
Estradiol acetate (EA), sold under the brand names Femtrace, Femring, and Menoring, is an estrogen medication which is used in hormone therapy for the treatment of menopausal symptoms in women. It is taken by mouth once daily or given as a vaginal ring once every three months.
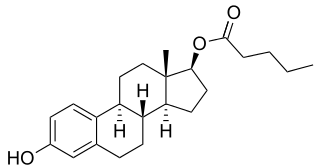
Estradiol valerate (EV), sold for use by mouth under the brand name Progynova and for use by injection under the brand names Delestrogen and Progynon Depot among others, is an estrogen medication. It is used in hormone therapy for menopausal symptoms and low estrogen levels, hormone therapy for transgender people, and in hormonal birth control. It is also used in the treatment of prostate cancer. The medication is taken by mouth or by injection into muscle or fat once every 1 to 4 weeks.
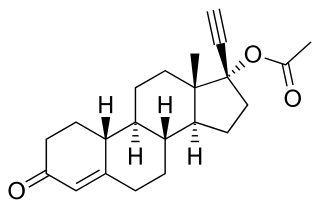
Norethisterone acetate (NETA), also known as norethindrone acetate and sold under the brand name Primolut-Nor among others, is a progestin medication which is used in birth control pills, menopausal hormone therapy, and for the treatment of gynecological disorders. The medication available in low-dose and high-dose formulations and is used alone or in combination with an estrogen. It is ingested orally.
Combined injectable contraceptives (CICs) are a form of hormonal birth control for women. They consist of monthly injections of combined formulations containing an estrogen and a progestin to prevent pregnancy.

Estradiol benzoate (EB), sold under the brand name Progynon-B among others, is an estrogen medication which is used in hormone therapy for menopausal symptoms and low estrogen levels in women, in hormone therapy for transgender women, and in the treatment of gynecological disorders. It is also used in the treatment of prostate cancer in men. Estradiol benzoate is used in veterinary medicine as well. When used clinically, the medication is given by injection into muscle usually two to three times per week.

Estradiol cypionate (EC), sold under the brand name Depo-Estradiol among others, is an estrogen medication which is used in hormone therapy for menopausal symptoms and low estrogen levels in women, in hormone therapy for trans women, and in hormonal birth control for women. It is given by injection into muscle once every 1 to 4 weeks.

Norethisterone enanthate (NETE), also known as norethindrone enanthate, is a form of hormonal birth control which is used to prevent pregnancy in women. It is used both as a form of progestogen-only injectable birth control and in combined injectable birth control formulations. It may be used following childbirth, miscarriage, or abortion. The failure rate per year in preventing pregnancy for the progestogen-only formulation is 2 per 100 women. Each dose of this form lasts two months with only up to two doses typically recommended.
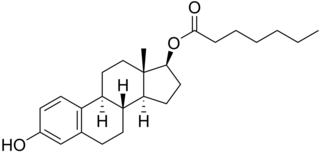
Estradiol enantate, also spelled estradiol enanthate and sold under the brand names Perlutal and Topasel among others, is an estrogen medication which is used in hormonal birth control for women. It is formulated in combination with dihydroxyprogesterone acetophenide, a progestin, and is used specifically as a combined injectable contraceptive. Estradiol enantate is not available for medical use alone. The medication, in combination with DHPA, is given by injection into muscle once a month.

Estradiol undecylate, also known as estradiol undecanoate and formerly sold under the brand names Delestrec and Progynon Depot 100 among others, is an estrogen medication which has been used in the treatment of prostate cancer in men. It has also been used as a part of hormone therapy for transgender women. Although estradiol undecylate has been used in the past, it was discontinued .The medication has been given by injection into muscle usually once a month.
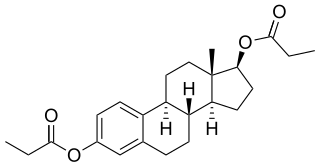
Estradiol dipropionate (EDP), sold under the brand names Agofollin, Di-Ovocylin, and Progynon DP among others, is an estrogen medication which has been used in hormone therapy for menopausal symptoms and low estrogen levels in women and in the treatment of gynecological disorders. It has also been used in feminizing hormone therapy for transgender women and in the treatment of prostate cancer in men. Although widely used in the past, estradiol dipropionate has largely been discontinued and is mostly no longer available today. It appears to remain in use only in Japan, Macedonia, and Australia. Estradiol dipropionate is given by injection into muscle at intervals ranging from once or twice a week to once every week and a half to two weeks.
An estrogen ester is an ester of an estrogen, most typically of estradiol but also of other estrogens such as estrone, estriol, and even nonsteroidal estrogens like diethylstilbestrol. Esterification renders estradiol into a prodrug of estradiol with increased resistance to first-pass metabolism, slightly improving its oral bioavailability. In addition, estrogen esters have increased lipophilicity, which results in a longer duration when given by intramuscular or subcutaneous injection due to the formation of a long-lasting local depot in muscle and fat. Conversely, this is not the case with intravenous injection or oral administration. Estrogen esters are rapidly hydrolyzed into their parent estrogen by esterases once they have been released from the depot. Because estradiol esters are prodrugs of estradiol, they are considered to be natural and bioidentical forms of estrogen.

Estradiol dienanthate (EDE), sold under the brand names Climacteron among others, is a long-acting estrogen medication which was previously used in menopausal hormone therapy for women and to suppress lactation in women. It was formulated in combination with estradiol benzoate (EB), a short-acting estrogen, and testosterone enanthate benzilic acid hydrazone (TEBH), a long-acting androgen/anabolic steroid. EDE has not been made available for medical use alone. The medication, in combination with EB and TEBH, was given by injection into muscle once or at regular intervals, for instance once every 6 weeks.

Estradiol benzoate butyrate (EBB), sold under the brand names Neolutin N, Redimen, Soluna, and Unijab and formerly known under the developmental code name Unimens, is an estrogen medication which is used in hormonal birth control for women. It is formulated in combination with dihydroxyprogesterone acetophenide, a progestin, and is used specifically as a combined injectable contraceptive. EBB is not available for medical use alone. The medication, in combination with DHPA, is given by injection into muscle once a month.

Estradiol (E2) is a medication and naturally occurring steroid hormone. It is an estrogen and is used mainly in menopausal hormone therapy and to treat low sex hormone levels in women. It is also used in hormonal birth control for women, in feminizing hormone therapy for transgender women, and in the treatment of hormone-sensitive cancers like prostate cancer in men and breast cancer in women, among other uses. Estradiol can be taken by mouth, held and dissolved under the tongue, as a gel or patch that is applied to the skin, in through the vagina, by injection into muscle or fat, or through the use of an implant that is placed into fat, among other routes.

Estrone (E1), sold under the brand names Estragyn, Kestrin, and Theelin among many others, is an estrogen medication and naturally occurring steroid hormone which has been used in menopausal hormone therapy and for other indications. It has been provided as an aqueous suspension or oil solution given by injection into muscle and as a vaginal cream applied inside of the vagina. It can also be taken by mouth as estradiol/estrone/estriol and in the form of prodrugs like estropipate and conjugated estrogens.

The pharmacology of estradiol, an estrogen medication and naturally occurring steroid hormone, concerns its pharmacodynamics, pharmacokinetics, and various routes of administration.

Estradiol benzoate/progesterone (EB/P4), sold under the brand names Duogynon and Sistocyclin among others, is a combination medication of estradiol benzoate (EB), an estrogen, and progesterone (P4), a progestogen. It has been formulated both as short-acting oil solutions and long-acting microcrystalline aqueous suspensions and is given by injection into muscle either once or continuously at regular intervals.

Estradiol valerate/hydroxyprogesterone caproate (EV/OHPC), sold under the brand names Gravibinon and Injectable No. 1 among others, is a combined estrogen and progestogen medication which is used in the treatment of threatened miscarriage and other indications and as a form of combined injectable birth control to prevent pregnancy. It contains estradiol valerate (EV), an estrogen, and hydroxyprogesterone caproate (OHPC), a progestin. The medication is given by injection into muscle once a day to once a month depending on the indication.

Lynestrenol phenylpropionate (LPP), also known as ethynylestrenol phenylpropionate, is a progestin and a progestogen ester which was developed for potential use as a progestogen-only injectable contraceptive by Organon but was never marketed. It was assessed at doses of 25 to 75 mg in an oil solution once a month by intramuscular injection. LPP was associated with high contraceptive failure at the low dose and with poor cycle control. The medication was found to produce estrogenic effects in the endometrium in women due to transformation into estrogenic metabolites.
Estradiol diundecylate/hydroxyprogesterone heptanoate/testosterone cyclohexylpropionate (EDU/OHPH/TCHP), sold under the brand name Trioestrine Retard, is an injectable combination medication of estradiol diundecylate (EDU), an estrogen, hydroxyprogesterone heptanoate (OHPH), a progestogen, and testosterone cyclohexylpropionate (TCHP), an androgen/anabolic steroid. It contained 2.25 mg EDU, 100 mg OHPH, and 67.5 mg TCHP in oil solution, was provided as ampoules, and was administered by intramuscular injection. The medication was manufactured by Roussel and Théramex and was marketed by 1953. It is no longer available.















