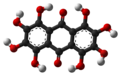
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name 1,2,3,4,5,6,7,8-Octahydroxyanthracene-9,10-dione | |||
| Other names Octahydroxyanthracenedione | |||
| Identifiers | |||
3D model (JSmol) | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
PubChem CID | |||
| UNII | |||
CompTox Dashboard (EPA) | |||
| |||
| |||
| Properties | |||
| C14H8O10 | |||
| Molar mass | 336.208 g·mol−1 | ||
| log P | −0.291 | ||
| Acidity (pKa) | 5.358 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
Octahydroxyanthraquinone is an organic compound with formula C
14H
8O
10, formally derived from anthraquinone by replacement of 8 hydrogen atoms by hydroxyl groups.
The compound was obtained in 1911 by Georg von Georgievics [1] [2] and can be obtained through oxidation of rufigallol (1,2,3,5,6,7-hexahydroxyanthraquinone) with boric acid and mercuric oxide in sulfuric acid at 250 °C (482 °F). [3]
Esters of octahydroxyanthraquinone, where all eight hydroxyls are replaced by straight-chain 1-alkanecarboxylate groups H
3C-(CH
2)n-COO-, with n between 6 and 14, are liquid crystals and have been studied for possible LCD applications. [3]
Octahydroxyanthraquinone is active against the malaria parasite, but rufigallol (1,2,3,5,6,7-hexahydroxyanthraquinone) is 22 times more potent. [4]