
An acid is a molecule or ion capable of donating a proton (hydrogen ion H+) (a Brønsted–Lowry acid), or, alternatively, capable of forming a covalent bond with an electron pair (a Lewis acid).

Amino acids are organic compounds that contain amine (–NH2) and carboxyl (–COOH) functional groups, along with a side chain (R group) specific to each amino acid. The key elements of an amino acid are carbon (C), hydrogen (H), oxygen (O), and nitrogen (N), although other elements are found in the side chains of certain amino acids. About 500 naturally occurring amino acids are known (though only 20 appear in the genetic code) and can be classified in many ways. They can be classified according to the core structural functional groups' locations as alpha- (α-), beta- (β-), gamma- (γ-) or delta- (δ-) amino acids; other categories relate to polarity, pH level, and side chain group type (aliphatic, acyclic, aromatic, containing hydroxyl or sulfur, etc.). In the form of proteins, amino acid residues form the second-largest component (water is the largest) of human muscles and other tissues. Beyond their role as residues in proteins, amino acids participate in a number of processes such as neurotransmitter transport and biosynthesis.
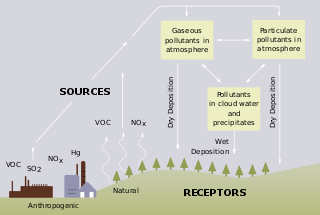
Acid rain is a rain or any other form of precipitation that is unusually acidic, meaning that it has elevated levels of hydrogen ions. It can have harmful effects on plants, aquatic animals, and infrastructure. Acid rain is caused by emissions of sulfur dioxide and nitrogen oxide, which react with the water molecules in the atmosphere to produce acids. Some governments have made efforts since the 1970s to reduce the release of sulfur dioxide and nitrogen oxide into the atmosphere with positive results. Nitrogen oxides can also be produced naturally by lightning strikes, and sulfur dioxide is produced by volcanic eruptions. Acid rain has been shown to have adverse impacts on forests, freshwaters, and soils, killing insect and aquatic life-forms, causing paint to peel, corrosion of steel structures such as bridges, and weathering of stone buildings and statues as well as having impacts on human health.

A carboxylic acid is an organic compound that contains a carboxyl group attached to an R-group. The general formula of a carboxylic acid is R–COOH, with R referring to the alkyl group. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion.

In chemistry, an ester is a chemical compound derived from an acid in which at least one –OH (hydroxyl) group is replaced by an –O–alkyl (alkoxy) group. Usually, esters are derived from substitution reaction of a carboxylic acid and an alcohol. Glycerides, which are fatty acid esters of glycerol, are important esters in biology, being one of the main classes of lipids, and making up the bulk of animal fats and vegetable oils. Esters with low molecular weight are commonly used as fragrances and found in essential oils and pheromones. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties, while polyesters are important plastics, with monomers linked by ester moieties. Esters usually have a sweet smell and are considered high-quality solvents for a broad array of plastics, plasticizers, resins, and lacquers. They are also one of the largest classes of synthetic lubricants on the commercial market.
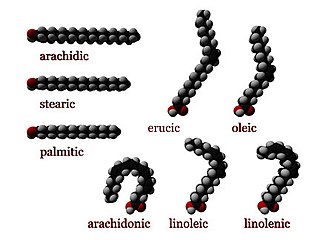
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with a long aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, from 4 to 28. Fatty acids are usually not found in organisms in their standalone form, but instead exist as three main classes of esters: triglycerides, phospholipids, and cholesteryl esters. In any of these forms, fatty acids are both important dietary sources of fuel for animals and they are important structural components for cells.

In nutrition, biology, and chemistry, fat usually means any ester of fatty acids, or a mixture of such compounds; most commonly those that occur in living beings or in food.
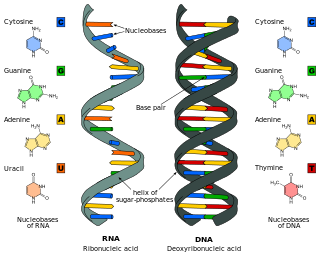
Nucleic acids are the biopolymers, or large biomolecules, essential to all known forms of life. The term nucleic acid is the overall name for DNA and RNA. They are composed of nucleotides, which are the monomers made of three components: a 5-carbon sugar, a phosphate group and a nitrogenous base. If the sugar is a compound ribose, the polymer is RNA ; if the sugar is derived from ribose as deoxyribose, the polymer is DNA.
Nitric acid (HNO3), also known as aqua fortis (Latin for "strong water") and spirit of niter, is a highly corrosive mineral acid.

In chemistry, pH (, denoting 'potential of hydrogen' or 'power of hydrogen') is a scale used to specify the acidity or basicity of an aqueous solution. Acidic solutions (solutions with higher concentrations of H+ ions) are measured to have lower pH values than basic or alkaline solutions.

Salicylic acid (from Latin salix, willow tree) is a lipophilic monohydroxybenzoic acid, a type of phenolic acid, and a beta hydroxy acid (BHA). It has the formula C7H6O3. This colorless crystalline organic acid is widely used in organic synthesis and functions as a plant hormone. It is derived from the metabolism of salicin.

Sulfuric acid (American spelling) or sulphuric acid (British spelling), also known as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with molecular formula H2SO4. It is a colourless, odourless, and viscous liquid that is soluble in water and is synthesized in reactions that are highly exothermic.

Citric acid is a weak organic acid that has the molecular formula C6H8O7. It occurs naturally in citrus fruits. In biochemistry, it is an intermediate in the citric acid cycle, which occurs in the metabolism of all aerobic organisms.

Chlamydia trachomatis, commonly known as chlamydia, is a bacterium that causes chlamydia, which can manifest in various ways, including: trachoma, lymphogranuloma venereum, nongonococcal urethritis, cervicitis, salpingitis, pelvic inflammatory disease. C. trachomatis is the most common infectious cause of blindness and the most common sexually transmitted bacterium.

The Actinomycetales are an order of Actinobacteria. A member of the order is often called an actinomycete.
The bacterium, despite its simplicity, contains a well-developed cell structure which is responsible for some of its unique biological structures and pathogenicity. Many structural features are unique to bacteria and are not found among archaea or eukaryotes. Because of the simplicity of bacteria relative to larger organisms and the ease with which they can be manipulated experimentally, the cell structure of bacteria has been well studied, revealing many biochemical principles that have been subsequently applied to other organisms.

Bacillus anthracis is the agent of anthrax—a common disease of livestock and, occasionally, of humans—and the only obligate pathogen within the genus Bacillus. This disease can be classified as a zoonosis, causing infected animals to transmit the disease to humans. B. anthracis is a Gram-positive, endospore-forming, rod-shaped bacterium, with a width of 1.0–1.2 µm and a length of 3–5 µm. It can be grown in an ordinary nutrient medium under aerobic or anaerobic conditions.

Acetic acid, systematically named ethanoic acid, is a colourless liquid organic compound with the chemical formula CH3COOH (also written as CH3CO2H, C2H4O2, or HC2H3O2). When undiluted, it is sometimes called glacial acetic acid. Vinegar is no less than 4% acetic acid by volume, making acetic acid the main component of vinegar apart from water. Acetic acid has a distinctive sour taste and pungent smell. In addition to household vinegar, it is mainly produced as a precursor to polyvinyl acetate and cellulose acetate. It is classified as a weak acid since it only partially dissociates in solution, but concentrated acetic acid is corrosive and can attack the skin.

Hydrochloric acid or muriatic acid is a colorless inorganic chemical system with the formula HCl. Hydrochloric acid has a distinctive pungent smell. It is classified as strongly acidic and can attack the skin over a wide composition range, since the hydrogen chloride completely dissociates in an aqueous solution.
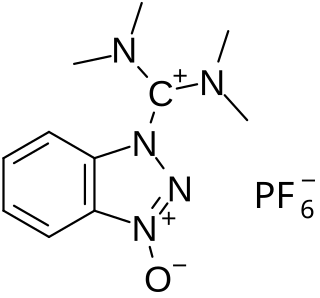
HBTU is a coupling reagent used in solid phase peptide synthesis. It was introduced in 1978 and shows resistance against racemization. It is used because of its mild activating properties.


















