
Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) are fully or partly halogenated hydrocarbons that contain carbon (C), hydrogen (H), chlorine (Cl), and fluorine (F), produced as volatile derivatives of methane, ethane, and propane.

Freon is a registered trademark of the Chemours Company and generic descriptor for a number of halocarbon products. They are stable, nonflammable, low toxicity gases or liquids which have generally been used as refrigerants and as aerosol propellants. These include chlorofluorocarbons and hydrofluorocarbons, both of which cause ozone depletion and contribute to global warming. 'Freon' is the brand name for the refrigerants R-12, R-13B1, R-22, R-410A, R-502, and R-503 manufactured by The Chemours Company, and so is not used to label all refrigerants of this type. They emit a strong smell similar to acetone. Freon has been found to cause damage to human health when inhaled in large amounts. Studies have been conducted in the pursuit to find beneficial reuses for gases under the Freon umbrella as an alternative to disposal of the gas.

A refrigerant is a working fluid used in cooling, heating or reverse cooling and heating of air conditioning systems and heat pumps where they undergo a repeated phase transition from a liquid to a gas and back again. Refrigerants are heavily regulated because of their toxicity and flammability and the contribution of CFC and HCFC refrigerants to ozone depletion and that of HFC refrigerants to climate change.

Hydrofluorocarbons (HFCs) are synthetic organic compounds that contain fluorine and hydrogen atoms, and are the most common type of organofluorine compounds. Most are gases at room temperature and pressure. They are frequently used in air conditioning and as refrigerants; R-134a (1,1,1,2-tetrafluoroethane) is one of the most commonly used HFC refrigerants. In order to aid the recovery of the stratospheric ozone layer, HFCs were adopted to replace the more potent chlorofluorocarbons (CFCs), which were phased out from use by the Montreal Protocol, and hydrochlorofluorocarbons (HCFCs) which are presently being phased out. HFCs replaced older chlorofluorocarbons such as R-12 and hydrochlorofluorocarbons such as R-21. HFCs are also used in insulating foams, aerosol propellants, as solvents and for fire protection.

Difluoromethane, also called difluoromethylene, HFC-32Methylene Fluoride or R-32, is an organic compound of the dihalogenoalkane variety. Invented in 1964 by Hoechst AG (not Daikin) it has the formula of CH2F2. It is a colorless gas in the ambient atmosphere and is slightly soluble in water, with a high thermal stability. Due to the low melting and boiling point, (−136.0 and −51.6 °C [−212.8 and −60.9 °F; 137.2 and 221.6 K] respectively) contact with this compound may result in frostbite. In the United States, the Clean Air Act Section 111 on Volatile Organic Compounds (VOC) has listed difluoromethane as an exception (since 1997) from the definition of VOC due to its low production of tropospheric ozone. Difluoromethane is commonly used in endothermic processes such as refrigeration or air conditioning.
1,1,1,2-Tetrafluoroethane (also known as norflurane (INN), R-134a, Klea 134a, Freon 134a, Forane 134a, Genetron 134a, Green Gas, Florasol 134a, Suva 134a, HFA-134a, or HFC-134a) is a hydrofluorocarbon (HFC) and haloalkane refrigerant with thermodynamic properties similar to R-12 (dichlorodifluoromethane) but with insignificant ozone depletion potential and a lower 100-year global warming potential (1,430, compared to R-12's GWP of 10,900). It has the formula CF3CH2F and a boiling point of −26.3 °C (−15.34 °F) at atmospheric pressure. R-134a cylinders are colored light blue. A phaseout and transition to HFO-1234yf and other refrigerants, with GWPs similar to CO2, began in 2012 within the automotive market.
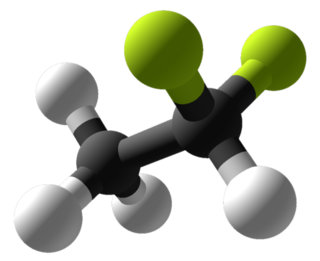
1,1-Difluoroethane, or DFE, is an organofluorine compound with the chemical formula C2H4F2. This colorless gas is used as a refrigerant, where it is often listed as R-152a (refrigerant-152a) or HFC-152a (hydrofluorocarbon-152a). It is also used as a propellant for aerosol sprays and in gas duster products. As an alternative to chlorofluorocarbons, it has an ozone depletion potential of zero, a lower global warming potential (124) and a shorter atmospheric lifetime (1.4 years).

A gas duster, also known as tinned wind, compressed air, or canned air, is a product used for cleaning or dusting electronic equipment and other sensitive devices that cannot be cleaned using water.
Trifluoroethane may refer to either of two isomeric fluorocarbons which differ by the location of attachment of the fluorine atoms:

Chlorodifluoromethane or difluoromonochloromethane is a hydrochlorofluorocarbon (HCFC). This colorless gas is better known as HCFC-22, or R-22, or CHClF
2. It was commonly used as a propellant and refrigerant. These applications were phased out under the Montreal Protocol in developed countries in 2020 due to the compound's ozone depletion potential (ODP) and high global warming potential (GWP), and in developing countries this process will be completed by 2030. R-22 is a versatile intermediate in industrial organofluorine chemistry, e.g. as a precursor to tetrafluoroethylene.
Fluoroform, or trifluoromethane, is the chemical compound with the formula CHF3. It is a hydrofluorocarbon as well as being a part of the haloforms, a class of compounds with the formula CHX3 with C3v symmetry. Fluoroform is used in diverse applications in organic synthesis. It is not an ozone depleter but is a greenhouse gas.
R-410A is a refrigerant used in air conditioning and heat pump applications. It is a zeotropic but near-azeotropic mixture of difluoromethane (CH2F2, called R-32) and pentafluoroethane (CHF2CF3, called R-125). R-410A is sold under the trademarked names AZ-20, EcoFluor R410, Forane 410A, Genetron R410A, Puron, and Suva 410A.
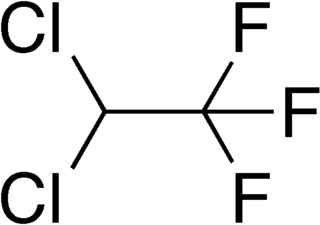
2,2-Dichloro-1,1,1-trifluoroethane or HCFC-123 is considered as an alternative to CFC-11 in low pressure refrigeration and HVAC systems, and should not be used in foam blowing processes or solvent applications. It is also the primary component of the Halotron I fire-extinguishing mixture.
Organofluorine chemistry describes the chemistry of organofluorine compounds, organic compounds that contain a carbon–fluorine bond. Organofluorine compounds find diverse applications ranging from oil and water repellents to pharmaceuticals, refrigerants, and reagents in catalysis. In addition to these applications, some organofluorine compounds are pollutants because of their contributions to ozone depletion, global warming, bioaccumulation, and toxicity. The area of organofluorine chemistry often requires special techniques associated with the handling of fluorinating agents.
Natural refrigerants are considered substances that serve as refrigerants in refrigeration systems. They are alternatives to synthetic refrigerants such as chlorofluorocarbon (CFC), hydrochlorofluorocarbon (HCFC), and hydrofluorocarbon (HFC) based refrigerants. Unlike other refrigerants, natural refrigerants can be found in nature and are commercially available thanks to physical industrial processes like fractional distillation, chemical reactions such as Haber process and spin-off gases. The most prominent of these include various natural hydrocarbons, carbon dioxide, ammonia, and water. Natural refrigerants are preferred actually in new equipment to their synthetic counterparts for their presumption of higher degrees of sustainability. With the current technologies available, almost 75 percent of the refrigeration and air conditioning sector has the potential to be converted to natural refrigerants.

Chloropentafluoroethane is a chlorofluorocarbon (CFC) once used as a refrigerant and also known as R-115 and CFC-115. Its production and consumption has been banned since 1 January 1996 under the Montreal Protocol because of its high ozone depletion potential and very long lifetime when released into the environment. CFC-115 is also a potent greenhouse gas.

1,1-Dichloro-1-fluoroethane is a haloalkane with the formula C
2H
3Cl
2F. It is one of the three isomers of dichlorofluoroethane. It belongs to the hydrochlorofluorocarbon (HCFC) family of man-made compounds that contribute significantly to both ozone depletion and global warming when released into the environment.

1-Chloro-1,1-difluoroethane (HCFC-142b) is a haloalkane with the chemical formula CH3CClF2. It belongs to the hydrochlorofluorocarbon (HCFC) family of man-made compounds that contribute significantly to both ozone depletion and global warming when released into the environment. It is primarily used as a refrigerant where it is also known as R-142b and by trade names including Freon-142b.
Fluorinated gases (F-gases) are a group of gases containing fluorine. They are divided into several types, the main of those are hydrofluorocarbons (HFCs), perfluorocarbons (PFCs), sulphur hexafluoride (SF6). They are used in refrigeration, air conditioning, heat pumps, fire suppression, electronics, aerospace, magnesium industry, foam and high voltage switchgear. As they are greenhouse gases with a strong global warming potential, their use is regulated.

1,1-Dichlorotetrafluoroethane is a chlorofluorocarbon also known as CFC-114a or R114a by American Society of Heating, Refrigerating, and Air Conditioning Engineers. It has two chlorine atoms on one carbon atom and none on the other. It is one of two isomers of dichlorotetrafluoroethane, the other being 1,2-dichlorotetrafluoroethane, also known as CFC-114.
















