
An oxide is a chemical compound that contains at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion of oxygen, an O2– (molecular) ion. Metal oxides thus typically contain an anion of oxygen in the oxidation state of −2. Most of the Earth's crust consists of solid oxides, the result of elements being oxidized by the oxygen in air or in water. Even materials considered pure elements often develop an oxide coating. For example, aluminium foil develops a thin skin of Al2O3 (called a passivation layer) that protects the foil from further corrosion. Certain elements can form multiple oxides, differing in the amounts of the element combining with the oxygen. Examples are carbon, iron, nitrogen (see nitrogen oxide), silicon, titanium, lithium, and aluminium. In such cases the oxides are distinguished by specifying the numbers of atoms involved, as in carbon monoxide and carbon dioxide, or by specifying the element's oxidation number, as in iron(II) oxide and iron(III) oxide.

Titanite, or sphene (from the Greek sphenos (σφηνώ), meaning wedge), is a calcium titanium nesosilicate mineral, CaTiSiO5. Trace impurities of iron and aluminium are typically present. Also commonly present are rare earth metals including cerium and yttrium; calcium may be partly replaced by thorium.

Calcium carbonate is a chemical compound with the formula CaCO3. It is a common substance found in rocks as the minerals calcite and aragonite (most notably as limestone, which is a type of sedimentary rock consisting mainly of calcite) and is the main component of eggshells, snail shells, seashells and pearls. Calcium carbonate is the active ingredient in agricultural lime and is created when calcium ions in hard water react with carbonate ions to create limescale. It has medical use as a calcium supplement or as an antacid, but excessive consumption can be hazardous and cause hypercalcemia and digestive issues.

Sulfur dioxide or sulphur dioxide is the chemical compound with the formula SO
2. It is a toxic gas responsible for the smell of burnt matches. It is released naturally by volcanic activity and is produced as a by-product of copper extraction and the burning of sulfur-bearing fossil fuels. Sulfur dioxide has a pungent smell like nitric acid.

Calcium oxide (CaO), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature. The broadly used term "lime" connotes calcium-containing inorganic materials, in which carbonates, oxides and hydroxides of calcium, silicon, magnesium, aluminium, and iron predominate. By contrast, quicklime specifically applies to the single chemical compound calcium oxide. Calcium oxide that survives processing without reacting in building products such as cement is called free lime.
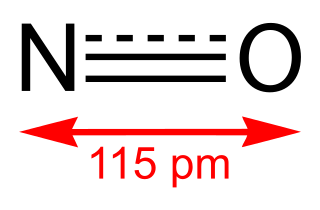
Nitric oxide is a colorless gas with the formula NO. It is one of the principal oxides of nitrogen. Nitric oxide is a free radical: it has an unpaired electron, which is sometimes denoted by a dot in its chemical formula. Nitric oxide is also a heteronuclear diatomic molecule, a class of molecules whose study spawned early modern theories of chemical bonding.
Carbonation is the chemical reaction of carbon dioxide to give carbonates, bicarbonates, and carbonic acid. In chemistry, the term is sometimes used in place of carboxylation, which refers to the formation of carboxylic acids.

Calcium hydroxide (traditionally called slaked lime) is an inorganic compound with the chemical formula Ca(OH)2. It is a colorless crystal or white powder and is produced when quicklime (calcium oxide) is mixed or slaked with water. It has many names including hydrated lime, caustic lime, builders' lime, slaked lime, cal, and pickling lime. Calcium hydroxide is used in many applications, including food preparation, where it has been identified as E number E526. Limewater, also called milk of lime, is the common name for a saturated solution of calcium hydroxide.
Calcination refers to heating (thermal treatment of) a solid chemical compound (e.g. carbonate ores) to high temperatures while staying below its melting point, in absence or limited supply of air or oxygen (O2), generally for the purpose of removing impurities or volatile substances and/or to incur thermal decomposition.

Hopcalite is the trade name for a number of mixtures that mainly consist of oxides of copper and manganese, which are used as catalysts for the conversion of carbon monoxide to carbon dioxide when exposed to the oxygen in the air at room temperature.
Pyrometallurgy is a branch of extractive metallurgy. It consists of the thermal treatment of minerals and metallurgical ores and concentrates to bring about physical and chemical transformations in the materials to enable recovery of valuable metals. Pyrometallurgical treatment may produce products able to be sold such as pure metals, or intermediate compounds or alloys, suitable as feed for further processing. Examples of elements extracted by pyrometallurgical processes include the oxides of less reactive elements like iron, copper, zinc, chromium, tin, and manganese.

Lime is a calcium-containing inorganic mineral composed primarily of oxides, and hydroxide, usually calcium oxide and/or calcium hydroxide. It is also the name for calcium oxide which occurs as a product of coal-seam fires and in altered limestone xenoliths in volcanic ejecta. The word lime originates with its earliest use as building mortar and has the sense of sticking or adhering.
Sodium oxide is a chemical compound with the formula Na2O. It is used in ceramics and glasses. The compound is the base anhydride of sodium hydroxide; when water is added to sodium oxide, NaOH is produced.

Calcium sulfide is the chemical compound with the formula CaS. This white material crystallizes in cubes like rock salt. CaS has been studied as a component in a process that would recycle gypsum, a product of flue-gas desulfurization. Like many salts containing sulfide ions, CaS typically has an odour of H2S, which results from small amount of this gas formed by hydrolysis of the salt.
A combination reaction (also known as a synthesis reaction) is a reaction where two or more elements or compounds (reactants) combine to form a single compound (product). Such reactions are represented by equations of the following form: X + Y → XY (A+B → AB). The combination of two or more elements to form one compound is called a combination reaction. In other words, when two or more elements or compounds react so as to form one single compound, then the chemical reaction that takes place is called a combination reaction. | a)- Between elements | C + O2 → CO2 | Carbon completely burnt in oxygen yields carbon dioxide |- | b) Between compounds | CaO + H2O → Ca(OH)2 | Calcium oxide (lime) combined with water gives calcium hydroxide (slaked lime) |- | c) Between elements and compounds | 2CO + O2 → 2CO2 | Oxygen combines with carbon monoxide,And carbon dioxide is formed. |}
A carbon dioxide scrubber is a piece of equipment that absorbs carbon dioxide (CO2). It is used to treat exhaust gases from industrial plants or from exhaled air in life support systems such as rebreathers or in spacecraft, submersible craft or airtight chambers. Carbon dioxide scrubbers are also used in controlled atmosphere (CA) storage. They have also been researched for carbon capture and storage as a means of combating climate change.
The sulfite process produces wood pulp that is almost pure cellulose fibers by treating wood chips with solutions of sulfite and bisulfite ions. These chemicals cleave the bonds between the cellulose and lignin components of the lignocellulose. A variety of sulfite/bisulfite salts are used, including sodium (Na+), calcium (Ca2+), potassium (K+), magnesium (Mg2+), and ammonium (NH4+). The lignin is converted to lignosulfonates, which are soluble and can be separated from the cellulose fibers. For the production of cellulose, the sulfite process competes with the Kraft process, which produces stronger fibers and is less environmentally costly.

Neptunium(IV) oxide, or neptunium dioxide, is a radioactive, olive green cubic crystalline solid with the formula NpO2. It is a common product of plutonium fission, and emits both α- and γ-particles.
A sulfite sulfate is a chemical compound that contains both sulfite and sulfate anions [SO3]2− [SO4]2−. These compounds were discovered in the 1980s as calcium and rare earth element salts. Minerals in this class were later discovered. Minerals may have sulfite as an essential component, or have it substituted for another anion as in alloriite. The related ions [O3SOSO2]2− and [(O2SO)2SO2]2− may be produced in a reaction between sulfur dioxide and sulfate and exist in the solid form as tetramethyl ammonium salts. They have a significant partial pressure of sulfur dioxide.











