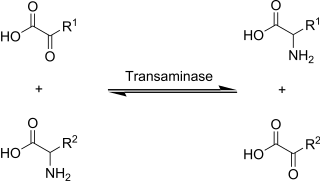
Transamination is a chemical reaction that transfers an amino group to a ketoacid to form new amino acids. This pathway is responsible for the deamination of most amino acids. This is one of the major degradation pathways which convert essential amino acids to non-essential amino acids.

Aspartate transaminase (AST) or aspartate aminotransferase, also known as AspAT/ASAT/AAT or (serum) glutamic oxaloacetic transaminase, is a pyridoxal phosphate (PLP)-dependent transaminase enzyme that was first described by Arthur Karmen and colleagues in 1954. AST catalyzes the reversible transfer of an α-amino group between aspartate and glutamate and, as such, is an important enzyme in amino acid metabolism. AST is found in the liver, heart, skeletal muscle, kidneys, brain, red blood cells and gall bladder. Serum AST level, serum ALT level, and their ratio are commonly measured clinically as biomarkers for liver health. The tests are part of blood panels.

Oxaloacetic acid (also known as oxalacetic acid or OAA) is a crystalline organic compound with the chemical formula HO2CC(O)CH2CO2H. Oxaloacetic acid, in the form of its conjugate base oxaloacetate, is a metabolic intermediate in many processes that occur in animals. It takes part in gluconeogenesis, the urea cycle, the glyoxylate cycle, amino acid synthesis, fatty acid synthesis and the citric acid cycle.

Transaminases or aminotransferases are enzymes that catalyze a transamination reaction between an amino acid and an α-keto acid. They are important in the synthesis of amino acids, which form proteins.

Amino acid synthesis is the set of biochemical processes by which the amino acids are produced. The substrates for these processes are various compounds in the organism's diet or growth media. Not all organisms are able to synthesize all amino acids. For example, humans can synthesize 11 of the 20 standard amino acids. These 11 are called the non-essential amino acids).
In medicine, the presence of elevated transaminases, commonly the transaminases alanine transaminase (ALT) and aspartate transaminase (AST), may be an indicator of liver dysfunction. Other terms include transaminasemia, transaminitis, and elevatedliver enzymes. Normal ranges for both ALT and AST vary by gender, age, and geography and are roughly 8-40 U/L. Mild transaminesemia refers to levels up to 250 U/L. Drug-induced increases such as that found with the use of anti-tuberculosis agents such as isoniazid are limited typically to below 100 U/L for either ALT or AST. Muscle sources of the enzymes, such as intense exercise, are unrelated to liver function and can markedly increase AST and ALT. Cirrhosis of the liver or fulminant liver failure secondary to hepatitis commonly reach values for both ALT and AST in the >1000 U/L range. Elevated transaminases that persist less than six months are termed "acute" in nature, and those values that persist for six months or more are termed "chronic" in nature.

In enzymology, 4-aminobutyrate transaminase, also called GABA transaminase or 4-aminobutyrate aminotransferase, or GABA-T, is an enzyme that catalyzes the chemical reaction:
In enzymology, an alanine-glyoxylate transaminase is an enzyme that catalyzes the chemical reaction
In enzymology, an aromatic-amino-acid-glyoxylate transaminase is an enzyme that catalyzes the chemical reaction
In enzymology, an aspartate-phenylpyruvate transaminase is an enzyme that catalyzes the chemical reaction
In enzymology, an aspartate-prephenate aminotransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a diaminobutyrate-2-oxoglutarate transaminase is an enzyme that catalyzes the chemical reaction
In enzymology, a glycine transaminase is an enzyme that catalyzes the chemical reaction
In enzymology, a kynurenine-glyoxylate transaminase is an enzyme that catalyzes the chemical reaction:
In enzymology, a methionine-glyoxylate transaminase is an enzyme that catalyzes the chemical reaction
In enzymology, a pyridoxamine-oxaloacetate transaminase is an enzyme that catalyzes the chemical reaction
In enzymology, a serine-glyoxylate transaminase is an enzyme that catalyzes the chemical reaction

Aspartate aminotransferase, mitochondrial is an enzyme that in humans is encoded by the GOT2 gene. Glutamic-oxaloacetic transaminase is a pyridoxal phosphate-dependent enzyme which exists in cytoplasmic and inner-membrane mitochondrial forms, GOT1 and GOT2, respectively. GOT plays a role in amino acid metabolism and the urea and Kreb's cycle. Also, GOT2 is a major participant in the malate-aspartate shuttle, which is a passage from the cytosol to the mitochondria. The two enzymes are homodimeric and show close homology. GOT2 has been seen to have a role in cell proliferation, especially in terms of tumor growth.

Aspartate aminotransferase, cytoplasmic is an enzyme that in humans is encoded by the GOT1 gene.
4-aminobutyrate---pyruvate transaminase is an enzyme with systematic name 4-aminobutanoate:pyruvate aminotransferase. This enzyme is a type of GABA transaminase, which degrades the neurotransmitter GABA. The enzyme catalyses the following chemical reaction








