procedure to visually examine the cervix as well as the vagina and vulva using a colposcope. Numbing should be requested prior to procedure.

Anal cancer is a cancer which arises from the anus, the distal opening of the gastrointestinal tract. Symptoms may include bleeding from the anus or a lump near the anus. Other symptoms may include pain, itchiness, or discharge from the anus. A change in bowel movements may also occur.

A precancerous condition is a condition, tumor or lesion involving abnormal cells which are associated with an increased risk of developing into cancer. Clinically, precancerous conditions encompass a variety of abnormal tissues with an increased risk of developing into cancer. Some of the most common precancerous conditions include certain colon polyps, which can progress into colon cancer, monoclonal gammopathy of undetermined significance, which can progress into multiple myeloma or myelodysplastic syndrome. and cervical dysplasia, which can progress into cervical cancer. Bronchial premalignant lesions can progress to squamous cell carcinoma of the lung.

A koilocyte is a squamous epithelial cell that has undergone a number of structural changes, which occur as a result of infection of the cell by human papillomavirus (HPV). Identification of these cells by pathologists can be useful in diagnosing various HPV-associated lesions.

Gardasil is an HPV vaccine for use in the prevention of certain strains of human papillomavirus (HPV). It was developed by Merck & Co. High-risk human papilloma virus (hr-HPV) genital infection is the most common sexually transmitted infection among women. The HPV strains that Gardasil protects against are sexually transmitted, specifically HPV types 6, 11, 16 and 18. HPV types 16 and 18 cause an estimated 70% of cervical cancers, and are responsible for most HPV-induced anal, vulvar, vaginal, and penile cancer cases. HPV types 6 and 11 cause an estimated 90% of genital warts cases. HPV type 16 is responsible for almost 90% of HPV-positive oropharyngeal cancers, and the prevalence is higher in males than females. Though Gardasil does not treat existing infection, vaccination is still recommended for HPV-positive individuals, as it may protect against one or more different strains of the disease.

Vulvar cancer is a cancer of the vulva, the outer portion of the female genitals. It most commonly affects the labia majora. Less often, the labia minora, clitoris, or vaginal glands are affected. Symptoms include a lump, itchiness, changes in the skin, or bleeding from the vulva.
Vaginal cancer is an extraordinarily rare form of cancer that develops in the tissue of the vagina. Primary vaginal cancer originates from the vaginal tissue – most frequently squamous cell carcinoma, but primary vaginal adenocarcinoma, sarcoma, and melanoma have also been reported – while secondary vaginal cancer involves the metastasis of a cancer that originated in a different part of the body. Secondary vaginal cancer is more common. Signs of vaginal cancer may include abnormal vaginal bleeding, dysuria, tenesmus, or pelvic pain, though as many as 20% of women diagnosed with vaginal cancer are asymptomatic at the time of diagnosis. Vaginal cancer occurs more frequently in women over age 50, and the mean age of diagnosis of vaginal cancer is 60 years. It often can be cured if found and treated in early stages. Surgery alone or surgery combined with pelvic radiation is typically used to treat vaginal cancer.
Vaginal intraepithelial neoplasia (VAIN) is a condition that describes premalignant histological findings in the vagina characterized by dysplastic changes.
HspE7 is an investigational therapeutic vaccine candidate being developed by Nventa Biopharmaceuticals for the treatment of precancerous and cancerous lesions caused by the human papillomavirus (HPV). HspE7 uses recombinant DNA technology to covalently fuse a heat shock protein (Hsp) to a target antigen, thereby stimulating cellular immune system responses to specific diseases. HspE7 is a patented construct consisting of the HPV Type 16 E7 protein and heat shock protein 65 (Hsp65) and is currently the only candidate using Hsp technology to target the over 20 million Americans already infected with HPV.
An anal Pap smear is the anal counterpart of the cervical Pap smear. It is used for the early detection of anal cancer. Some types of human papillomavirus (HPV) can cause anal cancer. Other HPV types cause anogenital warts. Cigarette smokers, men who have sex with men, individuals with a history of immunosuppression and women with a history of cervical, vaginal and vulval cancer are at increased risk of getting anal cancer. Vaccination against HPV before initial sexual exposure can reduce the risk of anal cancer.
The Bethesda system (TBS), officially called The Bethesda System for Reporting Cervical Cytology, is a system for reporting cervical or vaginal cytologic diagnoses, used for reporting Pap smear results. It was introduced in 1988 and revised in 1991, 2001, and 2014. The name comes from the location of the conference, sponsored by the National Institutes of Health, that established the system.
Cervicography is a diagnostic medical procedure in which a non-physician takes pictures of the cervix and submits them to a physician for interpretation. Other related procedures are speculoscopy and colposcopy. The procedure is considered a screening test for cervical cancer and is complementary to Pap smear. The technique was initially developed by Adolf Stafl, MD, of Medical College of Wisconsin in 1981.
Epithelial dysplasia, a term becoming increasingly referred to as intraepithelial neoplasia, is the sum of various disturbances of epithelial proliferation and differentiation as seen microscopically. Individual cellular features of dysplasia are called epithelial atypia.
Neuroendocrine carcinoma of the cervix is best defined separately:Neuroendocrine: Of, relating to, or involving the interaction between the nervous system and the hormones of the endocrine glands.Carcinoma: An invasive malignant tumor derived from epithelial tissue that tends to metastasize to other areas of the body.

Cervical cancer screening is a medical screening test designed to identify risk of cervical cancer. Cervical screening may involve looking for viral DNA, and/or to identify abnormal, potentially precancerous cells within the cervix as well as cells that have progressed to early stages of cervical cancer. One goal of cervical screening is to allow for intervention and treatment so abnormal lesions can be removed prior to progression to cancer. An additional goal is to decrease mortality from cervical cancer by identifying cancerous lesions in their early stages and providing treatment prior to progression to more invasive disease.
Pamela Toliman is a medical researcher from Papua New Guinea (PNG) who has researched areas such as sexually transmitted diseases, HIV/AIDS, cervical cancer and COVID-19.