Related Research Articles

Tannins are a class of astringent, polyphenolic biomolecules that bind to and precipitate proteins and various other organic compounds including amino acids and alkaloids.

Flavan-3-ols are a subgroup of flavonoids. They are derivatives of flavans that possess a 2-phenyl-3,4-dihydro-2H-chromen-3-ol skeleton. Flavan-3-ols are structurally diverse and include a range of compounds, such as catechin, epicatechin gallate, epigallocatechin, epigallocatechin gallate, proanthocyanidins, theaflavins, thearubigins. They play a part in plant defense and are present in the majority of plants.

Polyphenols are a large family of naturally occurring phenols. They are abundant in plants and structurally diverse. Polyphenols include flavonoids, tannic acid, and ellagitannin, some of which have been used historically as dyes and for tanning garments.

Procyanidins are members of the proanthocyanidin class of flavonoids. They are oligomeric compounds, formed from catechin and epicatechin molecules. They yield cyanidin when depolymerized under oxidative conditions.

The phenolic content in wine refers to the phenolic compounds—natural phenol and polyphenols—in wine, which include a large group of several hundred chemical compounds that affect the taste, color and mouthfeel of wine. These compounds include phenolic acids, stilbenoids, flavonols, dihydroflavonols, anthocyanins, flavanol monomers (catechins) and flavanol polymers (proanthocyanidins). This large group of natural phenols can be broadly separated into two categories, flavonoids and non-flavonoids. Flavonoids include the anthocyanins and tannins which contribute to the color and mouthfeel of the wine. The non-flavonoids include the stilbenoids such as resveratrol and phenolic acids such as benzoic, caffeic and cinnamic acids.

The color of wine is one of the most easily recognizable characteristics of wines. Color is also an element in wine tasting since heavy wines generally have a deeper color. The accessory traditionally used to judge the wine color was the tastevin, a shallow cup allowing one to see the color of the liquid in the dim light of a cellar. The color is an element in the classification of wines.

Prodelphinidin is a name for the polymeric tannins composed of gallocatechin. It yields delphinidin during depolymerisation under oxidative conditions.

Leucocyanidin is a colorless chemical compound that is a member of the class of natural products known as leucoanthocyanidins.

Oenin is an anthocyanin. It is the 3-glucoside of malvidin. It is one of the red pigments found in the skin of purple grapes and in wine.

Procyanidin C2 is a B type proanthocyanidin trimer, a type of condensed tannin.

Procyanidin B2 is a B type proanthocyanidin. Its structure is (−)-Epicatechin-(4β→8)-(−)-epicatechin.
A type proanthocyanidins are a specific type of proanthocyanidins, which are a class of flavonoid. Proanthocyanidins fall under a wide range of names in the nutritional and scientific vernacular, including oligomeric proanthocyanidins, flavonoids, polyphenols, condensed tannins, and OPCs. Proanthocyanidins were first popularized by French scientist Jacques Masquelier.

Procyanidin B1 is a procyanidin dimer.
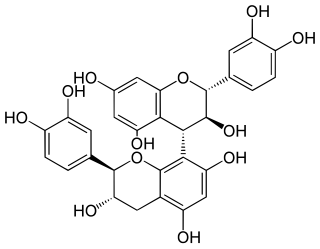
Procyanidin B3 is a B type proanthocyanidin. Procyanidin B3 is a catechin dimer.

Procyanidin B5 is a B type proanthocyanidin.
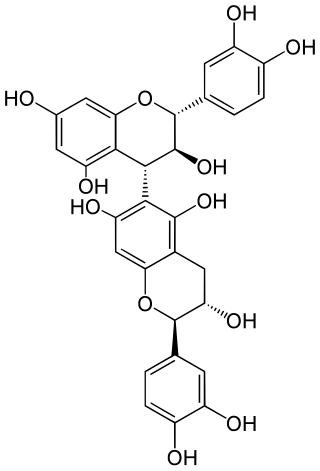
Procyanidin B6 is a B type proanthocyanidin.

Procyanidin C1 (PCC1) is a B type proanthocyanidin. It is an epicatechin trimer found in grape, unripe apples, and cinnamon.

Condensed tannins are polymers formed by the condensation of flavans. They do not contain sugar residues.
B type proanthocyanidins are a specific type of proanthocyanidin, which are a class of flavanoids. They are oligomers of flavan-3-ols.

Catechin-7-O-glucoside is a flavan-3-ol glycoside formed from catechin.
References
- ↑ Schwitters, Bert (1995). OPC in Practice. Publishing rights search incomplete. p. 15. ISBN 978-8886035132.
- 1 2 "Scientific Opinion on the substantiation of a health claim related to CranMax® and reduction of the risk of urinary tract infection by inhibiting the adhesion of certain bacteria in the urinary tract pursuant to Article 14 of Regulation (EC) No 1924/2006". EFSA Journal. 12 (5): 3657 (11 pgs). 2014. doi: 10.2903/j.efsa.2014.3657 .
A cause and effect relationship has not been established between the consumption of CranMax® and reduction of the risk of urinary tract infection by inhibiting the adhesion of certain bacteria in the urinary tract
- 1 2 Williams, Gabrielle; Stothart, Christopher I.; Hahn, Deirdre; Stephens, Jacqueline H.; Craig, Jonathan C.; Hodson, Elisabeth M. (2023-11-10). "Cranberries for preventing urinary tract infections". The Cochrane Database of Systematic Reviews. 2023 (11): CD001321. doi:10.1002/14651858.CD001321.pub7. ISSN 1469-493X. PMC 10636779. PMID 37947276.
- 1 2 Mateos-Martín ML, Fuguet E, Quero C, et al. (2012). "New identification of proanthocyanidins in cinnamon (Cinnamomum zeylanicum L.) using MALDI-TOF/TOF mass spectrometry". Analytical and Bioanalytical Chemistry. 402 (3): 1327–1336. doi:10.1007/s00216-011-5557-3. hdl:10261/88579. PMID 22101466. S2CID 23120410.
- ↑ Souquet, J; Cheynier, Véronique; Brossaud, Franck; Moutounet, Michel (1996). "Polymeric proanthocyanidins from grape skins". Phytochemistry. 43 (2): 509–512. Bibcode:1996PChem..43..509S. doi:10.1016/0031-9422(96)00301-9.
- 1 2 3 4 5 "USDA Database for the Proanthocyanidin Content of Selected Foods – 2004" (PDF). USDA. 2004. Retrieved 24 April 2014.
- ↑ Vivas, N; Nonier, M; Pianet, I; Vivasdegaulejac, N; Fouquet, E (2006). "Proanthocyanidins from Quercus petraea and Q. robur heartwood: quantification and structures". Comptes Rendus Chimie. 9: 120–126. doi:10.1016/j.crci.2005.09.001.
- ↑ Pacheco-Palencia LA, Mertens-Talcott S, Talcott ST (Jun 2008). "Chemical composition, antioxidant properties, and thermal stability of a phytochemical enriched oil from Acai (Euterpe oleracea Mart.)". J Agric Food Chem. 56 (12): 4631–4636. doi:10.1021/jf800161u. PMID 18522407.
- ↑ Hammerstone, John F.; Lazarus, Sheryl A.; Schmitz, Harold H. (August 2000). "Procyanidin content and variation in some commonly consumed foods". The Journal of Nutrition. 130 (8S Suppl): 2086S–2092S. doi: 10.1093/jn/130.8.2086S . PMID 10917927.
Figure 5
- ↑ Rohdewald, P (2002). "A review of the French maritime pine bark extract (Pycnogenol), a herbal medication with a diverse clinical pharmacology". International Journal of Clinical Pharmacology and Therapeutics. 40 (4): 158–168. doi:10.5414/cpp40158. PMID 11996210.
- ↑ Hatano, T; Miyatake, H; Natsume, M; Osakabe, N; Takizawa, T; Ito, H; Yoshida, T (2002). "Proanthocyanidin glycosides and related polyphenols from cacao liquor and their antioxidant effects". Phytochemistry. 59 (7): 749–758. Bibcode:2002PChem..59..749H. doi:10.1016/S0031-9422(02)00051-1. PMID 11909632.
- ↑ Merghem, R.; Jay, M.; Brun, N.; Voirin, B. (2004). "Qualitative analysis and HPLC isolation and identification of procyanidins fromvicia faba". Phytochemical Analysis. 15 (2): 95–99. Bibcode:2004PChAn..15...95M. doi:10.1002/pca.731. PMID 15116939.
- ↑ Van Der Poel, A. F. B.; Dellaert, L. M. W.; Van Norel, A.; Helsper, J. P. F. G. (2007). "The digestibility in piglets of faba bean (Vicia faba L.) as affected by breeding towards the absence of condensed tannins". British Journal of Nutrition. 68 (3): 793–800. doi: 10.1079/BJN19920134 . PMID 1493141.
- ↑ Griffiths, D. W. (1981). "The polyphenolic content and enzyme inhibitory activity of testas from bean (Vicia faba) and pea (Pisum spp.) varieties". Journal of the Science of Food and Agriculture. 32 (8): 797–804. Bibcode:1981JSFA...32..797G. doi:10.1002/jsfa.2740320808.
- ↑ Qa’Dan, F.; Petereit, F.; Mansoor, K.; Nahrstedt, A. (2006). "Antioxidant oligomeric proanthocyanidins fromCistus salvifolius". Natural Product Research. 20 (13): 1216–1224. doi:10.1080/14786410600899225. PMID 17127512. S2CID 254865.
- 1 2 Luca SV, Macovei I, Bujor A, Trifan A (2020). "Bioactivity of dietary polyphenols: The role of metabolites". Critical Reviews in Food Science and Nutrition . 60 (4): 626–659. doi:10.1080/10408398.2018.1546669. PMID 30614249. S2CID 58651581.
- ↑ Bogs, J.; Jaffe, F. W.; Takos, A. M.; Walker, A. R.; Robinson, S. P. (2007). "The Grapevine Transcription Factor VvMYBPA1 Regulates Proanthocyanidin Synthesis during Fruit Development". Plant Physiology. 143 (3): 1347–1361. doi:10.1104/pp.106.093203. PMC 1820911 . PMID 17208963.
- ↑ Cabrera, A.; Martin, A. (2009). "Genetics of tannin content and its relationship with flower and testa colours in Vicia faba". The Journal of Agricultural Science. 113: 93–98. doi:10.1017/S0021859600084665. S2CID 84456901.
- ↑ Kristensen, H.; Aastrup, S. (1986). "A non-destructive screening method for proanthocyanidin-free barley mutants". Carlsberg Research Communications. 51 (7): 509–513. doi: 10.1007/BF02906893 .
- ↑ Grape Seed Extract, White paper, The Grape Seed Method Evaluation Committee, Under the Auspices of NNFA ComPli Archived 2002-12-24 at the Wayback Machine
- ↑ Stringano, E; Gea, A; Salminen, J. P.; Mueller-Harvey, I (2011). "Simple solution for a complex problem: Proanthocyanidins, galloyl glucoses and ellagitannins fit on a single calibration curve in high performance-gel permeation chromatography". Journal of Chromatography A. 1218 (43): 7804–7812. doi:10.1016/j.chroma.2011.08.082. PMID 21930278.
- ↑ Engström, M. T.; Pälijärvi, M; Fryganas, C; Grabber, J. H.; Mueller-Harvey, I; Salminen, J. P. (2014). "Rapid Qualitative and Quantitative Analyses of Proanthocyanidin Oligomers and Polymers by UPLC-MS/MS". Journal of Agricultural and Food Chemistry. 62 (15): 3390–3399. doi:10.1021/jf500745y. PMID 24665824.
- ↑ Torres, J. L.; Lozano, C. (2001). "Chromatographic characterization of proanthocyanidins after thiolysis with cysteamine". Chromatographia. 54 (7–8): 523–526. doi:10.1007/BF02491211. S2CID 95355684.
- ↑ Jorgensen, Emily M.; Marin, Anna B.; Kennedy, James A. (2004). "Analysis of the Oxidative Degradation of Proanthocyanidins under Basic Conditions". Journal of Agricultural and Food Chemistry. 52 (8): 2292–2296. doi:10.1021/jf035311i. PMID 15080635.
- ↑ Li, Hui-Jing; Deinzer, Max L (2007). "Tandem mass spectrometry for sequencing proanthocyanidins". Analytical Chemistry. 79 (4): 1739–1748. doi:10.1021/ac061823v. PMID 17297981. INIST 18534021.
- ↑ Carpenter JL, Caruso FL, Tata A, Vorsa N, Neto CC (2014). "Variation in proanthocyanidin content and composition among commonly grown North American cranberry cultivars (Vaccinium macrocarpon)". J Sci Food Agric. 94 (13): 2738–2745. Bibcode:2014JSFA...94.2738C. doi:10.1002/jsfa.6618. PMID 24532348.
- ↑ Taheri, Rod; Connolly, Bryan A.; Brand, Mark H.; Bolling, Bradley W. (2013). "Underutilized Chokeberry (Aronia melanocarpa, Aronia arbutifolia, Aronia prunifolia) Accessions Are Rich Sources of Anthocyanins, Flavonoids, Hydroxycinnamic Acids, and Proanthocyanidins". Journal of Agricultural and Food Chemistry. 61 (36): 8581–8588. doi:10.1021/jf402449q. PMID 23941506.
- ↑ Rösch, Daniel R.; Mügge, Clemens; Fogliano, Vincenzo; Kroh, Lothar W. (2004). "Antioxidant Oligomeric Proanthocyanidins from Sea Buckthorn (Hippophaë rhamnoides) Pomace". Journal of Agricultural and Food Chemistry. 52 (22): 6712–6718. doi:10.1021/jf040241g. PMID 15506806.
- ↑ Kandil, F. E.; Song, L.; Pezzuto, J. M.; Marley, K.; Seigler, D. S.; Smith, M. A. L. (2000). "Isolation of oligomeric proanthocyanidins from flavonoid-producing cell cultures". In Vitro Cellular & Developmental Biology - Plant. 36 (6): 492–500. doi:10.1007/s11627-000-0088-1. S2CID 25781920.
- ↑ Amil-Ruiz, F.; Blanco-Portales, R.; Munoz-Blanco, J.; Caballero, J. L. (2011). "The Strawberry Plant Defense Mechanism: A Molecular Review". Plant and Cell Physiology. 52 (11): 1873–1903. doi: 10.1093/pcp/pcr136 . PMID 21984602.
- ↑ Matthews, S.; Mila, I.; Scalbert, A.; Pollet, B.; Lapierre, C.; Hervé Du Penhoat, C. L. M.; Rolando, C.; Donnelly, D. M. X. (1997). "Method for Estimation of Proanthocyanidins Based on Their Acid Depolymerization in the Presence of Nucleophiles". Journal of Agricultural and Food Chemistry. 45 (4): 1195–1201. doi:10.1021/jf9607573.
- 1 2 Liu, H; Zou, T; Gao, J. M.; Gu, L (2013). "Depolymerization of cranberry procyanidins using (+)-catechin, (-)-epicatechin, and (-)-epigallocatechin gallate as chain breakers". Food Chemistry. 141 (1): 488–494. doi:10.1016/j.foodchem.2013.03.003. PMID 23768384.
- ↑ Kennedy, J. A.; Jones, G. P. (2001). "Analysis of Proanthocyanidin Cleavage Products Following Acid-Catalysis in the Presence of Excess Phloroglucinol". Journal of Agricultural and Food Chemistry. 49 (4): 1740–1746. doi:10.1021/jf001030o. PMID 11308320.
- ↑ Sears, K. D.; Casebier, R. L. (1968). "Cleavage of proanthocyanidins with thioglycollic acid". Chemical Communications (22): 1437. doi:10.1039/C19680001437.
- ↑ Vernhet, A.; Dubascoux, S. P.; Cabane, B.; Fulcrand, H. L. N.; Dubreucq, E.; Poncet-Legrand, C. L. (2011). "Characterization of oxidized tannins: Comparison of depolymerization methods, asymmetric flow field-flow fractionation and small-angle X-ray scattering". Analytical and Bioanalytical Chemistry. 401 (5): 1559–1569. doi:10.1007/s00216-011-5076-2. PMID 21573842. S2CID 4645218., Vernhet, A.; Dubascoux, S. P.; Cabane, B.; Fulcrand, H. L. N.; Dubreucq, E.; Poncet-Legrand, C. L. (2011). "Characterization of oxidized tannins: Comparison of depolymerization methods, asymmetric flow field-flow fractionation and small-angle X-ray scattering". Analytical and Bioanalytical Chemistry. 401 (5): 1559–1569. doi:10.1007/s00216-011-5076-2. PMID 21573842. S2CID 4645218.
- ↑ Lange, B. M.; Lapierre, C.; Sandermann Jr, H. (1995). "Elicitor-Induced Spruce Stress Lignin (Structural Similarity to Early Developmental Lignins)". Plant Physiology. 108 (3): 1277–1287. doi:10.1104/pp.108.3.1277. PMC 157483 . PMID 12228544.
- ↑ Douglas-Fir Bark: Characterization of a Condensed Tannin Extract, by Hong-Keun Song, A thesis submitted to Oregon State University in partial fulfillment of the requirements for the degree of Master of Science, December 13, 1984
- ↑ Zhang, L. L.; Lin, Y. M. (2008). "HPLC, NMR and MALDI-TOF MS Analysis of Condensed Tannins from Lithocarpus glaber Leaves with Potent Free Radical Scavenging Activity". Molecules. 13 (12): 2986–2997. doi: 10.3390/molecules13122986 . PMC 6245341 . PMID 19052523.
- ↑ Howell, AB (2007). "Bioactive compounds in cranberries and their role in prevention or urinary tract infections". Mol Nutr Food Res. 51 (6): 732–737. doi:10.1002/mnfr.200700038. PMID 17487930.
- ↑ Luís, Â; Domingues, F; Pereira, L (10 March 2017). "Can Cranberries Contribute to Reduce the Incidence of Urinary Tract Infections? A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Clinical Trials". Journal of Urology. 198 (3): 614–621. doi:10.1016/j.juro.2017.03.078. PMID 28288837. S2CID 206632675.
- ↑ "Recurrent Uncomplicated Urinary Tract Infections in Women: AUA/CUA/SUFU Guideline (2019)". www.auanet.org. American Urological Association. 2019. Retrieved 2019-11-11.
- ↑ Corder, R.; Mullen, W.; Khan, N. Q.; Marks, S. C.; Wood, E. G.; Carrier, M. J.; Crozier, A. (2006). "Oenology: Red wine procyanidins and vascular health". Nature. 444 (7119): 566. Bibcode:2006Natur.444..566C. doi: 10.1038/444566a . PMID 17136085. S2CID 4303406.
- ↑ Absalon, C; Fabre, S; Tarascou, I; Fouquet, E; Pianet, I (2011). "New strategies to study the chemical nature of wine oligomeric procyanidins". Analytical and Bioanalytical Chemistry. 401 (5): 1485–1495. doi:10.1007/s00216-011-4988-1. PMID 21573848. S2CID 8145537.
- ↑ Gonzalo-Diago, A; Dizy, M; Fernández-Zurbano, P (2013). "Taste and mouthfeel properties of red wines proanthocyanidins and their relation to the chemical composition". Journal of Agricultural and Food Chemistry. 61 (37): 8861–8870. doi:10.1021/jf401041q. hdl:10261/144130. PMID 23889258.
- ↑ Sánchez-Moreno, Concepción; Cao, Guohua; Ou, Boxin; Prior, Ronald L. (2003). "Anthocyanin and Proanthocyanidin Content in Selected White and Red Wines. Oxygen Radical Absorbance Capacity Comparison with Nontraditional Wines Obtained from Highbush Blueberry". Journal of Agricultural and Food Chemistry. 51 (17): 4889–4896. doi:10.1021/jf030081t. PMID 12903941.
- ↑ Robertson, Nina U.; Schoonees, Anel; Brand, Amanda; Visser, Janicke (29 September 2020). "Pine bark (Pinus spp.) extract for treating chronic disorders". The Cochrane Database of Systematic Reviews. 2020 (9): CD008294. doi:10.1002/14651858.CD008294.pub5. ISSN 1469-493X. PMC 8094515 . PMID 32990945.
- 1 2 Wu X, Gu L, Prior RL, McKay S (2004). "Characterization of anthocyanins and proanthocyanidins in some cultivars of Ribes, Aronia, and Sambucus and their antioxidant capacity". Journal of Agricultural and Food Chemistry. 52 (26): 7846–7856. doi:10.1021/jf0486850. PMID 15612766.