
Voacangine is an alkaloid found predominantly in the root bark of the Voacanga africana tree, as well as in other plants such as Tabernanthe iboga, Tabernaemontana africana, Trachelospermum jasminoides, Tabernaemontana divaricata and Ervatamia yunnanensis. It is an iboga alkaloid which commonly serves as a precursor for the semi-synthesis of ibogaine. It has been demonstrated in animals to have similar anti-addictive properties to ibogaine itself. It also potentiates the effects of barbiturates. Under UV-A and UV-B light its crystals fluoresce blue-green, and it is soluble in ethanol.

Indole alkaloids are a class of alkaloids containing a structural moiety of indole; many indole alkaloids also include isoprene groups and are thus called terpene indole or secologanin tryptamine alkaloids. Containing more than 4100 known different compounds, it is one of the largest classes of alkaloids. Many of them possess significant physiological activity and some of them are used in medicine. The amino acid tryptophan is the biochemical precursor of indole alkaloids.
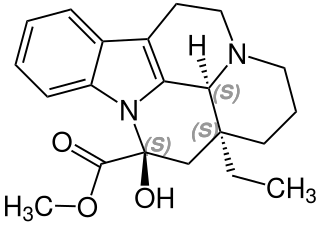
Vincamine is a monoterpenoid indole alkaloid found in the leaves of Vinca minor, comprising about 25–65% of its indole alkaloids by weight. It can also be synthesized from related alkaloids.

Tabernaemontana corymbosa is a species of plant in the family Apocynaceae. It is native to Brunei, China, Indonesia, Laos, Malaysia, Myanmar, Singapore, Thailand, and Vietnam. Glossy green leaves and faintly sweet scented flower. Flowers continuously all year. Frost tolerant. Grows to about 2 metres. Likes full sun to part shade. A number of cultivars are available.
Strictosidine synthase (EC 4.3.3.2) is an enzyme in alkaloid biosynthesis that catalyses the condensation of tryptamine with secologanin to form strictosidine in a formal Pictet–Spengler reaction:

Coronaridine, also known as 18-carbomethoxyibogamine, is an alkaloid found in Tabernanthe iboga and related species, including Tabernaemontana divaricata for which it was named.

Ibogamine is an anti-convulsant, anti-addictive, CNS stimulant alkaloid found in Tabernanthe iboga and Crepe Jasmine. Basic research related to how addiction affects the brain has used this chemical.

Voacamine, also known under the older names voacanginine and vocamine, is a naturally occurring dimeric indole alkaloid of the secologanin type, found in a number of plants, including Voacanga africana and Tabernaemontana divaricata. It is approved for use as an antimalarial drug in several African countries. Voacamine exhibits cannabinoid CB1 receptor antagonistic activity.
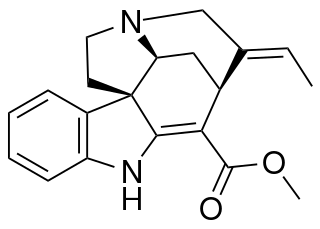
Akuammicine is a monoterpene indole alkaloid of the Vinca sub-group. It is found in the Apocynaceae family of plants including Picralima nitida, Vinca minor and the Aspidosperma.

Catharanthine is a terpene indole alkaloid produced by the medicinal plant Catharanthus roseus and Tabernaemontana divaricata. Catharanthine is derived from strictosidine, but the exact mechanism by which this happens is currently unknown. Catharanthine is one of the two precursors that form vinblastine, the other being vindoline.

Affinine is a monoterpenoid indole alkaloid which can be isolated from plants of the genus Tabernaemontana. Structurally it can be considered a member of the vobasine alkaloid family and may be synthesized from tryptophan. Limited pharmacological testing has indicated that it may be an effective inhibitor of both acetylcholinesterase and butyrylcholinesterase.
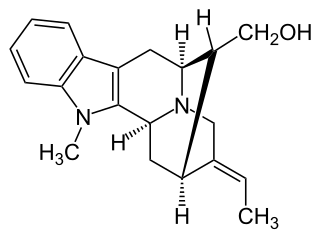
Affinisine is a monoterpenoid indole alkaloid which can be isolated from plants of the genus Tabernaemontana. Structurally, it can be considered a member of the sarpagine alkaloid family and may be synthesized from tryptophan via a Pictet-Spengler reaction.
Iboga-type alkaloids are a set of monoterpene indole alkaloids comprising naturally occurring compounds found in Tabernanthe and Tabernaemontana, as well as synthetic structural analogs. Naturally occurring iboga-type alkaloids include ibogamine, ibogaine, tabernanthine, and other substituted ibogamines. Many iboga-type alkaloids display biological activities such as cardiac toxicity and psychoactive effects, and some have been studied as potential treatments for drug addiction.

Apparicine is a monoterpenoid tricyclic indole alkaloid. It is named after Apparicio Duarte, a Brazilian botanist who studied the Aspidosperma species from which apparicine was first isolated. It was the first member of the vallesamine group of indole alkaloids to be isolated and have its structure established, which was first published in 1965. It has also been known by the synonyms gomezine, pericalline, and tabernoschizine.

Tabernaemontanine is a naturally occurring monoterpene indole alkaloid found in several species in the genus Tabernaemontana including Tabernaemontana divaricata.

Conophylline is a autophagy inducing vinca alkaloid found in plants of the genus Tabernaemontana. Among the many functional groups in this molecule is an epoxide: the compound where that ring is replaced with a double bond is called conophyllidine and this co-occurs in the same plants.

Ervatinine is an anti-corrosive indole alkaloid from Ervatamia coronaria.

Vobasine is a naturally occurring monoterpene indole alkaloid found in several species in the genus Tabernaemontana including Tabernaemontana divaricata.

Voacristine is a indole alkaloid occurring in Voacanga and Tabernaemontana genus. It is also an iboga type alkaloid.
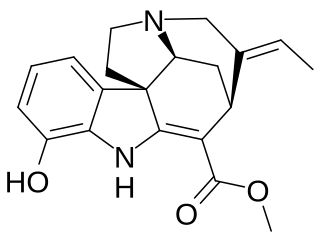
Vinervine is a monoterpene indole alkaloid of the Vinca sub-group. It is a derivative of akuammicine, with one additional hydroxy (OH) group in the indole portion, hence it is also known as 12-hydroxyakuammicine.



















