In chemistry, a structural isomer of a compound is another compound whose molecule has the same number of atoms of each element, but with logically distinct bonds between them. The term metamer was formerly used for the same concept.

In organic chemistry, the phenyl group, or phenyl ring, is a cyclic group of atoms with the formula C6H5, and is often represented by the symbol Ph. The phenyl group is closely related to benzene and can be viewed as a benzene ring, minus a hydrogen, which may be replaced by some other element or compound to serve as a functional group. A phenyl group has six carbon atoms bonded together in a hexagonal planar ring, five of which are bonded to individual hydrogen atoms, with the remaining carbon bonded to a substituent. Phenyl groups are commonplace in organic chemistry. Although often depicted with alternating double and single bonds, the phenyl group is chemically aromatic and has equal bond lengths between carbon atoms in the ring.

1,4-Dichlorobenzene (1,4-DCB, p-DCB, or para-dichlorobenzene, sometimes abbreviated as PDCB or para) is an aryl chloride and isomer of dichlorobenzene with the formula C6H4Cl2. This colorless solid has a strong odor. The molecule consists of a benzene ring with two chlorine atoms (replacing hydrogen atoms) on opposing sites of the ring.
In organic chemistry, an aryl halide is an aromatic compound in which one or more hydrogen atoms, directly bonded to an aromatic ring are replaced by a halide. Haloarenes are different from haloalkanes because they exhibit many differences in methods of preparation and properties. The most important members are the aryl chlorides, but the class of compounds is so broad that there are many derivatives and applications.

Triazines are a class of nitrogen-containing heterocycles. The parent molecules' molecular formula is C3H3N3. They exist in three isomeric forms, 1,3,5-triazines being common.

Chlorobenzene (abbreviated PhCl) is an aryl chloride and the simplest of the chlorobenzenes, consisting of a benzene ring substituted with one chlorine atom. Its chemical formula is C6H5Cl. This colorless, flammable liquid is a common solvent and a widely used intermediate in the manufacture of other chemicals.

1,3,5-Triazido-2,4,6-trinitrobenzene, also known as TATNB (triazidotrinitrobenzene) and TNTAZB (trinitrotriazidobenzene), is an aromatic high explosive composed of a benzene ring with three azido groups (-N3) and three nitro groups (-NO2) alternating around the ring, giving the chemical formula C6(N3)3(NO2)3. Its detonation velocity is 7,350 meters per second, which is comparable to TATB (triaminotrinitrobenzene).
Hexachlorobenzene, or perchlorobenzene, is an aryl chloride and a six-substituted chlorobenzene with the molecular formula C6Cl6. It is a fungicide formerly used as a seed treatment, especially on wheat to control the fungal disease bunt. It has been banned globally under the Stockholm Convention on Persistent Organic Pollutants.
Bromobenzenes are a group of aryl bromides/halobenzenes consisting of one or more bromine atoms as substituents on a benzene core. They have the formula C6H6–nBrn, where n = 1–6 is the number of bromine atoms. Depending on the number of bromine substituents, there may be several constitutional isomers possible.
Chlorobenzenes are a group of aryl chlorides/halobenzenes consisting of one or more chlorine atoms as substituents on a benzene core. They have the formula C6H6–nCln, where n = 1–6 is the number of chlorine atoms. Depending on the number of chlorine substituents, there may be several constitutional isomers possible.
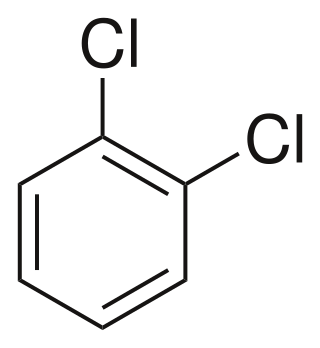
1,2-Dichlorobenzene, or orthodichlorobenzene (ODCB), is an aryl chloride and isomer of dichlorobenzene with the formula C6H4Cl2. This colourless liquid is poorly soluble in water but miscible with most organic solvents. It is a derivative of benzene, consisting of two adjacent chlorine atoms.
1,2,4-Trichlorobenzene is an organochlorine compound, one of three isomers of trichlorobenzene. It is a derivative of benzene with three chloride substituents. It is a colorless liquid used as a solvent for a variety of compounds and materials.

Trichlorobenzene (TCB) may refer to any of three chlorobenzenes with the molecular formula C6H3Cl3. Trichlorobenzenes are man-made chemical compounds that occur in three different forms. Even though the forms have the same molecular weight and molecular formulae, they are structurally different by the positions of the chlorine atoms attached to the benzene ring. 1,2,3-Trichlorobenzene and 1,3,5-trichlorobenzene are colorless solids, but 1,2,4-trichlorobenzene is a colorless oil. The isomers may also have different chemical and toxicological properties.

Hexachlorocyclohexane (HCH), C
6H
6Cl
6, is any of several polyhalogenated organic compounds consisting of a six-carbon ring with one chlorine and one hydrogen attached to each carbon. This structure has nine stereoisomers, which differ by the stereochemistry of the individual chlorine substituents on the cyclohexane. It is sometimes erroneously called "benzene hexachloride" (BHC). They have been used as models for analyzing the effects of different geometric positions of the large atoms with dipolar bonds on the stability of the cyclohexane conformation. The isomers are poisonous, pesticidal, and persistent organic pollutants, to varying degrees.

Pentachlorobenzene (PeCB) is an aryl chloride and a five-substituted chlorobenzene with the molecular formula C6HCl5 which is a chlorinated aromatic hydrocarbon. It consists of a benzene ring substituted with five chlorine atoms. PeCB was once used industrially for a variety of uses, but because of environmental concerns there are currently no large scale uses of PeCB. Pentachlorobenzene is a known persistent organic pollutant (POP) and banned globally by the Stockholm Convention on Persistent Organic Pollutants in 2009.

4-Nitrochlorobenzene is the organic compound with the formula ClC6H4NO2. It is a pale yellow solid. 4-Nitrochlorobenzene is a common intermediate in the production of a number of industrially useful compounds, including antioxidants commonly found in rubber. Other isomers with the formula ClC6H4NO2 include 2-nitrochlorobenzene and 3-nitrochlorobenzene.
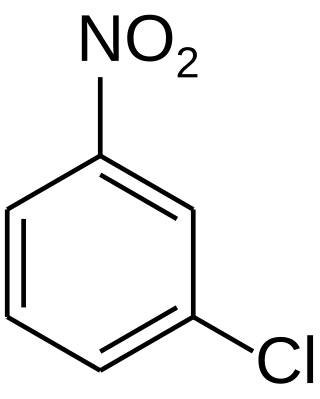
3-Nitrochlorobenzene is an organic compound with the formula C6H4ClNO2. It is a yellow crystalline solid that is important as a precursor to other compounds due to the two reactive sites present on the molecule.
1,2,3-Trichlorobenzene is an organochlorine compound with the chemical formula C6H3Cl3. This is one of three isomers of trichlorobenzene; the two others are 1,2,4-Trichlorobenzene and 1,3,5-Trichlorobenzene.
Tetrachlorobenzene is any of three isomeric chlorobenzenes with the molecular formula C6H2Cl4. They differ by the positions of the chlorine atoms around the ring. Tetrachlorobenzenes are colorless crystalline compounds.
Fluorobenzenes are a group of aryl fluorides/halobenzenes consisting of one or more fluorine atoms as substituents on a benzene core. They have the formula C6H6–nFn, where n = 1–6 is the number of fluorine atoms. Depending on the number of fluorine substituents, there may be several constitutional isomers possible.













