IARC group 2B substances, mixtures and exposure circumstances are those that have been classified as "possibly carcinogenic to humans" by the International Agency for Research on Cancer (IARC) as This category is used when there is limited evidence of carcinogenicity in humans and less than sufficient evidence of carcinogenicity in experimental animals. It may also be used when there is insufficient evidence of carcinogenicity in humans but sufficient evidence in experimental animals. In some cases, an agent, mixture, or exposure circumstance with inadequate evidence of carcinogenicity in humans but limited evidence in experimental animals, combined with supporting evidence from other relevant data, may be included in this group.
2,4-Dinitrophenylhydrazine (2,4-DNPH or DNPH) is the organic compound C6H3(NO2)2NHNH2. DNPH is a red to orange solid. It is a substituted hydrazine. The solid is relatively sensitive to shock and friction. For this reason DNPH is usually handled as a wet powder. DNPH is a precursor to the drug Sivifene.

The Knorr pyrrole synthesis is a widely used chemical reaction that synthesizes substituted pyrroles (3). The method involves the reaction of an α-amino-ketone (1) and a compound containing an electron-withdrawing group α to a carbonyl group (2).

Amitraz is a non-systemic acaricide and insecticide and has also been described as a scabicide. It was first synthesized by the Boots Co. in England in 1969. Amitraz has been found to have an insect repellent effect, works as an insecticide and also as a pesticide synergist. Its effectiveness is traced back on alpha-adrenergic agonist activity, interaction with octopamine receptors of the central nervous system and inhibition of monoamine oxidases and prostaglandin synthesis. Therefore, it leads to overexcitation and consequently paralysis and death in insects. Because amitraz is less harmful to mammals, amitraz is among many other purposes best known as insecticide against mite- or tick-infestation of dogs. It is also widely used in the beekeeping industry as a control for the Varroa destructor mite, although there are recent reports of resistance.

Phenoxy herbicides are two families of chemicals that have been developed as commercially important herbicides, widely used in agriculture. They share the part structure of phenoxyacetic acid.
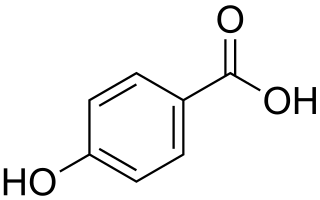
4-Hydroxybenzoic acid, also known as p-hydroxybenzoic acid (PHBA), is a monohydroxybenzoic acid, a phenolic derivative of benzoic acid. It is a white crystalline solid that is slightly soluble in water and chloroform but more soluble in polar organic solvents such as alcohols and acetone. 4-Hydroxybenzoic acid is primarily known as the basis for the preparation of its esters, known as parabens, which are used as preservatives in cosmetics and some ophthalmic solutions. It is isomeric with 2-hydroxybenzoic acid, known as salicylic acid, a precursor to aspirin, and with 3-hydroxybenzoic acid.

2,4-Dimethyl-6-tert-butylphenol is the organic compound with the formula Me2(tert-Bu)C6H2OH (Me = methyl, tert-Bu = tertiary butyl). It is a colorless oil that is classified as an alkylated phenol.

N-Morpholinyllysergamide (LSM-775) is a derivative of ergine. It is less potent than LSD but is reported to have some LSD-like effects at doses ranging from 75 to 700 micrograms and a shorter duration. There are fewer signs of cardiovascular stimulation and peripheral toxicity with LSM-775 compared to LSD.

2,4 Dienoyl-CoA reductase also known as DECR1 is an enzyme which in humans is encoded by the DECR1 gene which resides on chromosome 8. This enzyme catalyzes the following reactions

Dichlorprop is a chlorophenoxy herbicide similar in structure to 2,4-D that is used to kill annual and perennial broadleaf weeds. It is a component of many common weedkillers. About 4 million pounds of dichlorprop are used annually in the United States.

1-Fluoro-2,4-dinitrobenzene is a chemical that reacts with the N-terminal amino acid of polypeptides. This can be helpful for sequencing proteins.
In enzymology, a 2,4-dichlorobenzoyl-CoA reductase (EC 1.3.1.63) is an enzyme that catalyzes the chemical reaction
2,4-Dichlorophenol (2,4-DCP) is a chlorinated derivative of phenol with the molecular formula Cl2C6H3OH. It is a white solid that is mildly acidic (pKa = 7.9). It is produced on a large scale as a precursor to the herbicide 2,4-dichlorophenoxyacetic acid (2,4-D).
2,4-DB or 4-(2,4-dichlorophenoxy)butyric acid is a selective systemic phenoxy herbicide used to control many annual and perennial broad-leaf weeds in alfalfa, peanuts, soybeans, and other crops. Its active metabolite, 2,4-D, inhibits growth at the tips of stems and roots. It is classified in toxicity class III. It shows some evidence of toxicity to dogs and cats, such as changes in body weight and reduced numbers of offspring, when fed 25-80 milligrams per kilogram of body weight for prolonged periods. Tests of carcinogenicity in this range yielded differing results. It is moderately toxic to fish.

Lysergic acid 2,4-dimethylazetidide (LA-SS-Az, LSZ) is an analog of LSD developed by the team led by David E. Nichols at Purdue University. It was developed as a rigid analog of LSD with the diethylamide group constrained into an azetidine ring in order to map the binding site at the 5-HT2A receptor. There are three possible stereoisomers around the azetidine ring, with the (S,S)-(+) isomer being the most active, slightly more potent than LSD itself in drug discrimination tests using trained rats.
This is an index of articles relating to pesticides.
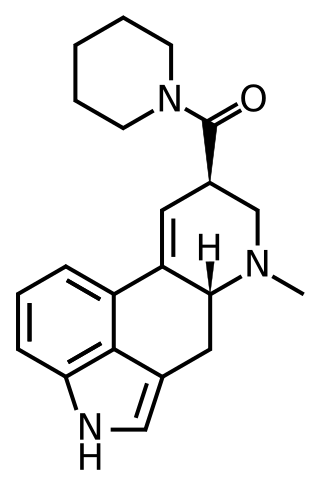
LSD-Pip is a compound from the ergoline family, related to LSD but with the N,N-diethyl substitution replaced by a piperidine group. It is more potent than the corresponding pyrrolidine and morpholine analogues, but is still several times less potent than LSD as a 5-HT2A agonist. Early studies suggested this compound to be inactive as a psychedelic in humans, though this does not seem to have been confirmed by any more recent work.

2,4-Dichlorophenoxyacetic acid is an organic compound with the chemical formula Cl2C6H3OCH2CO2H. It is usually referred to by its ISO common name 2,4-D. It is a systemic herbicide that kills most broadleaf weeds by causing uncontrolled growth, but most grasses such as cereals, lawn turf, and grassland are relatively unaffected.

Umbellic acid is a hydroxycinnamic acid. It is an isomer of caffeic acid.

Pelanserin (TR2515) is a chemical compound that acts as an antagonist of the 5-HT2 and α1-adrenergic receptors.
















