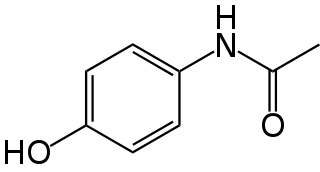
Paracetamol is a non-opioid analgesic and antipyretic agent used to treat fever and mild to moderate pain. It is a widely used over the counter medication and common brand names include Tylenol and Panadol.

Ibuprofen is a nonsteroidal anti-inflammatory drug (NSAID) that is used to relieve pain, fever, and inflammation. This includes painful menstrual periods, migraines, and rheumatoid arthritis. It may also be used to close a patent ductus arteriosus in a premature baby. It can be used orally or intravenously. It typically begins working within an hour.

Over-the-counter (OTC) drugs are medicines sold directly to a consumer without a requirement for a prescription from a healthcare professional, as opposed to prescription drugs, which may be supplied only to consumers possessing a valid prescription. In many countries, OTC drugs are selected by a regulatory agency to ensure that they contain ingredients that are safe and effective when used without a physician's care. OTC drugs are usually regulated according to their active pharmaceutical ingredient (API) rather than final products. By regulating APIs instead of specific drug formulations, governments allow manufacturers the freedom to formulate ingredients, or combinations of ingredients, into proprietary mixtures.

Brasso is a metal polish designed to remove tarnish from brass, copper, chrome and stainless steel. It is available either directly as a liquid or as an impregnated wadding pad.

Dihydrocodeine is a semi-synthetic opioid analgesic prescribed for pain or severe dyspnea, or as an antitussive, either alone or compounded with paracetamol (acetaminophen) or aspirin. It was developed in Germany in 1908 and first marketed in 1911.

Excedrin is an over-the-counter headache pain reliever, typically in the form of tablets or caplets. It contains paracetamol (acetaminophen), aspirin, and caffeine. It was manufactured by Bristol-Myers Squibb until it was purchased by Novartis in July 2005 along with other products from BMS's over-the-counter business. As of March 2015, GSK holds majority ownership of Excedrin through a joint venture transaction with Novartis. On 18 July 2022, GSK spun off its consumer healthcare business to Haleon.
Mr Sheen is the brand name of a range of cleaning materials, mainly floor and furniture polish, created in Australia in the 1950s by Samuel Taylor Pty Ltd. An aerosol, the product uses a combination of silicones, waxes and solvents and is currently manufactured by Reckitt. Alongside Australia, Mr Sheen is also available in Jamaica, Kenya, Ireland and the United Kingdom, as well as in Zimbabwe, Zambia and South Africa where it is called Mr Min and in Argentina where it is known as Mr Frend. The Mr Sheen character and branding, despite numerous revamps, has a high level of recognition amongst consumers, so much so that the company has been able to go for periods of time without even advertising the product, such as in the early 1990s.

Cepacol is an American brand of personal hygiene products, as well as for relief of sore throat. It is distributed in the US by Reckitt. The brand was originally owned by J.B. Williams. Following acquisition by Combe Incorporated, Combe Incorporated sold Cepacol to Reckitt Benckiser in 2011.

Reckitt Benckiser Group plc, trading as Reckitt, is a British multinational consumer goods company headquartered in Slough, England. It is a producer of health, hygiene and nutrition products. The company was formed in March 1999 by the merger of British company Reckitt & Colman plc and Dutch company Benckiser N.V.

Strepsils is a brand of throat lozenges manufactured by Reckitt. Strepsils throat lozenges are used to relieve discomfort caused by mouth and throat infections.

Solpadeine is the brand name of a range of analgesic medication containing various amounts of paracetamol, ibuprofen, caffeine and codeine, made by Omega Pharma. The range was previously made by GlaxoSmithKline, which sold its portfolio of over-the-counter drugs to Omega Pharma in 2012.
Migraleve is the brand name of a range of migraine-relief medications made by Pfizer.
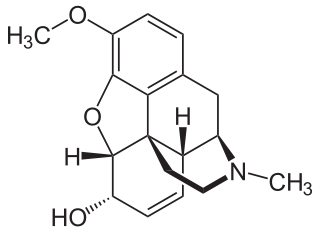
Codeine is an opiate and prodrug of morphine mainly used to treat pain, coughing, and diarrhea. It is also commonly used as a recreational drug. It is found naturally in the sap of the opium poppy, Papaver somniferum. It is typically used to treat mild to moderate degrees of pain. Greater benefit may occur when combined with paracetamol (acetaminophen) or a nonsteroidal anti-inflammatory drug (NSAID) such as aspirin or ibuprofen. Evidence does not support its use for acute cough suppression in children or adults. In Europe, it is not recommended as a cough medicine in those under 12 years of age. It is generally taken by mouth. It typically starts working after half an hour, with maximum effect at two hours. Its effects last for about four to six hours. Codeine exhibits abuse potential similar to other opioid medications, including a risk of habituation and overdose.
Harpic is the brand name of a toilet cleaner launched in the United Kingdom in 1932 by Reckitt and Sons. It is currently available in Africa, the Middle East, South Asia, the Asia-Pacific, Europe, and the Americas. The toilet cleaning products marketed under the brand name include liquids, tablets, wipes, brush systems, toilet rim blocks, and in-cistern blocks.
This is a partial list of notable price fixing and bid rigging cases.
Crookes Healthcare is a healthcare manufacturer based in Nottingham, England, and a subsidiary of Reckitt. It manufactures some of the best-known health remedies and over-the-counter drugs sold by British pharmacies.

Vanish is a brand of stain removing products, owned by the product manufacturer Reckitt, and sold in Australia, India, Indonesia, Russia, South Africa, Latin America and parts of Europe.

Kenneth John Harvey AM is an Australian public health doctor, currently Honorary Adjunct Associate Professor at the Institute for Evidence-Based Healthcare in Bond University. Described by The Age as an "anti-quackery crusader", Harvey is an advocate of evidence-based medicine and a critic of pharmaceutical marketing and unproven diet products. He is the president of Friends of Science in Medicine. In 2017, Harvey was awarded a Member of the Order of Australia (AM) for his "significant service to community health and the pharmaceutical industry”.
Paul Edward Rolan is an Australian physician, medical researcher, academic, and public health advocate. He specializes in the management of chronic pain and headache, and development of novel treatments.














