
Hepadnaviridae is a family of viruses. Humans, apes, and birds serve as natural hosts. There are currently 18 species in this family, divided among 5 genera. Its best-known member is hepatitis B virus. Diseases associated with this family include: liver infections, such as hepatitis, hepatocellular carcinomas, and cirrhosis. It is the sole accepted family in the order Blubervirales.

Measles morbillivirus(MeV), also called measles virus (MV), is a single-stranded, negative-sense, enveloped, non-segmented RNA virus of the genus Morbillivirus within the family Paramyxoviridae. It is the cause of measles. Humans are the natural hosts of the virus; no animal reservoirs are known to exist.
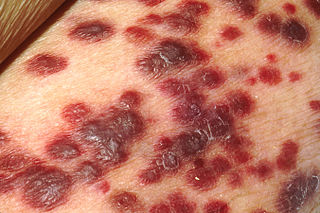
Kaposi's sarcoma-associated herpesvirus (KSHV) is the ninth known human herpesvirus; its formal name according to the International Committee on Taxonomy of Viruses (ICTV) is Human gammaherpesvirus 8, or HHV-8 in short. Like other herpesviruses, its informal names are used interchangeably with its formal ICTV name. This virus causes Kaposi's sarcoma, a cancer commonly occurring in AIDS patients, as well as primary effusion lymphoma, HHV-8-associated multicentric Castleman's disease and KSHV inflammatory cytokine syndrome. It is one of seven currently known human cancer viruses, or oncoviruses. Even after many years since the discovery of KSHV/HHV8, there is no known cure for KSHV associated tumorigenesis.

Human herpesvirus 6 (HHV-6) is the common collective name for human betaherpesvirus 6A (HHV-6A) and human betaherpesvirus 6B (HHV-6B). These closely related viruses are two of the nine known herpesviruses that have humans as their primary host.

Herpesviridae is a large family of DNA viruses that cause infections and certain diseases in animals, including humans. The members of this family are also known as herpesviruses. The family name is derived from the Greek word ἕρπειν, referring to spreading cutaneous lesions, usually involving blisters, seen in flares of herpes simplex 1, herpes simplex 2 and herpes zoster (shingles). In 1971, the International Committee on the Taxonomy of Viruses (ICTV) established Herpesvirus as a genus with 23 viruses among four groups. As of 2020, 115 species are recognized, all but one of which are in one of the three subfamilies. Herpesviruses can cause both latent and lytic infections.

Feline viral rhinotracheitis (FVR) is an upper respiratory or pulmonary infection of cats caused by Felid alphaherpesvirus 1 (FeHV-1), of the family Herpesviridae. It is also commonly referred to as feline influenza, feline coryza, and feline pneumonia but, as these terms describe other very distinct collections of respiratory symptoms, they are misnomers for the condition. Viral respiratory diseases in cats can be serious, especially in catteries and kennels. Causing one-half of the respiratory diseases in cats, FVR is the most important of these diseases and is found worldwide. The other important cause of feline respiratory disease is feline calicivirus.

Gammaherpesvirinae is a subfamily of viruses in the order Herpesvirales and in the family Herpesviridae. Viruses in Gammaherpesvirinae are distinguished by reproducing at a more variable rate than other subfamilies of Herpesviridae. Mammals serve as natural hosts. There are 43 species in this subfamily, divided among 7 genera with three species unassigned to a genus. Diseases associated with this subfamily include: HHV-4: infectious mononucleosis. HHV-8: Kaposi's sarcoma.
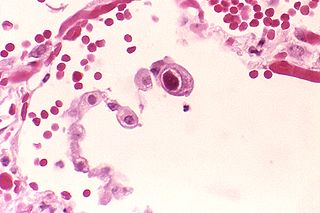
Betaherpesvirinae is a subfamily of viruses in the order Herpesvirales and in the family Herpesviridae. Mammals serve as natural hosts. There are 26 species in this subfamily, divided among 5 genera. Diseases associated with this subfamily include: human cytomegalovirus (HHV-5): congenital CMV infection; HHV-6: 'sixth disease' ; HHV-7: symptoms analogous to the 'sixth disease'.
Anthony (Tony) Charles Minson, PhD, FMedSci is a British virologist known for his work on the biology of herpesviruses, and a university administrator. He was the Senior Pro-Vice-Chancellor of the University of Cambridge from 2003 to 2009. He is an emeritus professor of virology at the university's Department of Pathology and an emeritus fellow of Wolfson College.
Human betaherpesvirus 7 (HHV-7) is one of nine known members of the Herpesviridae family that infects humans. HHV-7 is a member of Betaherpesvirinae, a subfamily of the Herpesviridae that also includes HHV-6 and Cytomegalovirus. HHV-7 often acts together with HHV-6, and the viruses together are sometimes referred to by their genus, Roseolovirus. HHV-7 was first isolated in 1990 from CD4+ T cells taken from peripheral blood lymphocytes.
Lymphocystivirus is a genus of viruses, in the family Iridoviridae. Fish serve as natural hosts. There are four species in this genus. Diseases associated with this genus include: tumor-like growths on the skin.

Alloherpesviridae is a family of viruses in the order Herpesvirales. This family includes the species that infect fish and amphibians. Phylogenetic studies have confirmed the validity of this family and suggest that it may be divided into two clades: one consisting of viruses from cyprinid and anguillid hosts and the other of viruses from ictalurid, salmonid, acipenserid, and ranid hosts. There are currently 13 species in this family, divided among four genera. A disease associated with this family includes channel catfish disease.
Ictalurivirus is a genus of viruses in the order Herpesvirales, in the family Alloherpesviridae. Fish serve as natural hosts. There are three species in this genus. Diseases associated with this genus include: channel catfish disease.
Cyvirus anguillidallo1, also known as Anguillid herpesvirus 1 (AngHV-1) is a species of virus in the genus Cyprinivirus, family Alloherpesviridae, and order Herpesvirales.
Cyvirus cyprinidallo1, also known as Cyprinid herpesvirus 1 (CyHV-1) is a species of virus in the genus Cyprinivirus, family Alloherpesviridae, and order Herpesvirales.
Acipenserid herpesvirus 2 (AciHV-2) is a virus in the genus Ictalurivirus, family Alloherpesviridae, and order Herpesvirales. It infects sturgeon of the family Acipenseridae.
Ictalurid herpesvirus 2 (IcHV-2) is a species of virus in the genus Ictalurivirus, family Alloherpesviridae, and order Herpesvirales.
Human betaherpesvirus 6A (HHV-6A) is a species of virus in the genus Roseolovirus, subfamily Betaherpesvirinae, family Herpesviridae, and order Herpesvirales.
Human betaherpesvirus 6B (HHV-6B) is a species of virus in the genus Roseolovirus, subfamily Betaherpesvirinae, family Herpesviridae, and order Herpesvirales.
Salmonid herpesvirus 3 (SalHV-3) is a species of virus in the genus Salmonivirus, family Alloherpesviridae, and order Herpesvirales.









