
A carcinogen is any substance, radionuclide, or radiation that promotes carcinogenesis, the formation of cancer. This may be due to the ability to damage the genome or to the disruption of cellular metabolic processes. Several radioactive substances are considered carcinogens, but their carcinogenic activity is attributed to the radiation, for example gamma rays and alpha particles, which they emit. Common examples of non-radioactive carcinogens are inhaled asbestos, certain dioxins, and tobacco smoke. Although the public generally associates carcinogenicity with synthetic chemicals, it is equally likely to arise from both natural and synthetic substances. Carcinogens are not necessarily immediately toxic; thus, their effect can be insidious.
Coal tar is a thick dark liquid which is a by-product of the production of coke and coal gas from coal. It is a type of creosote. It has both medical and industrial uses. Medicinally it is a topical medication applied to skin to treat psoriasis and seborrheic dermatitis (dandruff). It may be used in combination with ultraviolet light therapy. Industrially it is a railroad tie preservative and used in the surfacing of roads. Coal tar was listed as a known human carcinogen in the first Report on Carcinogens from the U.S. Federal Government.
Sodium laureth sulfate (SLES), an accepted contraction of sodium lauryl ether sulfate (SLES), also called sodium alkylethersulfate, is an anionic detergent and surfactant found in many personal care products and for industrial uses. SLES is an inexpensive and very effective foaming agent. SLES, sodium lauryl sulfate (SLS), ammonium lauryl sulfate (ALS), and sodium pareth sulfate are surfactants that are used in many cosmetic products for their cleaning and emulsifying properties. It is derived from palm kernel oil or coconut oil. In herbicides, it is used as a surfactant to improve absorption of the herbicidal chemicals and reduces time the product takes to be rainfast, when enough of the herbicidal agent will be absorbed.

Formaldehyde is a naturally occurring organic compound with the formula CH2O and structure H−CHO. The pure compound is a pungent, colourless gas that polymerises spontaneously into paraformaldehyde, hence it is stored as an aqueous solution (formalin), which is also used to store animal specimens. It is the simplest of the aldehydes. The common name of this substance comes from its similarity and relation to formic acid.
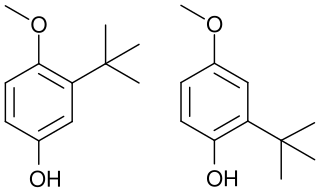
Butylated hydroxyanisole (BHA) is an antioxidant consisting of a mixture of two isomeric organic compounds, 2-tert-butyl-4-hydroxyanisole and 3-tert-butyl-4-hydroxyanisole. It is prepared from 4-methoxyphenol and isobutylene. It is a waxy solid used as a food additive with the E number E320. The primary use for BHA is as an antioxidant and preservative in food, food packaging, animal feed, cosmetics, rubber, and petroleum products. BHA also is commonly used in medicines, such as cholecalciferol, isotretinoin, lovastatin, and simvastatin, among others.
Acetaldehyde (IUPAC systematic name ethanal) is an organic chemical compound with the formula CH3CHO, sometimes abbreviated by chemists as MeCHO (Me = methyl). It is a colorless liquid or gas, boiling near room temperature. It is one of the most important aldehydes, occurring widely in nature and being produced on a large scale in industry. Acetaldehyde occurs naturally in coffee, bread, and ripe fruit, and is produced by plants. It is also produced by the partial oxidation of ethanol by the liver enzyme alcohol dehydrogenase and is a contributing cause of hangover after alcohol consumption. Pathways of exposure include air, water, land, or groundwater, as well as drink and smoke. Consumption of disulfiram inhibits acetaldehyde dehydrogenase, the enzyme responsible for the metabolism of acetaldehyde, thereby causing it to build up in the body.

Mineral oil is any of various colorless, odorless, light mixtures of higher alkanes from a mineral source, particularly a distillate of petroleum, as distinct from usually edible vegetable oils.
Acrylonitrile is an organic compound with the formula CH2CHCN and the structure H2C=CH−C≡N. It is a colorless, volatile liquid although commercial samples can be yellow due to impurities. It has a pungent odor of garlic or onions. In terms of its molecular structure, it consists of a vinyl group linked to a nitrile. It is an important monomer for the manufacture of useful plastics such as polyacrylonitrile. It is reactive and toxic at low doses. Acrylonitrile was first synthesized by the French chemist Charles Moureu (1863–1929) in 1893.

Nitrobenzene is an organic compound with the chemical formula C6H5NO2. It is a water-insoluble pale yellow oil with an almond-like odor. It freezes to give greenish-yellow crystals. It is produced on a large scale from benzene as a precursor to aniline. In the laboratory, it is occasionally used as a solvent, especially for electrophilic reagents.

1,4-Dioxane is a heterocyclic organic compound, classified as an ether. It is a colorless liquid with a faint sweet odor similar to that of diethyl ether. The compound is often called simply dioxane because the other dioxane isomers are rarely encountered.

The International Agency for Research on Cancer is an intergovernmental agency forming part of the World Health Organization of the United Nations. Its role is to conduct and coordinate research into the causes of cancer. It also collects and publishes surveillance data regarding the occurrence of cancer worldwide.
Substances, mixtures, and exposure circumstances in this list have been classified as group 1 by the International Agency for Research on Cancer (IARC): The agent (mixture) is carcinogenic to humans. The exposure circumstance entails exposures that are carcinogenic to humans. This category is used when there is sufficient evidence of carcinogenicity in humans. Exceptionally, an agent (mixture) may be placed in this category when evidence of carcinogenicity in humans is less than sufficient but there is sufficient evidence of carcinogenicity in experimental animals and strong evidence in exposed humans that the agent (mixture) acts through a relevant mechanism of carcinogenicity.
Substances, mixtures and exposure circumstances in this list have been classified by the International Agency for Research on Cancer (IARC) as group 2B: The agent (mixture) is "possibly carcinogenic to humans". The exposure circumstance entails exposures that are possibly carcinogenic to humans. This category is used for agents, mixtures and exposure circumstances for which there is limited evidence of carcinogenicity in humans and less than sufficient evidence of carcinogenicity in experimental animals. It may also be used when there is inadequate evidence of carcinogenicity in humans but there is sufficient evidence of carcinogenicity in experimental animals. In some instances, an agent, mixture or exposure circumstance for which there is inadequate evidence of carcinogenicity in humans but limited evidence of carcinogenicity in experimental animals together with supporting evidence from other relevant data may be placed in this group. Further details can be found in the preamble to the IARC Monographs.

Diethyl sulfate (DES) is a highly toxic, combustible, and likely carcinogenic chemical compound with the formula (C2H5)2SO4. It occurs as a colorless, oily liquid with a faint peppermint odor and is corrosive to tissue and metals. Diethyl sulfate is used as an alkylating agent to prepare ethyl derivatives of phenols, amines, and thiols. It is used to manufacture dyes and textiles.

Potassium bromate (KBrO3), is a bromate of potassium and takes the form of white crystals or powder. It is a strong oxidizing agent.
Cocamidopropyl betaine (CAPB) is a mixture of closely related organic compounds derived from coconut oil and dimethylaminopropylamine. CAPB is available as a viscous pale yellow solution and it is used as a surfactant in personal care products. The name reflects that the major part of the molecule, the lauric acid group, is derived from coconut oil. Cocamidopropyl betaine to a significant degree has replaced cocamide DEA.
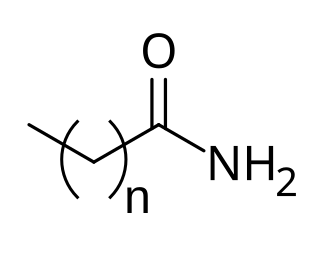
Cocamide is a mixture of amides manufactured from the fatty acids obtained from coconut oil. As coconut oil contains about 50% of lauric acid, in formulas only the 12-carbon chains tend to be considered. Therefore the formula of cocamide can be written as CH3(CH2)10CONH2, though the number of carbon atoms in the chains varies.

Diethanolamine, often abbreviated as DEA or DEOA, is an organic compound with the formula HN(CH2CH2OH)2. Pure diethanolamine is a white solid at room temperature, but its tendencies to absorb water and to supercool meaning that it is often encountered as a colorless, viscous liquid. Diethanolamine is polyfunctional, being a secondary amine and a diol. Like other organic amines, diethanolamine acts as a weak base. Reflecting the hydrophilic character of the secondary amine and hydroxyl groups, DEA is soluble in water. Amides prepared from DEA are often also hydrophilic. In 2013, the chemical was classified by the International Agency for Research on Cancer as "possibly carcinogenic to humans" (Group 2B).

In molecular genetics, a DNA adduct is a segment of DNA bound to a cancer-causing chemical. This process could lead to the development of cancerous cells, or carcinogenesis. DNA adducts in scientific experiments are used as biomarkers of exposure. They are especially useful in quantifying an organism's exposure to a carcinogen. The presence of such an adduct indicates prior exposure to a potential carcinogen, but it does not necessarily indicate the presence of cancer in the subject animal.

Lorenzo (Renzo) Tomatis was an Italian physician and experimental oncologist who researched carcinogenesis and its primary prevention















