
The endocrine system is a messenger system in an organism comprising feedback loops of hormones that are released by internal glands directly into the circulatory system and that target and regulate distant organs. In vertebrates, the hypothalamus is the neural control center for all endocrine systems.

The pituitary gland is an endocrine gland in vertebrates. In humans, the pituitary gland is located at the base of the brain, protruding off the bottom of the hypothalamus. The human pituitary gland is oval shaped, about the size of a chickpea, and weighs 0.5 grams (0.018 oz) on average.

Adrenocorticotropic hormone is a polypeptide tropic hormone produced by and secreted by the anterior pituitary gland. It is also used as a medication and diagnostic agent. ACTH is an important component of the hypothalamic-pituitary-adrenal axis and is often produced in response to biological stress. Its principal effects are increased production and release of cortisol and androgens by the zona fasiculata and zona reticularis, respectively. ACTH is also related to the circadian rhythm in many organisms.

The hypothalamus is a small part of the brain that contains a number of nuclei with a variety of functions. One of the most important functions is to link the nervous system to the endocrine system via the pituitary gland. The hypothalamus is located below the thalamus and is part of the limbic system. It forms the ventral part of the diencephalon. All vertebrate brains contain a hypothalamus. In humans, it is the size of an almond.

Prolactin (PRL), also known as lactotropin and mammotropin, is a protein best known for its role in enabling mammals to produce milk. It is influential in over 300 separate processes in various vertebrates, including humans. Prolactin is secreted from the pituitary gland in response to eating, mating, estrogen treatment, ovulation and nursing. It is secreted heavily in pulses in between these events. Prolactin plays an essential role in metabolism, regulation of the immune system and pancreatic development.
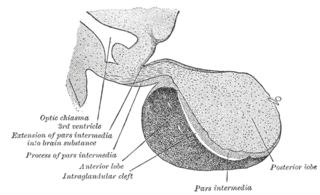
A major organ of the endocrine system, the anterior pituitary is the glandular, anterior lobe that together with the posterior lobe makes up the pituitary gland (hypophysis) which, in humans, is located at the base of the brain, protruding off the bottom of the hypothalamus.

The posterior pituitary is the posterior lobe of the pituitary gland which is part of the endocrine system. The posterior pituitary is not glandular as is the anterior pituitary. Instead, it is largely a collection of axonal projections from the hypothalamus that terminate behind the anterior pituitary, and serve as a site for the secretion of neurohypophysial hormones directly into the blood. The hypothalamic–neurohypophyseal system is composed of the hypothalamus, posterior pituitary, and these axonal projections.
A lactotropic cell is a cell in the anterior pituitary which produces prolactin in response to hormonal signals including dopamine, thyrotropin-releasing hormone and estrogen, which are stimulatory. Prolactin is responsible for actions needed for body homeostasis, the development of breasts, and for lactation. The inhibitory effects of dopamine override the stimulatory effects of TRH in non-pregnant, non-lactating sexually mature females. Depending on the sex of the individual, lactotropic cells account for 20% - 50% of all cells in the anterior pituitary gland. The inhibitory effects of dopamine override the stimulatory effects of TRH in non-pregnant, non-lactating sexually mature females. Other regulators include oxytocin and progesterone.
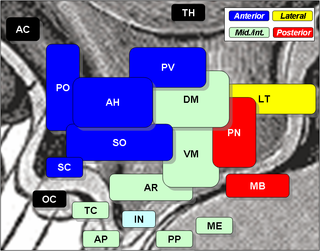
The arcuate nucleus of the hypothalamus is an aggregation of neurons in the mediobasal hypothalamus, adjacent to the third ventricle and the median eminence. The arcuate nucleus includes several important and diverse populations of neurons that help mediate different neuroendocrine and physiological functions, including neuroendocrine neurons, centrally projecting neurons, and astrocytes. The populations of neurons found in the arcuate nucleus are based on the hormones they secrete or interact with and are responsible for hypothalamic function, such as regulating hormones released from the pituitary gland or secreting their own hormones. Neurons in this region are also responsible for integrating information and providing inputs to other nuclei in the hypothalamus or inputs to areas outside this region of the brain. These neurons, generated from the ventral part of the periventricular epithelium during embryonic development, locate dorsally in the hypothalamus, becoming part of the ventromedial hypothalamic region. The function of the arcuate nucleus relies on its diversity of neurons, but its central role is involved in homeostasis. The arcuate nucleus provides many physiological roles involved in feeding, metabolism, fertility, and cardiovascular regulation.

Pituitary adenomas are tumors that occur in the pituitary gland. Most pituitary tumors are benign, approximately 35% are invasive and just 0.1% to 0.2% are carcinomas. Pituitary adenomas represent from 10% to 25% of all intracranial neoplasms and the estimated prevalence rate in the general population is approximately 17%.

Endocrine glands are ductless glands of the endocrine system that secrete their products, hormones, directly into the blood. The major glands of the endocrine system include the pineal gland, pituitary gland, pancreas, ovaries, testicles, thyroid gland, parathyroid gland, hypothalamus and adrenal glands. The hypothalamus and pituitary glands are neuroendocrine organs.
Neuroendocrinology is the branch of biology which studies the interaction between the nervous system and the endocrine system; i.e. how the brain regulates the hormonal activity in the body. The nervous and endocrine systems often act together in a process called neuroendocrine integration, to regulate the physiological processes of the human body. Neuroendocrinology arose from the recognition that the brain, especially the hypothalamus, controls secretion of pituitary gland hormones, and has subsequently expanded to investigate numerous interconnections of the endocrine and nervous systems.

Multiple endocrine neoplasia type 1 (MEN-1) is one of a group of disorders, the multiple endocrine neoplasias, that affect the endocrine system through development of neoplastic lesions in pituitary, parathyroid gland and pancreas. Individuals suffering from this disorder are prone to developing multiple endocrine and nonendocrine tumors. It was first described by Paul Wermer in 1954.
The prolactin receptor (PRLR) is a type I cytokine receptor encoded in humans by the PRLR gene on chromosome 5p13-14. It is the receptor for prolactin (PRL). The PRLR can also bind to and be activated by growth hormone (GH) and human placental lactogen (hPL). The PRLR is expressed in the mammary glands, pituitary gland, and other tissues. It plays an important role in lobuloalveolar development of the mammary glands during pregnancy and in lactation.

The hypophyseal portal system is a system of blood vessels in the microcirculation at the base of the brain, connecting the hypothalamus with the anterior pituitary. Its main function is to quickly transport and exchange hormones between the hypothalamus arcuate nucleus and anterior pituitary gland. The capillaries in the portal system are fenestrated which allows a rapid exchange between the hypothalamus and the pituitary. The main hormones transported by the system include gonadotropin-releasing hormone, corticotropin-releasing hormone, growth hormone–releasing hormone, and thyrotropin-releasing hormone.
Stress hormones are secreted by endocrine glands to modify one's internal environment during the times of stress. By performing various functions such as mobilizing energy sources, increasing heart rate, and downregulating metabolic processes which are not immediately necessary, stress hormones promote the survival of the organism. The secretions of some hormones are also downplayed during stress. Stress hormones include, but are not limited to:
Non-tropic hormones are hormones that directly stimulate target cells to induce effects. This differs from the tropic hormones, which act on another endocrine gland. Non-tropic hormones are those that act directly on targeted tissues or cells, and not on other endocrine gland to stimulate release of other hormones. Many hormones act in a chain reaction. Tropic hormones usually act in the beginning of the reaction stimulating other endocrine gland to eventually release non-tropic hormones. These are the ones that act in the end of the chain reaction on other cells that are not part of other endocrine gland. The Hypothalamic-pituitary-adrenal axis is a perfect example of this chain reaction. The reaction begins in the hypothalamus with a release of corticotropin-releasing hormone/factor. This stimulates the anterior pituitary and causes it to release Adrenocorticotropic hormone to the adrenal glands. Lastly, cortisol (non-tropic) is secreted from the adrenal glands and goes into the bloodstream where it can have more widespread effects on organs and tissues. Since cortisol is what finally reaches other tissues in the body, it is a non-tropic hormone. CRH and ACTH are tropic hormones because they act on the anterior pituitary gland and adrenal glands, respectively, both of which are endocrine glands. Non-tropic hormones are thus often the last piece of a larger process and chain of hormone secretion. Both tropic and non-tropic hormones are necessary for proper endocrine function. For example, if ACTH is inhibited, cortisol can no longer be released because the chain reaction has been interrupted. Some examples of non-tropic hormones are:
Hypothalamic–pituitary hormones are hormones that are produced by the hypothalamus and pituitary gland. Although the organs in which they are produced are relatively small, the effects of these hormones cascade throughout the body. They can be classified as a hypothalamic–pituitary axis of which the adrenal, gonadal, thyroid, somatotropic, and prolactin axes are branches.
Hypogonadotropic hypogonadism (HH), is due to problems with either the hypothalamus or pituitary gland affecting the hypothalamic-pituitary-gonadal axis. Hypothalamic disorders result from a deficiency in the release of gonadotropic releasing hormone (GnRH), while pituitary gland disorders are due to a deficiency in the release of gonadotropins from the anterior pituitary. GnRH is the central regulator in reproductive function and sexual development via the HPG axis. GnRH is released by GnRH neurons, which are hypothalamic neuroendocrine cells, into the hypophyseal portal system acting on gonadotrophs in the anterior pituitary. The release of gonadotropins, LH and FSH, act on the gonads for the development and maintenance of proper adult reproductive physiology. LH acts on Leydig cells in the male testes and theca cells in the female. FSH acts on Sertoli cells in the male and follicular cells in the female. Combined this causes the secretion of gonadal sex steroids and the initiation of folliculogenesis and spermatogenesis. The production of sex steroids forms a negative feedback loop acting on both the anterior pituitary and hypothalamus causing a pulsatile secretion of GnRH. GnRH neurons lack sex steroid receptors and mediators such as kisspeptin stimulate GnRH neurons for pulsatile secretion of GnRH.
Pulsatile secretion is a biochemical phenomenon observed in a wide variety of cell and tissue types, in which chemical products are secreted in a regular temporal pattern. The most common cellular products observed to be released in this manner are intercellular signaling molecules such as hormones or neurotransmitters. Examples of hormones that are secreted pulsatilely include insulin, thyrotropin, TRH, gonadotropin-releasing hormone (GnRH) and growth hormone (GH). In the nervous system, pulsatility is observed in oscillatory activity from central pattern generators. In the heart, pacemakers are able to work and secrete in a pulsatile manner. A pulsatile secretion pattern is critical to the function of many hormones in order to maintain the delicate homeostatic balance necessary for essential life processes, such as development and reproduction. Variations of the concentration in a certain frequency can be critical to hormone function, as evidenced by the case of GnRH agonists, which cause functional inhibition of the receptor for GnRH due to profound downregulation in response to constant (tonic) stimulation. Pulsatility may function to sensitize target tissues to the hormone of interest and upregulate receptors, leading to improved responses. This heightened response may have served to improve the animal's fitness in its environment and promote its evolutionary retention.











