The management of HIV/AIDS normally includes the use of multiple antiretroviral drugs as a strategy to control HIV infection. There are several classes of antiretroviral agents that act on different stages of the HIV life-cycle. The use of multiple drugs that act on different viral targets is known as highly active antiretroviral therapy (HAART). HAART decreases the patient's total burden of HIV, maintains function of the immune system, and prevents opportunistic infections that often lead to death. HAART also prevents the transmission of HIV between serodiscordant same-sex and opposite-sex partners so long as the HIV-positive partner maintains an undetectable viral load.
Protease inhibitors (PIs) are medications that act by interfering with enzymes that cleave proteins. Some of the most well known are antiviral drugs widely used to treat HIV/AIDS, hepatitis C and COVID-19. These protease inhibitors prevent viral replication by selectively binding to viral proteases and blocking proteolytic cleavage of protein precursors that are necessary for the production of infectious viral particles.

Ritonavir, sold under the brand name Norvir, is an antiretroviral medication used along with other medications to treat HIV/AIDS. This combination treatment is known as highly active antiretroviral therapy (HAART). Ritonavir is a protease inhibitor and is used with other protease inhibitors. It may also be used in combination with other medications to treat hepatitis C and COVID-19. It is taken by mouth. Tablets of ritonavir are not bioequivalent to capsules, as the tablets may result in higher peak plasma concentrations.
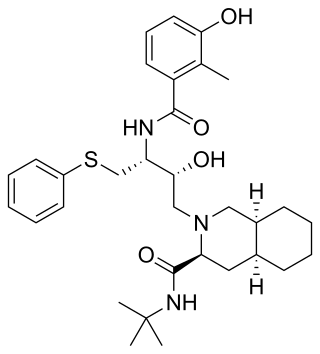
Nelfinavir, sold under the brand name Viracept, is an antiretroviral medication used in the treatment of HIV/AIDS. Nelfinavir belongs to the class of drugs known as protease inhibitors (PIs) and like other PIs is almost always used in combination with other antiretroviral drugs.

Darunavir (DRV), sold under the brand name Prezista among others, is an antiretroviral medication used to treat and prevent HIV/AIDS. It is generally recommended for use with other antiretrovirals. It is often used with low doses of ritonavir or cobicistat to increase darunavir levels. It may be used for prevention after a needlestick injury or other potential exposure. It is taken by mouth once to twice a day.

Rudi Pauwels is a Belgian pharmacologist and biotech entrepreneur.

Camostat is a serine protease inhibitor. Serine protease enzymes have a variety of functions in the body, and so camostat has a diverse range of uses. Camostat is approved in Japan for the treatment of chronic pancreatitis and postoperative reflux esophagitis. The oral proteolytic enzyme inhibitor has been on the market since 1985 under the trade name Foipan Tablets. The manufacturer is Ono Pharmaceutical. The drug is used in the treatment of some forms of cancer and is also effective against some viral infections, as well as inhibiting fibrosis in liver or kidney disease or pancreatitis.

Nafamostatmesylate (INN), a synthetic serine protease inhibitor, it is a short-acting anticoagulant, and is also used for the treatment of pancreatitis. It also has some potential antiviral and anti-cancer properties. Nafamostat is a fast-acting proteolytic inhibitor and used during hemodialysis to prevent the proteolysis of fibrinogen into fibrin. The mechanism of action of Nafamostat is as a slow tight-binding substrate, trapping the target protein in the acyl-enzyme intermediate form, resulting in apparent observed inhibition.
CCR5 receptor antagonists are a class of small molecules that antagonize the CCR5 receptor. The C-C motif chemokine receptor CCR5 is involved in the process by which HIV, the virus that causes AIDS, enters cells. Hence antagonists of this receptor are entry inhibitors and have potential therapeutic applications in the treatment of HIV infections.
Non-nucleoside reverse-transcriptase inhibitors (NNRTIs) are antiretroviral drugs used in the treatment of human immunodeficiency virus (HIV). NNRTIs inhibit reverse transcriptase (RT), an enzyme that controls the replication of the genetic material of HIV. RT is one of the most popular targets in the field of antiretroviral drug development.
Many major physiological processes depend on regulation of proteolytic enzyme activity and there can be dramatic consequences when equilibrium between an enzyme and its substrates is disturbed. In this prospective, the discovery of small-molecule ligands, like protease inhibitors, that can modulate catalytic activities has an enormous therapeutic effect. Hence, inhibition of the HIV protease is one of the most important approaches for the therapeutic intervention in HIV infection and their development is regarded as major success of structure-based drug design. They are highly effective against HIV and have, since the 1990s, been a key component of anti-retroviral therapies for HIV/AIDS.

The 3C-like protease (3CLpro) or main protease (Mpro), formally known as C30 endopeptidase or 3-chymotrypsin-like protease, is the main protease found in coronaviruses. It cleaves the coronavirus polyprotein at eleven conserved sites. It is a cysteine protease and a member of the PA clan of proteases. It has a cysteine-histidine catalytic dyad at its active site and cleaves a Gln–(Ser/Ala/Gly) peptide bond.
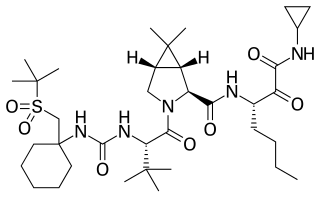
Narlaprevir, is an inhibitor of NS3/4A serine protease, intended for the treatment of chronic hepatitis C caused by genotype 1 virus in combination with other antiviral drugs.

Danoprevir (INN) is an orally available 15-membered macrocyclic peptidomimetic inhibitor of NS3/4A HCV protease. It contains acylsulfonamide, fluoroisoindole and tert-butyl carbamate moieties. Danoprevir is a clinical candidate based on its favorable potency profile against multiple HCV genotypes 1–6 and key mutants (GT1b, IC50 = 0.2–0.4 nM; replicon GT1b, EC50 = 1.6 nM). Danoprevir has been evaluated in an open-label, single arm clinical trial in combination with ritonavir for treating COVID-19 and favourably compared to lopinavir/ritonavir in a second trial.

Mozenavir (DMP-450) is an antiviral drug which was developed as a treatment for HIV/AIDS. It acts as an HIV protease inhibitor and binds to this target with high affinity, however despite promising results in early testing, mozenavir was unsuccessful in human clinical trials. Studies continue into related derivatives.
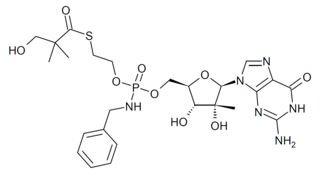
IDX-184 is an antiviral drug which was developed as a treatment for hepatitis C, acting as a NS5B RNA polymerase inhibitor. While it showed reasonable effectiveness in early clinical trials it did not progress past Phase IIb. However research using this drug has continued as it shows potentially useful activity against other emerging viral diseases such as Zika virus, and coronaviruses including MERS, and SARS-CoV-2.

Valopicitabine (NM-283) is an antiviral drug which was developed as a treatment for hepatitis C, though only progressed as far as Phase III clinical trials. It acts as an RNA-dependent RNA polymerase inhibitor. It is a prodrug which is converted inside the body to the active form, 2'-C-methylcytidine triphosphate.

Nirmatrelvir is an antiviral medication developed by Pfizer which acts as an orally active 3C-like protease inhibitor. It is part of a nirmatrelvir/ritonavir combination used to treat COVID-19 and sold under the brand name Paxlovid.

Lufotrelvir (PF-07304814) is an antiviral drug developed by Pfizer which acts as a 3CL protease inhibitor. It is a prodrug with the phosphate group being cleaved in vivo to yield the active agent PF-00835231. Lufotrelvir is in human clinical trials for the treatment of COVID-19, and shows good activity against COVID-19 including several variant strains, but unlike the related drug nirmatrelvir it is not orally active and must be administered by intravenous infusion, and so has been the less favoured candidate for clinical development overall.

Ensitrelvir, sold under the brand name Xocova is an antiviral medication used as a treatment for COVID-19. It was developed by Shionogi in partnership with Hokkaido University and acts as an orally active 3C-like protease inhibitor. It is taken by mouth.















