
In organic chemistry, a dicarbonyl is a molecule containing two carbonyl groups. Although this term could refer to any organic compound containing two carbonyl groups, it is used more specifically to describe molecules in which both carbonyls are in close enough proximity that their reactivity is changed, such as 1,2-, 1,3-, and 1,4-dicarbonyls. Their properties often differ from those of monocarbonyls, and so they are usually considered functional groups of their own. These compounds can have symmetrical or unsymmetrical substituents on each carbonyl, and may also be functionally symmetrical or unsymmetrical.
Cyclohexa-1,3-diene (also known as Benzane) is an organic compound with the formula (C2H4)(CH)4. It is a colorless, flammable liquid. Its refractive index is 1.475 (20 °C, D). It is one of two isomers of cyclohexadiene, the other being 1,4-cyclohexadiene.

A silabenzene is a heteroaromatic compound containing one or more silicon atoms instead of carbon atoms in benzene. A single substitution gives silabenzene proper; additional substitutions give a disilabenzene, trisilabenzene, etc.
In organic chemistry, dihydroxybenzenes (benzenediols) are organic compounds in which two hydroxyl groups are substituted onto a benzene ring. These aromatic compounds are classed as phenols. There are three structural isomers: 1,2-dihydroxybenzene is commonly known as catechol, 1,3-dihydroxybenzene is commonly known as resorcinol, and 1,4-dihydroxybenzene is commonly known as hydroquinone.
There are three distinct chemical compounds which are dichlorobenzenes:

The Controlled Drugs and Substances Act is Canada's federal drug control statute. Passed in 1996 under Prime Minister Jean Chrétien's government, it repeals the Narcotic Control Act and Parts III and IV of the Food and Drugs Act, and establishes eight Schedules of controlled substances and two Classes of precursors. It provides that "The Governor in Council may, by order, amend any of Schedules I to VIII by adding to them or deleting from them any item or portion of an item, where the Governor in Council deems the amendment to be necessary in the public interest."
Thiazine is an organic compound containing a ring of four carbon, one nitrogen and one sulfur atom. There are three isomers of thiazine, 1,2-thiazine, 1,3-thiazine, and 1,4-thiazine, which differ by the arrangement of the nitrogen and sulfur atoms in the ring.

In organic chemistry, dimethoxybenzene is an organic compound which is derived from benzene by substituting two methoxy groups. Dimethoxybenzene comes in three structural isomers:
Bromobenzenes are a group of aryl bromides/halobenzenes consisting of one or more bromine atoms as substituents on a benzene core. They have the formula C6H6–nBrn, where n = 1–6 is the number of bromine atoms. Depending on the number of bromine substituents, there may be several constitutional isomers possible.
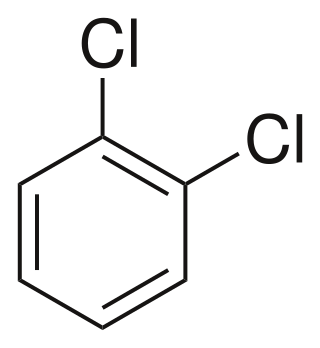
1,2-Dichlorobenzene, or orthodichlorobenzene (ODCB), is an aryl chloride and isomer of dichlorobenzene with the formula C6H4Cl2. This colourless liquid is poorly soluble in water but miscible with most organic solvents. It is a derivative of benzene, consisting of two adjacent chlorine atoms.

A dithiane is a heterocyclic compound composed of a cyclohexane core structure wherein two methylene bridges are replaced by sulfur. The three isomeric parent heterocycles are 1,2-dithiane, 1,3-dithiane and 1,4-dithiane. They are all colorless solids.

Divinylbenzene (DVB) is an organic compound with the chemical formula C6H4(CH=CH2)2 and structure H2C=CH−C6H4−HC=CH2. It is related to styrene by the addition of a second vinyl group. It is a colorless liquid manufactured by the thermal dehydrogenation of isomeric diethylbenzenes. Under synthesis conditions, o-divinylbenzene converts to naphthalene and thus is not a component of the usual mixtures of DVB.
1,4-Dibromobenzene (p-dibromobenzene) is an aryl bromide and isomer of dibromobenzene that is solid at room temperature. It has a strong smell similar to that of the lighter chlorine analogue. It can be used as a precursor to the dye 6,6-Dibromoindigo.
Dibromobenzenes are a group of bromobenzenes with the formula C6H4Br2, consisting of two bromine atoms bonded to a central benzene ring.
Dinitrobenzenes are nitrobenzenes composed of a benzene ring and two nitro group (-NO2) substituents. The three possible arrangements of the nitro groups afford three isomers, 1,2-dinitrobenzene, 1,3-dinitrobenzene, and 1,4-dinitrobenzene. Each isomer has the chemical formula C6H4N2O4 and a molar mass of about 168.11 g/mol. 1,3-Dinitrobenzene is the most common isomer and it is used in the manufacture of explosives.

Diethylbenzene (DEB) refers to any of three isomers with the formula C6H4(C2H5)2. Each consists of a benzene ring and two ethyl substituents. The meta and para have the greater commercial significance. All are colorless liquids.
The C2benzenes are a class of organic aromatic compounds which contain a benzene ring and two other carbon atoms. For the hydrocarbons with no further unsaturation, there are four isomers. There are three xylenes and one ethylbenzene. The substances are:

The C3-benzenes are a class of organic aromatic compounds which contain a benzene ring and three other carbon atoms. For the hydrocarbons with no further unsaturation, there are four isomers. The chemical formula for all the saturated isomers is C9H12.
The diisopropylbenzenes(DIPB) are organic compounds with the formula C6H4(CH(CH3)2)2. Three isomers exist: 1,2- 1,3-, and 1,4-diisopropylbenzene. All are colorless liquids, immiscible in water, with similar boiling points. They are classified are aromatic hydrocarbons bearing a pair of isopropyl (CH(CH3)2) substituents. DIPB has been referred to as "a common diluent" alongside hexane.
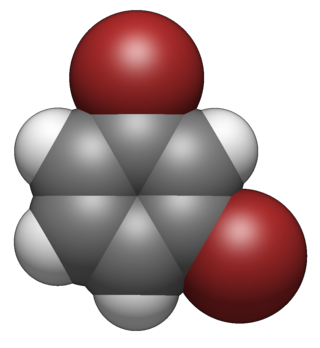
1,3-Dibromobenzene (m-dibromobenzene) is an aryl bromide and isomer of dibromobenzene that is a colorless liquid at room temperature.











