Related Research Articles

Estradiol (E2), also spelled oestradiol, is an estrogen steroid hormone and the major female sex hormone. It is involved in the regulation of the estrous and menstrual female reproductive cycles. Estradiol is responsible for the development of female secondary sexual characteristics such as the breasts, widening of the hips and a female-associated pattern of fat distribution. It is also important in the development and maintenance of female reproductive tissues such as the mammary glands, uterus and vagina during puberty, adulthood and pregnancy. It also has important effects in many other tissues including bone, fat, skin, liver, and the brain.

Estriol (E3), also spelled oestriol, is a steroid, a weak estrogen, and a minor female sex hormone. It is one of three major endogenous estrogens, the others being estradiol and estrone. Levels of estriol in women who are not pregnant are almost undetectable. However, during pregnancy, estriol is synthesized in very high quantities by the placenta and is the most produced estrogen in the body by far, although circulating levels of estriol are similar to those of other estrogens due to a relatively high rate of metabolism and excretion. Relative to estradiol, both estriol and estrone have far weaker activity as estrogens.
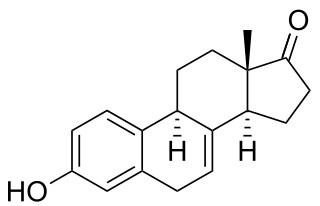
Equilin is a naturally occurring estrogen sex hormone found in horses as well as a medication. It is one of the estrogens present in the estrogen mixtures known as conjugated estrogens and esterified estrogens. CEEs is the most commonly used form of estrogen in hormone replacement therapy (HRT) for menopausal symptoms in the United States. Estrone sulfate is the major estrogen in CEEs while equilin sulfate is the second major estrogen in the formulation, present as about 25% of the total.

Conjugated estrogens (CEs), or conjugated equine estrogens (CEEs), sold under the brand name Premarin among others, is an estrogen medication which is used in menopausal hormone therapy and for various other indications. It is a mixture of the sodium salts of estrogen conjugates found in horses, such as estrone sulfate and equilin sulfate. CEEs are available in the form of both natural preparations manufactured from the urine of pregnant mares and fully synthetic replications of the natural preparations. They are formulated both alone and in combination with progestins such as medroxyprogesterone acetate. CEEs are usually taken by mouth, but can also be given by application to the skin or vagina as a cream or by injection into a blood vessel or muscle.

An estrogen conjugate is a conjugate of an endogenous estrogen. They occur naturally in the body as metabolites of estrogens and can be reconverted back into estrogens. They serve as a circulating reservoir for estrogen, particularly in the case of orally administered pharmaceutical estradiol. Estrogen conjugates include sulfate and/or glucuronide conjugates of estradiol, estrone, and estriol:

Estradiol sulfate (E2S), or 17β-estradiol 3-sulfate, is a natural, endogenous steroid and an estrogen ester. E2S itself is biologically inactive, but it can be converted by steroid sulfatase into estradiol, which is a potent estrogen. Simultaneously, estrogen sulfotransferases convert estradiol to E2S, resulting in an equilibrium between the two steroids in various tissues. Estrone and E2S are the two immediate metabolic sources of estradiol. E2S can also be metabolized into estrone sulfate (E1S), which in turn can be converted into estrone and estradiol. Circulating concentrations of E2S are much lower than those of E1S. High concentrations of E2S are present in breast tissue, and E2S has been implicated in the biology of breast cancer via serving as an active reservoir of estradiol.

17β-Dihydroequilin is a naturally occurring estrogen sex hormone found in horses as well as a medication. As the C3 sulfate ester sodium salt, it is a minor constituent (1.7%) of conjugated estrogens. However, as equilin, with equilin sulfate being a major component of CEEs, is transformed into 17β-dihydroequilin in the body, analogously to the conversion of estrone into estradiol, 17β-dihydroequilin is, along with estradiol, the most important estrogen responsible for the effects of CEEs.

17β-Dihydroequilenin, or β-dihydroequilenin, also known as δ6,8-17β-estradiol or 6,8-didehydro-17β-estradiol, as well as estra-1,3,5(10),6,8-pentaen-3,17β-diol, is a naturally occurring steroidal estrogen found in horses which is closely related to equilin, equilenin, and estradiol, and, as the 3-sulfate ester sodium salt, is a minor constituent (0.5%) of conjugated estrogens (Premarin). 17β-Dihydroequilenin has unexpectedly shown a selective estrogen receptor modulator (SERM)-like profile of estrogenic activity in studies with monkeys, in which beneficial effects on bone and the cardiovascular system were noted but proliferative responses in breast and endometrium were not observed.

8,9-Dehydroestrone, or Δ8-estrone, also known as estra-1,3,5(10),8-tetraen-3-ol-17-one, is a naturally occurring estrogen found in horses which is closely related to equilin, equilenin, and estrone, and, as the 3-sulfate ester sodium salt, is a minor constituent (3.5%) of conjugated estrogens (Premarin). It produces 8,9-dehydro-17β-estradiol as an important active metabolite, analogously to conversion of estrone or estrone sulfate into estradiol. The compound was first described in 1997. In addition to 8,9-dehydroestrone and 8,9-dehydro-17β-estradiol, 8,9-dehydro-17α-estradiol is likely also to be present in conjugated estrogens, but has not been identified at this time.

8,9-Dehydroestradiol, or Δ8-17β-estradiol, also known as estra-1,3,5(10),8-tetraen-17β-ol-3-one, is a naturally occurring steroidal estrogen found in horses which is closely related to equilin, equilenin, and estradiol, and, as the 3-sulfate ester sodium salt, is a minor constituent of conjugated estrogens (Premarin). It is also an important active metabolite of 8,9-dehydroestrone, analogously to conversion of estrone or estrone sulfate into estradiol.

Estriol glucuronide (E3G), or oestriol glucuronide, also known as estriol monoglucuronide, as well as estriol 16α-β-D-glucosiduronic acid, is a natural, steroidal estrogen and the glucuronic acid conjugate of estriol. It occurs in high concentrations in the urine of pregnant women as a reversibly formed metabolite of estriol. Estriol glucuronide is a prodrug of estriol, and was the major component of Progynon and Emmenin, estrogenic products manufactured from the urine of pregnant women that were introduced in the 1920s and 1930s and were the first orally active estrogens. Emmenin was succeeded by Premarin, which is sourced from the urine of pregnant mares and was introduced in 1941. Premarin replaced Emmenin due to the fact that it was easier and less expensive to produce.

Estradiol glucuronide, or estradiol 17β-D-glucuronide, is a conjugated metabolite of estradiol. It is formed from estradiol in the liver by UDP-glucuronyltransferase via attachment of glucuronic acid and is eventually excreted in the urine by the kidneys. It has much higher water solubility than does estradiol. Glucuronides are the most abundant estrogen conjugates.
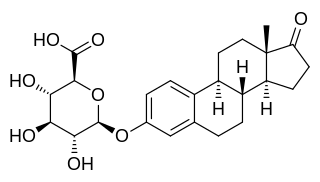
Estrone glucuronide, or estrone-3-D-glucuronide, is a conjugated metabolite of estrone. It is formed from estrone in the liver by UDP-glucuronyltransferase via attachment of glucuronic acid and is eventually excreted in the urine by the kidneys. It has much higher water solubility than does estrone. Glucuronides are the most abundant estrogen conjugates and estrone glucuronide is the dominant metabolite of estradiol.

2-Hydroxyestrone (2-OHE1), also known as estra-1,3,5(10)-trien-2,3-diol-17-one, is an endogenous, naturally occurring catechol estrogen and a major metabolite of estrone and estradiol. It is formed irreversibly from estrone in the liver and to a lesser extent in other tissues via 2-hydroxylation mediated by cytochrome P450 enzymes, mainly the CYP3A and CYP1A subfamilies. 2-OHE1 is the most abundant catechol estrogen in the body.

4-Hydroxyestradiol (4-OHE2), also known as estra-1,3,5(10)-triene-3,4,17β-triol, is an endogenous, naturally occurring catechol estrogen and a minor metabolite of estradiol. It is estrogenic, similarly to many other hydroxylated estrogen metabolites such as 2-hydroxyestradiol, 16α-hydroxyestrone, estriol (16α-hydroxyestradiol), and 4-hydroxyestrone but unlike 2-hydroxyestrone.
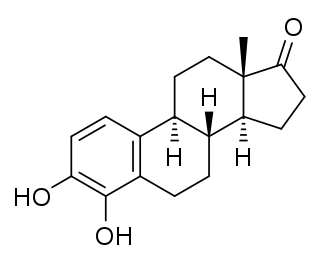
4-Hydroxyestrone (4-OHE1), also known as estra-1,3,5(10)-triene-3,4-diol-17-one, is an endogenous, naturally occurring catechol estrogen and a minor metabolite of estrone and estradiol. It is estrogenic, similarly to many other hydroxylated estrogen metabolites such as 2-hydroxyestradiol, 16α-hydroxyestrone, estriol (16α-hydroxyestradiol), and 4-hydroxyestradiol but unlike 2-hydroxyestrone.
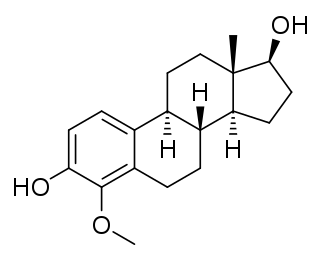
4-Methoxyestradiol (4-ME2) is an endogenous, naturally occurring methoxylated catechol estrogen and metabolite of estradiol that is formed by catechol O-methyltransferase via the intermediate 4-hydroxyestradiol. It has estrogenic activity similarly to estrone and 4-hydroxyestrone.

Estrone sulfate (E1S) is an estrogen medication and naturally occurring steroid hormone. It is used in menopausal hormone therapy among other indications. As the sodium salt, it is the major estrogen component of conjugated estrogens (Premarin) and esterified estrogens. In addition, E1S is used on its own as the piperazine salt estropipate. The compound also occurs as a major and important metabolite of estradiol and estrone. E1S is most commonly taken by mouth, but in the form of Premarin can also be taken by parenteral routes such as transdermal, vaginal, and injection.
17β-Methyl-17α-dihydroequilenin, also known as 17β-methyl-6,8-didehydro-17α-estradiol, is a synthetic steroidal estrogen which was never marketed. It is the C17β methylated derivative of 17α-dihydroequilenin, an equine estrogen and constituent of conjugated estrogens (Premarin). 17α-Dihydroequilenin itself is an analogue of 17α-estradiol, the C17 epimer of estradiol. NCI-122 has respective relative binding affinities of about 8.1% and 16% for the ERα and ERβ when compared to estradiol. It is far less potent as an estrogen in comparison to estradiol, with relative estrogenic potencies at the ERα and ERβ of 1.4% and 0.81%, respectively. Nonetheless, NCI-122 acts as a full agonist of the ERα and has estrogenic activity similar to that of estradiol at sufficiently high concentrations. The mechanisms of the lower potency of NCI-122 and related estrogens relative to estradiol have been studied.
References
- 1 2 3 Kuhl H (2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration" (PDF). Climacteric. 8 Suppl 1: 3–63. doi:10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- 1 2 3 Bhavnani BR, Tam SP, Lu X (October 2008). "Structure activity relationships and differential interactions and functional activity of various equine estrogens mediated via estrogen receptors (ERs) ERalpha and ERbeta". Endocrinology. 149 (10): 4857–70. doi: 10.1210/en.2008-0304 . PMID 18599548.
- 1 2 3 Bhavnani BR (June 2003). "Estrogens and menopause: pharmacology of conjugated equine estrogens and their potential role in the prevention of neurodegenerative diseases such as Alzheimer's". J. Steroid Biochem. Mol. Biol. 85 (2–5): 473–82. doi:10.1016/s0960-0760(03)00220-6. PMID 12943738. S2CID 45552896.