
Ascorbic acid is an organic compound with formula C
6H
8O
6, originally called hexuronic acid. It is a white solid, but impure samples can appear yellowish. It dissolves freely in water to give mildly acidic solutions. It is a mild reducing agent.

Food additives are substances added to food to preserve flavor or enhance taste, appearance, or other sensory qualities. Some additives, such as vinegar (pickling), salt (salting), smoke (smoking) and sugar (crystallization), have been used for centuries to preserve food. This allows for longer-lasting foods, such as bacon, sweets or wines.

Vitamin C is a water-soluble vitamin found in citrus and other fruits, berries and vegetables. It is also a generic prescription medication and in some countries is sold as a non-prescription dietary supplement. As a therapy, it is used to prevent and treat scurvy, a disease caused by vitamin C deficiency.

E numbers, short for Europe numbers, are codes for substances used as food additives, including those found naturally in many foods, such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly found on food labels, their safety assessment and approval are the responsibility of the European Food Safety Authority (EFSA). The fact that an additive has an E number implies that its use was at one time permitted in products for sale in the European Single Market; some of these additives are no longer allowed today.

Olestra is a fat substitute food additive that adds no metabolizable calories to products. It has been used in the preparation of otherwise high-fat foods, thereby lowering or eliminating their fat content.
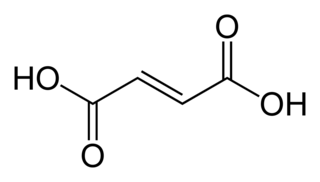
Fumaric acid or trans-butenedioic acid is an organic compound with the formula HO2CCH=CHCO2H. A white solid, fumaric acid occurs widely in nature. It has a fruit-like taste and has been used as a food additive. Its E number is E297. The salts and esters are known as fumarates. Fumarate can also refer to the C
4H
2O2−
4 ion (in solution). Fumaric acid is the trans isomer of butenedioic acid, while maleic acid is the cis isomer.

Canthaxanthin is a keto-carotenoid pigment widely distributed in nature. Carotenoids belong to a larger class of phytochemicals known as terpenoids. The chemical formula of canthaxanthin is C40H52O2. It was first isolated in edible mushrooms. It has also been found in green algae, bacteria, crustaceans, and bioaccumulates in fish such as carp, golden grey mullet, seabream and trush wrasse.

Erythorbic acid is a stereoisomer of ascorbic acid. It is synthesized by a reaction between methyl 2-keto-D-gluconate and sodium methoxide. It can also be synthesized from sucrose or by strains of Penicillium that have been selected for this feature. It is denoted by E number E315, and is widely used as an antioxidant in processed foods.

Sodium propanoate or sodium propionate is the sodium salt of propionic acid which has the chemical formula Na(C2H5COO). This white crystalline solid is deliquescent in moist air.
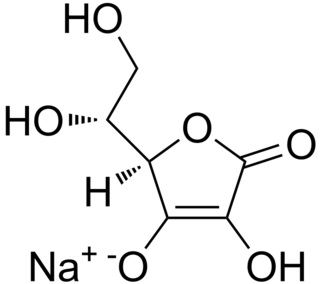
Sodium erythorbate (C6H7NaO6) is a food additive used predominantly in meats, poultry, and soft drinks. Chemically, it is the sodium salt of erythorbic acid.

Apocarotenal, or trans-β-apo-8'-carotenal, is a carotenoid found in spinach and citrus fruits. Like other carotenoids, apocarotenal plays a role as a precursor of vitamin A, even though it has 50% less pro-vitamin A activity than β-carotene. The empirical chemical formula for apocarotenal is C30H40O.

Mineral ascorbates are a group of salts of ascorbic acid. They are composed of a mineral cation bonded to ascorbate.
Ascorbyl stearate (C24H42O7) is an ester formed from ascorbic acid and stearic acid. In addition to its use as a source of vitamin C, it is used as an antioxidant food additive in margarine (E number E305). The USDA limits its use to 0.02% individually or in conjunction with other antioxidants.

Calcium benzoate refers to the calcium salt of benzoic acid. When used in the food industry as a preservative, its E number is E213 ; it is approved for use as a food additive in the EU, USA and Australia and New Zealand.

Calcium ascorbate is a compound with the molecular formula CaC12H14O12. It is the calcium salt of ascorbic acid, one of the mineral ascorbates. It is approximately 10% calcium by mass.

Sodium ascorbate is one of a number of mineral salts of ascorbic acid (vitamin C). The molecular formula of this chemical compound is C6H7NaO6. As the sodium salt of ascorbic acid, it is known as a mineral ascorbate. It has not been demonstrated to be more bioavailable than any other form of vitamin C supplement.

Potassium ascorbate is a compound with formula KC6H7O6. It is the potassium salt of ascorbic acid (vitamin C) and a mineral ascorbate. As a food additive, it has E number E303, INS number 303. Although it is not a permitted food additive in the UK, USA and the EU, it is approved for use in Australia and New Zealand. According to some studies, it has shown a strong antioxidant activity and antitumoral properties.
Benzene in soft drinks is of potential concern due to the carcinogenic nature of the molecule. This contamination is a public health concern and has caused significant outcry among environmental and health advocates. Benzene levels are regulated in drinking water nationally and internationally, and in bottled water in the United States, but only informally in soft drinks. The benzene forms from decarboxylation of the preservative benzoic acid in the presence of ascorbic acid and metal ions that act as catalysts, especially under heat and light. Hot peppers naturally contain vitamin C so the observation about soft drinks applies to pepper sauces containing sodium benzoate, like Texas Pete.

A dough conditioner, flour treatment agent, improving agent or bread improver is any ingredient or chemical added to bread dough to strengthen its texture or otherwise improve it in some way. Dough conditioners may include enzymes, yeast nutrients, mineral salts, oxidants and reductants, bleaching agents and emulsifiers. They are food additives combined with flour to improve baking functionality. Flour treatment agents are used to increase the speed of dough rising and to improve the strength and workability of the dough.
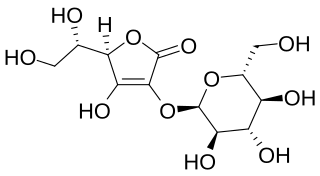
Ascorbyl glucoside (AA-2G) is an ascorbic acid derivative that contains at least one glycosyl group. Ascorbyl glucoside is commonly used in cosmetic products to administer vitamin C topically. Ascorbyl glucoside exhibits superior stability and penetration ability compared to ascorbyl phosphate salts, but the rate of its in vivo conversion to ascorbic acid is not known. Ascorbyl glucosides such as AA-2G, like many other derivatives of the ascorbic acid, show antiscorbutic effects. It is also sometimes used in skin whitening products.
















