
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation. It is a form of luminescence. In most cases, the emitted light has a longer wavelength, and therefore a lower photon energy, than the absorbed radiation. A perceptible example of fluorescence occurs when the absorbed radiation is in the ultraviolet region of the electromagnetic spectrum, while the emitted light is in the visible region; this gives the fluorescent substance a distinct color that can only be seen when the substance has been exposed to UV light. Fluorescent materials cease to glow nearly immediately when the radiation source stops, unlike phosphorescent materials, which continue to emit light for some time after.

Optical brighteners, optical brightening agents (OBAs), fluorescent brightening agents (FBAs), or fluorescent whitening agents (FWAs), are chemical compounds that absorb light in the ultraviolet and violet region of the electromagnetic spectrum, and re-emit light in the blue region through the phenomenon of fluorescence. These additives are often used to enhance the appearance of color of fabric and paper, causing a "whitening" effect; they make intrinsically yellow/orange materials look less so, by compensating the deficit in blue and purple light reflected by the material, with the blue and purple optical emission of the fluorophore.

Acridine is an organic compound and a nitrogen heterocycle with the formula C13H9N. Acridines are substituted derivatives of the parent ring. It is a planar molecule that is structurally related to anthracene with one of the central CH groups replaced by nitrogen. Like the related molecules pyridine and quinoline, acridine is mildly basic. It is an almost colorless solid, which crystallizes in needles. There are few commercial applications of acridines; at one time acridine dyes were popular, but they are now relegated to niche applications, such as with acridine orange. The name is a reference to the acrid odour and acrid skin-irritating effect of the compound.

A blacklight, also called a UV-A light, Wood's lamp, or ultraviolet light, is a lamp that emits long-wave (UV-A) ultraviolet light and very little visible light. One type of lamp has a violet filter material, either on the bulb or in a separate glass filter in the lamp housing, which blocks most visible light and allows through UV, so the lamp has a dim violet glow when operating. Blacklight lamps which have this filter have a lighting industry designation that includes the letters "BLB". This stands for "blacklight blue". A second type of lamp produces ultraviolet but does not have the filter material, so it produces more visible light and has a blue color when operating. These tubes are made for use in "bug zapper" insect traps, and are identified by the industry designation "BL". This stands for "blacklight".

Richard "Ric" Berger (1894–1984) was a Swiss professor of design, decoration, and art history. He is best known for his numerous newspaper articles about historical monuments, mainly in the French-speaking part of Switzerland, including his own drawings of the buildings. Through these articles, he contributed to an increased interest in historical monuments and settings among many hitherto uninformed people, and probably also contributed indirectly to a wider interest in preserving and saving historical monuments from destruction.

Staining is a technique used to enhance contrast in samples, generally at the microscopic level. Stains and dyes are frequently used in histology, in cytology, and in the medical fields of histopathology, hematology, and cytopathology that focus on the study and diagnoses of diseases at the microscopic level. Stains may be used to define biological tissues, cell populations, or organelles within individual cells.
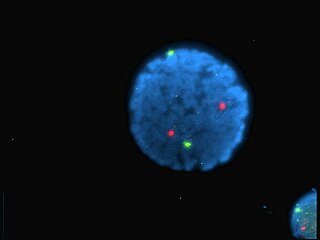
A fluorophore is a fluorescent chemical compound that can re-emit light upon light excitation. Fluorophores typically contain several combined aromatic groups, or planar or cyclic molecules with several π bonds.
The Bernthsen acridine synthesis is the chemical reaction of a diarylamine heated with a carboxylic acid and zinc chloride to form a 9-substituted acridine.
Proflavine, also called proflavin and diaminoacridine, is an acriflavine derivative, a disinfectant bacteriostatic against many gram-positive bacteria. It has been used in the form of the dihydrochloride and hemisulfate salts as a topical antiseptic, and was formerly used as a urinary antiseptic.
IARC group 3 substances, chemical mixtures and exposure circumstances are those that can not be classified in regard to their carcinogenicity to humans by the International Agency for Research on Cancer (IARC). This category is used most commonly for agents, mixtures and exposure circumstances for which the level of evidence of carcinogenicity is inadequate in humans and inadequate or limited in experimental animals. Exceptionally, agents (mixtures) for which the evidence of carcinogenicity is inadequate in humans, but sufficient in experimental animals may be placed in this category when there is strong evidence that the mechanism of carcinogenicity in experimental animals does not operate in humans. Agents, mixtures and exposure circumstances that do not fall into any other group are also placed in this category.

Acriflavine is a topical antiseptic. It has the form of an orange or brown powder. It may be harmful in the eyes or if inhaled. It is a dye and it stains the skin and may irritate. The hydrochloride form is more irritating than the neutral form. It is derived from acridine. Commercial preparations are often mixtures with proflavine. It is known by a variety of commercial names.

Heike Gabriela Drechsler is a German former track and field athlete who represented East Germany and later Germany. One of the most successful long jumpers of all-time, she is a former world record holder and ranks third on the all-time list with her legal best of 7.48 metres in 1988. Her marginally wind-assisted jump of 7.63 metres (+2.1) in 1992 at altitude in Sestriere, is still the furthest a woman has ever long jumped. She is the only woman who has won two Olympic gold medals in the long jump, winning in 1992 and 2000.

Lily Kronberger, also spelled Lili Kronberger, was a Hungarian figure skater competitive during the early years of modern figure skating. She was Hungary’s first World Champion.

Benzanthrone (BZA) is a polycyclic aromatic hydrocarbon. It is a yellow solid. Its derivatives are used as a dyestuff intermediate for anthraquinone-based dyes. Dehydrogenative coupling gives violanthrone. It is prepared by reduction of anthroquinone to anthrone followed by alkylation with a mixture of glycerol and sulfuric acid.

Acridine orange is an organic compound that serves as a nucleic acid-selective fluorescent dye with cationic properties useful for cell cycle determination. Acridine orange is cell-permeable, which allows the dye to interact with DNA by intercalation, or RNA via electrostatic attractions. When bound to DNA, acridine orange is very similar spectrally to an organic compound known as fluorescein. Acridine orange and fluorescein have a maximum excitation at 502nm and 525 nm (green). When acridine orange associates with RNA, the fluorescent dye experiences a maximum excitation shift from 525 nm (green) to 460 nm (blue). The shift in maximum excitation also produces a maximum emission of 650 nm (red). Acridine orange is able to withstand low pH environments, allowing the fluorescent dye to penetrate acidic organelles such as lysosomes and phagolysosomes that are membrane-bound organelles essential for acid hydrolysis or for producing products of phagocytosis of apoptotic cells. Acridine orange is used in epifluorescence microscopy and flow cytometry. The ability to penetrate the cell membranes of acidic organelles and cationic properties of acridine orange allows the dye to differentiate between various types of cells. The shift in maximum excitation and emission wavelengths provides a foundation to predict the wavelength at which the cells will stain.
Franz "Bimbo" Binder was an Austrian football player and coach who played as a forward. Internationally he represented the Austria national football team and the Germany national football team during the Anschluss.

Phenanthridine is a nitrogen heterocyclic compound that is the basis of DNA-binding fluorescent dyes through intercalation. Examples of such dyes are ethidium bromide and propidium iodide. It is an isomer of acridine.

The Lurgrotte karst cave is the largest cave in the Eastern Alps of Styria, Austria. It is located about 16 km (9.9 mi) north of Graz and crosses the Tannenben karst region. The cave has two accessible entrances, one at the village of Semriach and the other at the village of Peggau. At the Semriach entrance, the Lur River sinks into the cave. At the Peggau entrance, the Schmelz River emerges from within the cave, flowing to the west and eventually joining the Mur River.

Archduchess Maria Antonietta of Austria, Princess of Tuscany was a member of the House of Habsburg-Lorraine. She served as the Princess-Abbess of the Theresian Royal and Imperial Convent in Hradčany from 1881 until her death in 1883.
Interlingue and Interlingua are constructed international auxiliary languages.



















