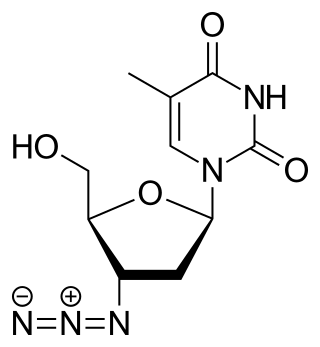
Zidovudine (ZDV), also known as azidothymidine (AZT), was the first antiretroviral medication used to prevent and treat HIV/AIDS. It is generally recommended for use in combination with other antiretrovirals. It may be used to prevent mother-to-child spread during birth or after a needlestick injury or other potential exposure. It is sold both by itself and together as lamivudine/zidovudine and abacavir/lamivudine/zidovudine. It can be used by mouth or by slow injection into a vein.
The management of HIV/AIDS normally includes the use of multiple antiretroviral drugs as a strategy to control HIV infection. There are several classes of antiretroviral agents that act on different stages of the HIV life-cycle. The use of multiple drugs that act on different viral targets is known as highly active antiretroviral therapy (HAART). HAART decreases the patient's total burden of HIV, maintains function of the immune system, and prevents opportunistic infections that often lead to death. HAART also prevents the transmission of HIV between serodiscordant same-sex and opposite-sex partners so long as the HIV-positive partner maintains an undetectable viral load.
The spread of HIV/AIDS has affected millions of people worldwide; AIDS is considered a pandemic. The World Health Organization (WHO) estimated that in 2016 there were 36.7 million people worldwide living with HIV/AIDS, with 1.8 million new HIV infections per year and 1 million deaths due to AIDS. Misconceptions about HIV and AIDS arise from several different sources, from simple ignorance and misunderstandings about scientific knowledge regarding HIV infections and the cause of AIDS to misinformation propagated by individuals and groups with ideological stances that deny a causative relationship between HIV infection and the development of AIDS. Below is a list and explanations of some common misconceptions and their rebuttals.
Protease inhibitors (PIs) are medications that act by interfering with enzymes that cleave proteins. Some of the most well known are antiviral drugs widely used to treat HIV/AIDS, hepatitis C and COVID-19. These protease inhibitors prevent viral replication by selectively binding to viral proteases and blocking proteolytic cleavage of protein precursors that are necessary for the production of infectious viral particles.
Reverse-transcriptase inhibitors (RTIs) are a class of antiretroviral drugs used to treat HIV infection or AIDS, and in some cases hepatitis B. RTIs inhibit activity of reverse transcriptase, a viral DNA polymerase that is required for replication of HIV and other retroviruses.

Zalcitabine, also called dideoxycytidine, is a nucleoside analog reverse-transcriptase inhibitor (NRTI) sold under the trade name Hivid. Zalcitabine was the third antiretroviral to be approved by the Food and Drug Administration (FDA) for the treatment of HIV/AIDS. It is used as part of a combination regimen.

Lamivudine, commonly called 3TC, is an antiretroviral medication used to prevent and treat HIV/AIDS. It is also used to treat chronic hepatitis B when other options are not possible. It is effective against both HIV-1 and HIV-2. It is typically used in combination with other antiretrovirals such as zidovudine, dolutegravir, and abacavir. Lamivudine may be included as part of post-exposure prevention in those who have been potentially exposed to HIV. Lamivudine is taken by mouth as a liquid or tablet.

Atazanavir, sold under the brand name Reyataz among others, is an antiretroviral medication used to treat HIV/AIDS. It is generally recommended for use with other antiretrovirals. It may be used for prevention after a needlestick injury or other potential exposure. It is taken by mouth.

Nevirapine (NVP), sold under the brand name Viramune among others, is a medication used to treat and prevent HIV/AIDS, specifically HIV-1. It is generally recommended for use with other antiretroviral medications. It may be used to prevent mother to child spread during birth but is not recommended following other exposures. It is taken by mouth.

Saquinavir, sold under the brand name Invirase among others, is an antiretroviral medication used together with other medications to treat or prevent HIV/AIDS. Typically it is used with ritonavir or lopinavir/ritonavir to increase its effect. It is taken by mouth.

Raltegravir, sold under the brand name Isentress, is an antiretroviral medication used, together with other medication, to treat HIV/AIDS. It may also be used, as part of post exposure prophylaxis, to prevent HIV infection following potential exposure. It is taken by mouth.
Many major physiological processes depend on regulation of proteolytic enzyme activity and there can be dramatic consequences when equilibrium between an enzyme and its substrates is disturbed. In this prospective, the discovery of small-molecule ligands, like protease inhibitors, that can modulate catalytic activities has an enormous therapeutic effect. Hence, inhibition of the HIV protease is one of the most important approaches for the therapeutic intervention in HIV infection and their development is regarded as major success of structure-based drug design. They are highly effective against HIV and have, since the 1990s, been a key component of anti-retroviral therapies for HIV/AIDS.
Discovery and development of nucleoside and nucleotide reverse-transcriptase inhibitors began in the 1980s when the AIDS epidemic hit Western societies. NRTIs inhibit the reverse transcriptase (RT), an enzyme that controls the replication of the genetic material of the human immunodeficiency virus (HIV). The first NRTI was zidovudine, approved by the U.S. Food and Drug Administration (FDA) in 1987, which was the first step towards treatment of HIV. Six NRTI agents and one NtRTI have followed. The NRTIs and the NtRTI are analogues of endogenous 2´-deoxy-nucleoside and nucleotide. Drug-resistant viruses are an inevitable consequence of prolonged exposure of HIV-1 to anti-HIV drugs.
The Berlin patient is an anonymous person from Berlin, Germany, who was described in 1998 as exhibiting prolonged "post-treatment control" of HIV viral load after HIV treatments were interrupted.

Jacques Leibowitch was a French medical doctor and clinical researcher known for his contributions to the knowledge and treatment of HIV and AIDS, starting with his initial designation of a human retrovirus as the cause of AIDS, and his ground-breaking use of triple combination therapy for the effective control of HIV in the patient. A practicing physician in the infectiology department of the Raymond Poincaré University Hospital of Garches, University lecturer Emeritus, he led the treatment program ICCARRE that proposes a dramatic reduction of weekly anti-HIV drug intake, down to 2-3 anti-viral pills a day taken 2 to 3 or 4 days a week, as opposed to the presently recommended seven days a week, as still universally prescribed. These reduced medical dosages are adequate, necessary and sufficient according to the results of his exploratory clinical research carried out since 2003. He is the author of the books "Un virus étrange venu d'ailleurs", and "Pour en finir avec le sida".

VIR-576 is an experimental drug that is under clinical trials for the treatment of HIV-1 infections. VIR-576 is synthetic peptide that binds to HIV-1's hydrophobic fusion peptide gp41, preventing the virus from inserting itself into a host cell's membrane to initiate an infection. This drug is a synthesized variant of a highly specific natural entry inhibitor designated as VIRIP.
Deborah Persaud is a Guyanese-born American virologist who primarily works on HIV/AIDS at Johns Hopkins Children's Center.
HIV in pregnancy is the presence of an HIV/AIDS infection in a woman while she is pregnant. There is a risk of HIV transmission from mother to child in three primary situations: pregnancy, childbirth, and while breastfeeding. This topic is important because the risk of viral transmission can be significantly reduced with appropriate medical intervention, and without treatment HIV/AIDS can cause significant illness and death in both the mother and child. This is exemplified by data from The Centers for Disease Control (CDC): In the United States and Puerto Rico between the years of 2014–2017, where prenatal care is generally accessible, there were 10,257 infants in the United States and Puerto Rico who were exposed to a maternal HIV infection in utero who did not become infected and 244 exposed infants who did become infected.

Abacavir/dolutegravir/lamivudine, sold under the brand name Triumeq among others, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. It is a combination of three medications with different and complementary mechanisms of action: abacavir, dolutegravir and lamivudine.
Viral load monitoring for HIV is the regular measurement of the viral load of individual HIV-positive people as part of their personal plan for treatment of HIV/AIDS. A count of the viral load is routine before the start of HIV treatment.