
Catharanthus is a genus of flowering plants in the family Apocynaceae. Like the genus Vinca, they are known commonly as periwinkles. There are eight known species. Seven are endemic to Madagascar, though one, C. roseus, is widely naturalized around the world. The eighth species, C. pusillus, is native to India and Sri Lanka. The name Catharanthus comes from the Greek for "pure flower".
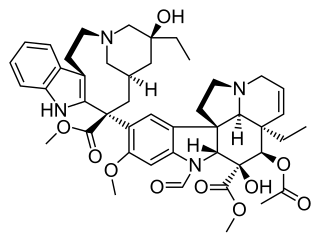
Vincristine, also known as leurocristine and marketed under the brand name Oncovin among others, is a chemotherapy medication used to treat a number of types of cancer. This includes acute lymphocytic leukemia, acute myeloid leukemia, Hodgkin's disease, neuroblastoma, and small cell lung cancer among others. It is given intravenously.

Catharanthus roseus, commonly known as bright eyes, Cape periwinkle, graveyard plant, Madagascar periwinkle, old maid, pink periwinkle, rose periwinkle, is a perennial species of flowering plant in the family Apocynaceae. It is native and endemic to Madagascar, but is grown elsewhere as an ornamental and medicinal plant, and now has a pantropical distribution. It is a source of the drugs vincristine and vinblastine, used to treat cancer. It was formerly included in the genus Vinca as Vinca rosea.

Vinca alkaloids are a set of anti-mitotic and anti-microtubule alkaloid agents originally derived from the periwinkle plant Catharanthus roseus and other vinca plants. They block beta-tubulin polymerization in a dividing cell.

Vindesine, also termed Eldisine, is a semisynthetic vinca alkaloid derived from the flowering plant Catharanthus roseus. Like the natural and semisynthetic vinca alkaloids derived from this plant, vindesine is an inhibitor of mitosis that is used as a chemotherapy drug. By inhibiting mitosis, vinedsine blocks the proliferation of cells, particularly the rapidly proliferation cells of certain types of cancer. It is used, generally in combination with other chemotherapeutic drugs, in the treatment of various malignancies such as leukaemia, lymphoma, melanoma, breast cancer, and lung cancer.
Ambovombe-Androy, or just Ambovombe, is a city in the far south of Madagascar, and the capital of the Androy region. Ambovombe has now acquired city status with an officially estimated population in 2018 of 114,230.
In enzymology, a tabersonine 16-hydroxylase (EC 1.14.13.73) is an enzyme that catalyzes the chemical reaction
Strictosidine synthase (EC 4.3.3.2) is an enzyme in alkaloid biosynthesis that catalyses the condensation of tryptamine with secologanin to form strictosidine in a formal Pictet–Spengler reaction:

Ajmalicine, also known as δ-yohimbine or raubasine, is an antihypertensive drug used in the treatment of high blood pressure. It has been marketed under numerous brand names including Card-Lamuran, Circolene, Cristanyl, Duxil, Duxor, Hydroxysarpon, Iskedyl, Isosarpan, Isquebral, Lamuran, Melanex, Raunatin, Saltucin Co, Salvalion, and Sarpan. It is an alkaloid found naturally in various plants such as Rauvolfia spp., Catharanthus roseus, and Mitragyna speciosa.
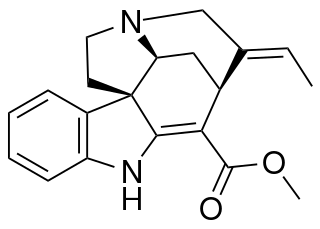
Akuammicine is a monoterpene indole alkaloid of the Vinca sub-group. It is found in the Apocynaceae family of plants including Picralima nitida, Vinca minor and the Aspidosperma.

Catharanthine is a terpene indole alkaloid produced by the medicinal plant Catharanthus roseus and Tabernaemontana divaricata. Catharanthine is derived from strictosidine, but the exact mechanism by which this happens is currently unknown. Catharanthine is one of the two precursors that form vinblastine, the other being vindoline.
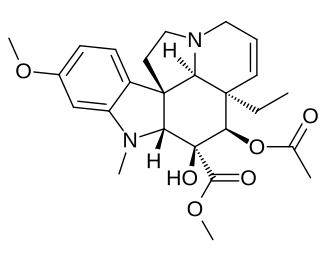
Vindoline is a chemical precursor to vinblastine. Vindoline is formed through biosynthesis from Tabersonine.

Serpentine is a terpene indole alkaloid produced by several members of the family Apocynaceae, including Catharanthus roseus and Rauvolfia serpentina.

Strictosidine is a natural chemical compound and is classified as a glucoalkaloid and a vinca alkaloid. It is formed by the Pictet–Spengler condensation reaction of tryptamine with secologanin, catalyzed by the enzyme strictosidine synthase. Thousands of strictosidine derivatives are sometimes referred to by the broad phrase of monoterpene indole alkaloids. Strictosidine is an intermediate in the biosynthesis of numerous pharmaceutically valuable metabolites including quinine, camptothecin, ajmalicine, serpentine, vinblastine, vincristine and mitragynine.

Tabersonine is a terpene indole alkaloid found in the medicinal plant Catharanthus roseus and also in the genus Voacanga. Tabersonine is hydroxylated at the 16 position by the enzyme tabersonine 16-hydroxylase (T16H) to form 16-hydroxytabersonine. The enzyme leading to its formation is currently unknown. Tabersonine is the first intermediate leading to the formation of vindoline one of the two precursors required for vinblastine biosynthesis.

16-Hydroxytabersonine is a terpene indole alkaloid produced by the plant Catharanthus roseus. The metabolite is an intermediate in the formation of vindoline, a precursor needed for formation of the pharmaceutically valuable vinblastine and vincristine. 16-hydroxytabersonine is formed from the hydroxylation of tabersonine by tabersonine 16-hydroxylase (T16H). Tabersonine 16-O-methyltransferase (16OMT) methylates the hydroxylated 16 position to form 16-methoxytabersonine.

16-Methoxytabersonine is a terpene indole alkaloid produced by the medicinal plant Catharanthus roseus. 16-methoxytabersonine is synthesized by methylation of the hydroxyl group at the 16 position of 16-hydroxytabersonine by tabersonine 16-O-methyltransferase (16OMT). The compound is a substrate for hydration by two concerted enzymes Tabersonine-3-Oxidase (T3O) and Tabersonine-3-Reductase (T3R), which leads to the formation of 3-hydroxy-16-methoxy-2,3-dihydrotabersonine.

Deacetylvindoline is a terpene indole alkaloid produced by Catharanthus roseus. Deacetylvindoline is the product of a hydroxylation of desacetoxyvindoline by deacetoxyvindoline 4-hydroxylase (D4H). It is a substrate for deacetylvindoline O-acetyltransferase (DAT) which acetylates a hydroxy group to form vindoline, one of the two immediate precursors for the formation of the pharmacetucially valuable bisindole alkaloid vinblastine.

Stemmadenine is a terpene indole alkaloid. Stemmadenine is believed to be formed from preakuammicine by a carbon-carbon bond cleavage. Cleavage of a second carbon-carbon bond is thought to form dehydrosecodine. The enzymes forming stemmadenine and using it as a substrate remain unknown to date. It is thought to be intermediate compound in many different biosynthetic pathways such as in Aspidosperma species. Many alkaloids are proposed to be produced through intermediate stemmadenine. Some of them are:

Catharanthine and vindoline are terpenoid indole alkaloids naturally produced within the Madagascar periwinkle plant whose dimerization produces the anti-cancer drugs vinblastine and vincristine. The precursor of catharanthine and vindoline is strictosidine, the common precursor of all indole alkaloids. The localization of catharanthine and vindoline within the plant tissue has been heavily studied in recent years with conflicting results. The dimerization of catharanthine and vindoline to form vinblastine and vincristine is catalyzed by a peroxidase and a reductase, and includes several intermediate compounds.
















