
Fluorescence is one of two kinds of emission of light by a substance that has absorbed light or other electromagnetic radiation. When exposed to ultraviolet radiation, many substances will glow (fluoresce) with colored visible light. The color of the light emitted depends on the chemical composition of the substance. Fluorescent materials generally cease to glow nearly immediately when the radiation source stops. This distinguishes them from the other type of light emission, phosphorescence. Phosphorescent materials continue to emit light for some time after the radiation stops.

Photoluminescence is light emission from any form of matter after the absorption of photons. It is one of many forms of luminescence and is initiated by photoexcitation, hence the prefix photo-. Following excitation, various relaxation processes typically occur in which other photons are re-radiated. Time periods between absorption and emission may vary: ranging from short femtosecond-regime for emission involving free-carrier plasma in inorganic semiconductors up to milliseconds for phosphoresence processes in molecular systems; and under special circumstances delay of emission may even span to minutes or hours.

Phosphorescence is a type of photoluminescence related to fluorescence. When exposed to light (radiation) of a shorter wavelength, a phosphorescent substance will glow, absorbing the light and reemitting it at a longer wavelength. Unlike fluorescence, a phosphorescent material does not immediately reemit the radiation it absorbs. Instead, a phosphorescent material absorbs some of the radiation energy and reemits it for a much longer time after the radiation source is removed.

Photochemistry is the branch of chemistry concerned with the chemical effects of light. Generally, this term is used to describe a chemical reaction caused by absorption of ultraviolet, visible (400–750 nm), or infrared radiation (750–2500 nm).

Photodynamic therapy (PDT) is a form of phototherapy involving light and a photosensitizing chemical substance used in conjunction with molecular oxygen to elicit cell death (phototoxicity).

Stokes shift is the difference between positions of the band maxima of the absorption and emission spectra of the same electronic transition. It is named after Irish physicist George Gabriel Stokes.
CIDNP, often pronounced like "kidnip", is a nuclear magnetic resonance (NMR) technique that is used to study chemical reactions that involve radicals. It detects the non-Boltzmann (non-thermal) nuclear spin state distribution produced in these reactions as enhanced absorption or emission signals.

Internal conversion is a transition from a higher to a lower electronic state in a molecule or atom. It is sometimes called "radiationless de-excitation", because no photons are emitted. It differs from intersystem crossing in that, while both are radiationless methods of de-excitation, the molecular spin state for internal conversion remains the same, whereas it changes for intersystem crossing. The energy of the electronically excited state is given off to vibrational modes of the molecule. The excitation energy is transformed into heat.
In particle physics, the quantum yield of a radiation-induced process is the number of times a specific event occurs per photon absorbed by the system.
Phosphorescent organic light-emitting diodes (PHOLED) are a type of organic light-emitting diode (OLED) that use the principle of phosphorescence to obtain higher internal efficiencies than fluorescent OLEDs. This technology is currently under development by many industrial and academic research groups.

Photosensitizers are light absorbers that alter the course of a photochemical reaction. They usually are catalysts. They can function by many mechanisms, sometimes they donate an electron to the substrate, sometimes they abstract a hydrogen atom from the substrate. At the end of this process, the photosensitizer returns to its ground state, where it remains chemically intact, poised to absorb more light. One branch of chemistry which frequently utilizes photosensitizers is polymer chemistry, using photosensitizers in reactions such as photopolymerization, photocrosslinking, and photodegradation. Photosensitizers are also used to generate prolonged excited electronic states in organic molecules with uses in photocatalysis, photon upconversion and photodynamic therapy. Generally, photosensitizers absorb electromagnetic radiation consisting of infrared radiation, visible light radiation, and ultraviolet radiation and transfer absorbed energy into neighboring molecules. This absorption of light is made possible by photosensitizers' large de-localized π-systems, which lowers the energy of HOMO and LUMO orbitals to promote photoexcitation. While many photosensitizers are organic or organometallic compounds, there are also examples of using semiconductor quantum dots as photosensitizers.

In chemistry, quenching refers to any process which decreases the fluorescent intensity of a given substance. A variety of processes can result in quenching, such as excited state reactions, energy transfer, complex-formation and collisions. As a consequence, quenching is often heavily dependent on pressure and temperature. Molecular oxygen, iodine ions and acrylamide are common chemical quenchers. The chloride ion is a well known quencher for quinine fluorescence. Quenching poses a problem for non-instant spectroscopic methods, such as laser-induced fluorescence.

Mostafa A. El-Sayed is an Egyptian-American physical chemist, nanoscience researcher, member of the National Academy of Sciences and US National Medal of Science laureate. He is known for the spectroscopy rule named after him, the El-Sayed rule.

In molecular spectroscopy, a Jablonski diagram is a diagram that illustrates the electronic states and often the vibrational levels of a molecule, and also the transitions between them. The states are arranged vertically by energy and grouped horizontally by spin multiplicity. Nonradiative transitions are indicated by squiggly arrows and radiative transitions by straight arrows. The vibrational ground states of each electronic state are indicated with thick lines, the higher vibrational states with thinner lines. The diagram is named after the Polish physicist Aleksander Jabłoński who first proposed it in 1933.

Fluorescence is used in the life sciences generally as a non-destructive way of tracking or analysing biological molecules. Some proteins or small molecules in cells are naturally fluorescent, which is called intrinsic fluorescence or autofluorescence. The intrinsic DNA fluorescence is very weak.Alternatively, specific or general proteins, nucleic acids, lipids or small molecules can be "labelled" with an extrinsic fluorophore, a fluorescent dye which can be a small molecule, protein or quantum dot. Several techniques exist to exploit additional properties of fluorophores, such as fluorescence resonance energy transfer, where the energy is passed non-radiatively to a particular neighbouring dye, allowing proximity or protein activation to be detected; another is the change in properties, such as intensity, of certain dyes depending on their environment allowing their use in structural studies.
Singlet fission is a spin-allowed process, unique to molecular photophysics, whereby one singlet excited state is converted into two triplet states. The phenomenon has been observed in molecular crystals, aggregates, disordered thin films, and covalently-linked dimers, where the chromophores are oriented such that the electronic coupling between singlet and the double triplet states is large. Being spin allowed, the process can occur very rapidly and out-compete radiative decay thereby producing two triplets with very high efficiency. The process is distinct from intersystem crossing, in that singlet fission does not involve a spin flip, but is mediated by two triplets coupled into an overall singlet. It has been proposed that singlet fission in organic photovoltaic devices could improve the photoconversion efficiencies.

9-Fluorenylidene is an aryl carbene derived from the bridging methylene group of fluorene. Fluorenylidene has the unusual property that the triplet ground state is only 1.1 kcal/mol lower in energy than the singlet state. For this reason, fluorenylidene has been studied extensively in organic chemistry.
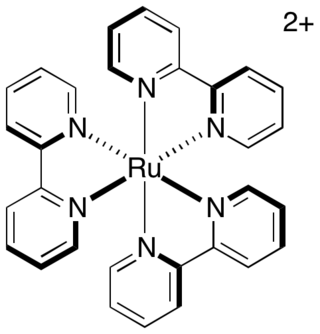
Photoredox catalysis is a branch of photochemistry that uses single-electron transfer. Photoredox catalysts are generally drawn from three classes of materials: transition-metal complexes, organic dyes, and semiconductors. While organic photoredox catalysts were dominant throughout the 1990s and early 2000s, soluble transition-metal complexes are more commonly used today.

Triplet-triplet annihilation (TTA) is an energy transfer mechanism where two molecules in their triplet excited states interact to form a ground state molecule and an excited molecule in its singlet state. This mechanism is example of Dexter energy transfer mechanism. In triplet-triplet annihilation, one molecule transfers its excited state energy to the second molecule, resulting in the first molecule returning to its ground state and the second molecule being promoted to a higher excited singlet state.
Thermally activated delayed fluorescence (TADF) is a process through which surrounding thermal energy changes population of excited states of molecular compounds and thus, alters light emission. The TADF process usually involves an excited molecular species in a triplet state, which commonly has a forbidden transition to the singlet ground state, termed phosphorescence. By absorbing nearby thermal energy, the triplet state can undergo reverse intersystem crossing (RISC) converting the triplet state population to an excited singlet state, which then emits light to the singlet ground state in a delayed process termed delayed fluorescence. Accordingly, in many cases, the TADF molecules show two types of emission, a delayed fluorescence and a prompt fluorescence. This is found for specific organic molecules, but also for selected organo-transition metal compounds, such as Cu(I) complexes. Along with traditional fluorescent molecules and phosphorescent molecules, TADF compounds belong to the three main light-emitting material groups used in organic light-emitting diodes (OLEDs).















