
Molecular engineering is an emerging field of study concerned with the design and testing of molecular properties, behavior and interactions in order to assemble better materials, systems, and processes for specific functions. This approach, in which observable properties of a macroscopic system are influenced by direct alteration of a molecular structure, falls into the broader category of “bottom-up” design.

A lithium-ion or Li-ion battery is a type of rechargeable battery which uses the reversible reduction of lithium ions to store energy. The negative electrode of a conventional lithium-ion cell is typically graphite, a form of carbon. This negative electrode is sometimes called the anode as it acts as an anode during discharge. The positive electrode is typically a metal oxide; the positive electrode is sometimes called the cathode as it acts as a cathode during discharge. Positive and negative electrodes remain positive and negative in normal use whether charging or discharging and are therefore clearer terms to use than anode and cathode which are reversed during charging.
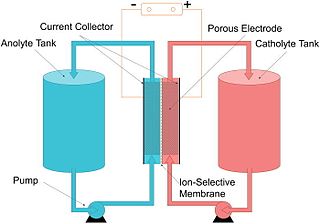
A flow battery, or redox flow battery, is a type of electrochemical cell where chemical energy is provided by two chemical components dissolved in liquids that are pumped through the system on separate sides of a membrane. Ion transfer inside the cell occurs through the membrane while both liquids circulate in their own respective space. Cell voltage is chemically determined by the Nernst equation and ranges, in practical applications, from 1.0 to 2.43 volts. The energy capacity is a function of the electrolyte volume and the power is a function of the surface area of the electrodes.
Bistriflimide, also known variously as bis(trifluoromethane)sulfonimide, bis(trifluoromethanesulfonyl)imide, bis(trifluoromethanesulfonyl)imidate (and variations thereof), informally and somewhat inaccurately as triflimide or triflimidate, or by the abbreviations TFSI or NTf2, is a non-coordinating anion with the chemical formula [(CF3SO2)2N]−. Its salts are typically referred to as being metal triflimidates.

A lithium-ion capacitor is a hybrid type of capacitor classified as a type of supercapacitor. It is called a hybrid because the anode is the same as those used in lithium-ion batteries and the cathode is the same as those used in supercapacitors. Activated carbon is typically used as the cathode. The anode of the LIC consists of carbon material which is often pre-doped with lithium ions. This pre-doping process lowers the potential of the anode and allows a relatively high output voltage compared to other supercapacitors.
An Ion gel is a composite material consisting of an ionic liquid immobilized by an inorganic or a polymer matrix. The material has the quality of maintaining high ionic conductivity while in the solid state. To create an ion gel, the solid matrix is mixed or synthesized in-situ with an ionic liquid. A common practice is to utilize a block copolymer which is polymerized in solution with an ionic liquid so that a self-assembled nanostructure is generated where the ions are selectively soluble. Ion gels can also be made using non-copolymer polymers such as cellulose, oxides such as silicon dioxide or refractory materials such as boron nitride.
The lithium–air battery (Li–air) is a metal–air electrochemical cell or battery chemistry that uses oxidation of lithium at the anode and reduction of oxygen at the cathode to induce a current flow.
A potassium-ion battery or K-ion battery is a type of battery and analogue to lithium-ion batteries, using potassium ions for charge transfer instead of lithium ions. It was invented by the Iranian/American chemist Ali Eftekhari in 2004.
The sodium-ion battery (NIB or SIB) is a type of rechargeable battery that uses sodium ions (Na+) as its charge carriers. In some cases, its working principle and cell construction are similar to those of lithium-ion battery (LIB) types, but it replaces lithium with sodium as the cathode material, which belongs to the same group in the periodic table as lithium and thus has similar chemical properties.
Aluminium-ion batteries are a class of rechargeable battery in which aluminium ions serve as charge carriers. Aluminium can exchange three electrons per ion. This means that insertion of one Al3+ is equivalent to three Li+ ions. Thus, since the ionic radii of Al3+ (0.54 Å) and Li+ (0.76 Å) are similar, significantly higher numbers of electrons and Al3+ ions can be accepted by cathodes with little damage. Al has 50 times (23.5 megawatt-hours m-3) the energy density of Li and is even higher than coal.
Research in lithium-ion batteries has produced many proposed refinements of lithium-ion batteries. Areas of research interest have focused on improving energy density, safety, rate capability, cycle durability, flexibility, and cost.

NASICON is an acronym for sodium (Na) Super Ionic CONductor, which usually refers to a family of solids with the chemical formula Na1+xZr2SixP3−xO12, 0 < x < 3. In a broader sense, it is also used for similar compounds where Na, Zr and/or Si are replaced by isovalent elements. NASICON compounds have high ionic conductivities, on the order of 10−3 S/cm, which rival those of liquid electrolytes. They are caused by hopping of Na ions among interstitial sites of the NASICON crystal lattice.
Magnesium batteries are batteries that utilize magnesium cations as the active charge transporting agents in solution and often as the elemental anode of an electrochemical cell. Both non-rechargeable primary cell and rechargeable secondary cell chemistries have been investigated. Magnesium primary cell batteries have been commercialised and have found use as reserve and general use batteries.
Yang Shao-Horn is a Chinese American scholar, Professor of Mechanical Engineering and Materials Science and Engineering and a member of Research Laboratory of Electronics at the Massachusetts Institute of Technology. She is known for research on understanding and controlling of processes for storing electrons in chemical bonds towards zero-carbon energy and chemicals.
An aqueous lithium-ion battery is a lithium-ion battery (Li-ion) that uses a concentrated saline solution as an electrolyte to facilitate the transfer of lithium ions between electrodes and induce an electrical current. In contrast to non-aqueous lithium-ion batteries, aqueous Li-ion batteries are nonflammable and do not pose any significant risks of explosion, because of the water-based nature of their electrolyte. They also lack the poisonous chemicals and environmental risks associated with their non-aqueous counterparts.
Calcium (ion) batteries are energy storage and delivery technologies (i.e., electro–chemical energy storage) that employ calcium ions (cations), Ca2+, as the active charge carrier in the electrolytes as well as in the electrodes (anode and cathode). Calcium (ion) batteries remain an active area of research, with studies and work persisting in the discovery and development of electrodes and electrolytes that enable stable, long-term battery operation.
Susan A. Odom was a Professor of Chemistry at the University of Kentucky who developed redox active organic compounds for energy storage applications.

Lithium lactate is a chemical compound, a salt of lithium and lactic acid with the formula CH3CH(OH)COOLi, an amorphous solid, very soluble in water.
Superconcentrated electrolytes, also known as water-in-salt or solvent-in-salt liquids, usually refer to chemical systems, which are liquid near room temperature and consist of a solvent-to-dissoved salt in a molar ratio near or smaller than ca. 4-8, i.e. where all solvent molecules are coordinated to cations, and no free solvent molecules remain. Since ca. 2010 such liquid electrolytes found several applications, primarily for batteries. In the case of lithium metal batteries and lithium-ion batteries most commonly used anions for superconcentrated electrolytes are those, that are large, asymmetric and rotationally-vibrationally flexible, such bis(trifluoromethanesulfonyl)amide and bis(fluorosulfonyl)amide. Noteworthy, lithium chloride and sodium perchlorate also form water-in-salt solutions.
A liquified gas electrolyte (LGE) is a battery/capacitor electrolyte made by compressing an ambient pressure gas into liquid form. Candidate gases are those composed of reasonably polar molecules that can be liquified at pressures low enough to be accommodate in a standard battery can.







