Prostaglandins (PG) are a group of physiologically active lipid compounds called eicosanoids having diverse hormone-like effects in animals. Prostaglandins have been found in almost every tissue in humans and other animals. They are derived enzymatically from the fatty acid arachidonic acid. Every prostaglandin contains 20 carbon atoms, including a 5-carbon ring. They are a subclass of eicosanoids and of the prostanoid class of fatty acid derivatives.
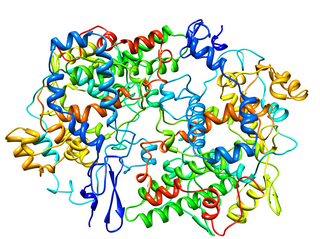
Cyclooxygenase (COX), officially known as prostaglandin-endoperoxide synthase (PTGS), is an enzyme that is responsible for biosynthesis of prostanoids, including thromboxane and prostaglandins such as prostacyclin, from arachidonic acid. A member of the animal-type heme peroxidase family, it is also known as prostaglandin G/H synthase. The specific reaction catalyzed is the conversion from arachidonic acid to prostaglandin H2 via a short-living prostaglandin G2 intermediate.

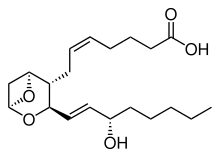
Prostacyclin (also called prostaglandin I2 or PGI2) is a prostaglandin member of the eicosanoid family of lipid molecules. It inhibits platelet activation and is also an effective vasodilator.

Thromboxane is a member of the family of lipids known as eicosanoids. The two major thromboxanes are thromboxane A2 and thromboxane B2. The distinguishing feature of thromboxanes is a 6-membered ether-containing ring.

Thromboxane A synthase 1 , also known as TBXAS1, is a cytochrome P450 enzyme that, in humans, is encoded by the TBXAS1 gene.

The thromboxane receptor (TP) also known as the prostanoid TP receptor is a protein that in humans is encoded by the TBXA2R gene, The thromboxane receptor is one among the five classes of prostanoid receptors and was the first eicosanoid receptor cloned. The TP receptor derives its name from its preferred endogenous ligand thromboxane A2.
A prostaglandin antagonist is a hormone antagonist acting upon one or more prostaglandins, a subclass of eicosanoid compounds which function as signaling molecules in numerous types of animal tissues.
Most of the eicosanoid receptors are integral membrane protein G protein-coupled receptors (GPCRs) that bind and respond to eicosanoid signaling molecules. Eicosanoids are rapidly metabolized to inactive products and therefore are short-lived. Accordingly, the eicosanoid-receptor interaction is typically limited to a local interaction: cells, upon stimulation, metabolize arachidonic acid to an eicosanoid which then binds cognate receptors on either its parent cell or on nearby cells to trigger functional responses within a restricted tissue area, e.g. an inflammatory response to an invading pathogen. In some cases, however, the synthesized eicosanoid travels through the blood to trigger systemic or coordinated tissue responses, e.g. prostaglandin (PG) E2 released locally travels to the hypothalamus to trigger a febrile reaction. An example of a non-GPCR receptor that binds many eicosanoids is the PPAR-γ nuclear receptor.
Prostaglandin receptors or prostanoid receptors represent a sub-class of cell surface membrane receptors that are regarded as the primary receptors for one or more of the classical, naturally occurring prostanoids viz., prostaglandin D2,, PGE2, PGF2alpha, prostacyclin (PGI2), thromboxane A2 (TXA2), and PGH2. They are named based on the prostanoid to which they preferentially bind and respond, e.g. the receptor responsive to PGI2 at lower concentrations than any other prostanoid is named the Prostacyclin receptor (IP). One exception to this rule is the receptor for thromboxane A2 (TP) which binds and responds to PGH2 and TXA2 equally well.
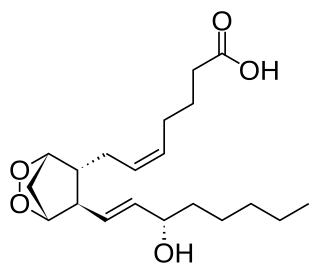
Prostaglandin H2 is a type of prostaglandin and a precursor for many other biologically significant molecules. It is synthesized from arachidonic acid in a reaction catalyzed by a cyclooxygenase enzyme. The conversion from Arachidonic acid to Prostaglandin H2 is a two step process. First, COX-1 catalyzes the addition of two free oxygens to form the 1,2-Dioxane bridge and a peroxide functional group to form Prostaglandin G2. Second, COX-2 reduces the peroxide functional group to a Secondary alcohol, forming Prostaglandin H2. Other peroxidases like Hydroquinone have been observed to reduce PGG2 to PGH2. PGH2 is unstable at room temperature, with a half life of 90-100 seconds, so it is often converted into a different prostaglandin.

Cyclooxygenase 1 (COX-1), also known as prostaglandin G/H synthase 1, prostaglandin-endoperoxide synthase 1 or prostaglandin H2 synthase 1, is an enzyme that in humans is encoded by the PTGS1 gene. In humans it is one of two cyclooxygenases.

Prostaglandin EP3 receptor (53kDa), also known as EP3, is a prostaglandin receptor for prostaglandin E2 (PGE2) encoded by the human gene PTGER3; it is one of four identified EP receptors, the others being EP1, EP2, and EP4, all of which bind with and mediate cellular responses to PGE2 and also, but generally with lesser affinity and responsiveness, certain other prostanoids (see Prostaglandin receptors). EP has been implicated in various physiological and pathological responses.

The Prostacyclin receptor, also termed the prostaglandin I2 receptor or just IP, is a receptor belonging to the prostaglandin (PG) group of receptors. IP binds to and mediates the biological actions of prostacyclin (also termed Prostaglandin I2, PGI2, or when used as a drug, epoprostenol). IP is encoded in humans by the PTGIR gene. While possessing many functions as defined in animal model studies, the major clinical relevancy of IP is as a powerful vasodilator: stimulators of IP are used to treat severe and even life-threatening diseases involving pathological vasoconstriction.

Aspirin causes several different effects in the body, mainly the reduction of inflammation, analgesia, the prevention of clotting, and the reduction of fever. Much of this is believed to be due to decreased production of prostaglandins and TXA2. Aspirin's ability to suppress the production of prostaglandins and thromboxanes is due to its irreversible inactivation of the cyclooxygenase (COX) enzyme. Cyclooxygenase is required for prostaglandin and thromboxane synthesis. Aspirin acts as an acetylating agent where an acetyl group is covalently attached to a serine residue in the active site of the COX enzyme. This makes aspirin different from other NSAIDs, which are reversible inhibitors. However, other effects of aspirin, such as uncoupling oxidative phosphorylation in mitochondria, and the modulation of signaling through NF-κB, are also being investigated. Some of its effects are like those of salicylic acid, which is not an acetylating agent.

11-Dehydrothromboxane B2 is produced from the breakdown of thromboxane A2. It is released by activated platelets and urine levels of 11-dehydro-TXB2 can be used to monitor the response to aspirin therapy when used to prevent heart disease and in diseases where platelet activation is prominent.
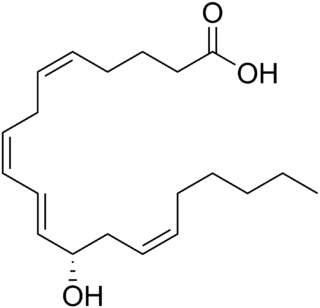
12-Hydroxyeicosatetraenoic acid (12-HETE) is a derivative of the 20 carbon polyunsaturated fatty acid, arachidonic acid, containing a hydroxyl residue at carbon 12 and a 5Z,8Z,10E,14Z Cis–trans isomerism configuration (Z=cis, E=trans) in its four double bonds. It was first found as a product of arachidonic acid metabolism made by human and bovine platelets through their 12S-lipoxygenase (i.e. ALOX12) enzyme(s). However, the term 12-HETE is ambiguous in that it has been used to indicate not only the initially detected "S" stereoisomer, 12S-hydroxy-5Z,8Z,10E,14Z-eicosatetraenoic acid (12(S)-HETE or 12S-HETE), made by platelets, but also the later detected "R" stereoisomer, 12(R)-hydroxy-5Z,8Z,10E,14Z-eicosatetraenoic acid (also termed 12(R)-HETE or 12R-HETE) made by other tissues through their 12R-lipoxygenase enzyme, ALOX12B. The two isomers, either directly or after being further metabolized, have been suggested to be involved in a variety of human physiological and pathological reactions. Unlike hormones which are secreted by cells, travel in the circulation to alter the behavior of distant cells, and thereby act as Endocrine signalling agents, these arachidonic acid metabolites act locally as Autocrine signalling and/or Paracrine signaling agents to regulate the behavior of their cells of origin or of nearby cells, respectively. In these roles, they may amplify or dampen, expand or contract cellular and tissue responses to disturbances.

12-Hydroxyheptadecatrienoic acid (also termed 12-HHT, 12(S)-hydroxyheptadeca-5Z,8E,10E-trienoic acid, or 12(S)-HHTrE) is a 17 carbon metabolite of the 20 carbon polyunsaturated fatty acid, arachidonic acid. It was discovered and structurally defined in 1973 by P. Wlodawer, Bengt I. Samuelsson, and M. Hamberg, as a product of arachidonic acid metabolism made by microsomes (i.e. endoplasmic reticulum) isolated from sheep seminal vesicle glands and by intact human platelets. 12-HHT is less ambiguously termed 12-(S)-hydroxy-5Z,8E,10E-heptadecatrienoic acid to indicate the S stereoisomerism of its 12-hydroxyl residue and the Z, E, and E cis-trans isomerism of its three double bonds. The metabolite was for many years thought to be merely a biologically inactive byproduct of prostaglandin synthesis. More recent studies, however, have attached potentially important activity to it.

20-Hydroxyeicosatetraenoic acid, also known as 20-HETE or 20-hydroxy-5Z,8Z,11Z,14Z-eicosatetraenoic acid, is an eicosanoid metabolite of arachidonic acid that has a wide range of effects on the vascular system including the regulation of vascular tone, blood flow to specific organs, sodium and fluid transport in the kidney, and vascular pathway remodeling. These vascular and kidney effects of 20-HETE have been shown to be responsible for regulating blood pressure and blood flow to specific organs in rodents; genetic and preclinical studies suggest that 20-HETE may similarly regulate blood pressure and contribute to the development of stroke and heart attacks. Additionally the loss of its production appears to be one cause of the human neurological disease, Hereditary spastic paraplegia. Preclinical studies also suggest that the overproduction of 20-HETE may contribute to the progression of certain human cancers, particularly those of the breast.

Multiplate multiple electrode aggregometry (MEA) is a test of platelet function in whole blood. The test can be used to diagnose platelet disorders, monitor antiplatelet therapy, and is also investigated as a potential predictor of transfusion requirements and bleeding risk in cardiac surgery.

Furegrelate, also known as 5-(3-pyridinylmethyl)benzofurancarboxylic acid, is a chemical compound with thromboxane enzyme inhibiting properties that was originally developed by Pharmacia Corporation as a drug to treat arrhythmias, ischaemic heart disorders, and thrombosis but was discontinued. It is commercially available in the form furegrelate sodium salt.