
In chemistry, an ester is a compound derived from an acid in which the hydrogen atom (H) of at least one acidic hydroxyl group of that acid is replaced by an organyl group. These compounds contain a distinctive functional group. Analogues derived from oxygen replaced by other chalcogens belong to the ester category as well. According to some authors, organyl derivatives of acidic hydrogen of other acids are esters as well, but not according to the IUPAC.
Mesitylene or 1,3,5-trimethylbenzene is a derivative of benzene with three methyl substituents positioned symmetrically around the ring. The other two isomeric trimethylbenzenes are 1,2,4-trimethylbenzene (pseudocumene) and 1,2,3-trimethylbenzene (hemimellitene). All three compounds have the formula C6H3(CH3)3, which is commonly abbreviated C6H3Me3. Mesitylene is a colorless liquid with sweet aromatic odor. It is a component of coal tar, which is its traditional source. It is a precursor to diverse fine chemicals. The mesityl group (Mes) is a substituent with the formula C6H2Me3 and is found in various other compounds.

Acetone peroxide is an organic peroxide and a primary explosive. It is produced by the reaction of acetone and hydrogen peroxide to yield a mixture of linear monomer and cyclic dimer, trimer, and tetramer forms. The monomer is dimethyldioxirane. The dimer is known as diacetone diperoxide (DADP). The trimer is known as triacetone triperoxide (TATP) or tri-cyclic acetone peroxide (TCAP). Acetone peroxide takes the form of a white crystalline powder with a distinctive bleach-like odor when impure, or a fruit-like smell when pure, and can explode powerfully if subjected to heat, friction, static electricity, concentrated sulfuric acid, strong UV radiation, or shock. Until about 2015, explosives detectors were not set to detect non-nitrogenous explosives, as most explosives used preceding 2015 were nitrogen-based. TATP, being nitrogen-free, has been used as the explosive of choice in several terrorist bomb attacks since 2001.

Cinnamic acid is an organic compound with the formula C6H5-CH=CH-COOH. It is a white crystalline compound that is slightly soluble in water, and freely soluble in many organic solvents. Classified as an unsaturated carboxylic acid, it occurs naturally in a number of plants. It exists as both a cis and a trans isomer, although the latter is more common.
The Perkin reaction is an organic reaction developed by English chemist William Henry Perkin in 1868 that is used to make cinnamic acids. It gives an α,β-unsaturated aromatic acid or α-substituted β-aryl acrylic acid by the aldol condensation of an aromatic aldehyde and an acid anhydride, in the presence of an alkali salt of the acid. The alkali salt acts as a base catalyst, and other bases can be used instead.
Barbituric acid or malonylurea or 6-hydroxyuracil is an organic compound based on a pyrimidine heterocyclic skeleton. It is an odorless powder soluble in water. Barbituric acid is the parent compound of barbiturate drugs, although barbituric acid itself is not pharmacologically active. The compound was first synthesised by Adolf von Baeyer.
The Claisen condensation is a carbon–carbon bond forming reaction that occurs between two esters or one ester and another carbonyl compound in the presence of a strong base. The reaction produces a β-keto ester or a β-diketone. It is named after Rainer Ludwig Claisen, who first published his work on the reaction in 1887. The reaction has often been displaced by diketene-based chemistry, which affords acetoacetic esters.
In organic chemistry, a cyanohydrin reaction is an organic reaction in which an aldehyde or ketone reacts with a cyanide anion or a nitrile to form a cyanohydrin. For example:
Chloral, also known as trichloroacetaldehyde or trichloroethanal, is the organic compound with the formula Cl3CCHO. This aldehyde is a colourless liquid that is soluble in a wide range of solvents. It reacts with water to form chloral hydrate, a once widely used sedative and hypnotic substance.

Wilhelm Rudolph Fittig was a German chemist. He discovered the pinacol coupling reaction, mesitylene, diacetyl and biphenyl. Fittig studied the action of sodium on ketones and hydrocarbons. He discovered the Fittig reaction or Wurtz–Fittig reaction for the synthesis of alkylbenzenes, he proposed a diketone structure for benzoquinone and isolated phenanthrene from coal tar. He discovered and synthesized the first lactones and investigated structures of piperine, naphthalene, and fluorene.

Benzylideneacetone is the organic compound described by the formula C6H5CH=CHC(O)CH3. Although both cis- and trans-isomers are possible for the α,β-unsaturated ketone, only the trans isomer is observed. Its original preparation demonstrated the scope of condensation reactions to construct new, complex organic compounds. Benzylideneacetone is used as a flavouring ingredient in food and perfumes.
Picoline refers to any of three isomers of methylpyridine (CH3C5H4N). They are all colorless liquids with a characteristic smell similar to that of pyridine. They are miscible with water and most organic solvents.
In organic chemistry, the Claisen–Schmidt condensation is the reaction between an aldehyde or ketone having an α-hydrogen with an aromatic carbonyl compound lacking an α-hydrogen. It can be considered as a specific variation of the aldol condensation. This reaction is named after two of its pioneering investigators Rainer Ludwig Claisen and J. Gustav Schmidt, who independently published on this topic in 1880 and 1881. An example is the synthesis of dibenzylideneacetone ( -1,5-diphenylpenta-1,4-dien-3-one).

The Stobbe condensation entails the reaction of an aldehyde or ketone with an ester of succinic acid to generate alkylidene succinic acid or related derivatives. The reaction consumes one equivalent of metal alkoxide. Commonly, diethylsuccinate is a component of the reaction. The usual product is salt of the half-ester. The Stobbe condensation is named after its discoverer, Hans Stobbe, whose work involved the sodium ethoxide-induced condensation of acetone and diethyl succinate.
Benzaldehyde (C6H5CHO) is an organic compound consisting of a benzene ring with a formyl substituent. It is among the simplest aromatic aldehydes and one of the most industrially useful.

2-Nitrobenzaldehyde is an organic compound with the formula O2NC6H4CHO. It is one of three isomers of nitrobenzaldehyde. It contains a nitro group adjacent to the formyl group.
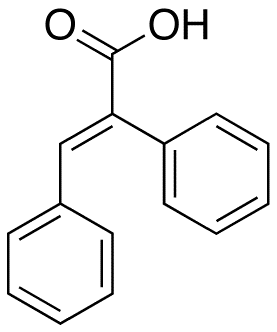
α-Phenylcinnamic acid is a phenylpropanoid, or, more specifically, a derivative of cinnamic acid. It has the formula C15H12O2 and appears as an off-white crystalline solid.

Acetone oxime (acetoxime) is the organic compound with the formula (CH3)2CNOH. It is the simplest example of a ketoxime. It is a white crystalline solid that is soluble in water, ethanol, ether, chloroform, and ligroin. It is used as a reagent in organic synthesis.

Georg Andreas Karl Städeler was a German chemist.













