Reactions
TMSNCS is useful reagent in organic chemistry. It is an ambident nucleophile, able to react with various alkyl halides, acetals, aldehydes, unsaturated compounds, aziridines, oxiranes, polycyclic aromatic hydrocarbons, and acetylated hexoses to form either thiocyanate or isothiocyanate structures. [6] As an electrophile, it can react with other nucleophiles to form thioamide types of structures, some of which can undergo subsequent reactions to form heterocycles.
Reactions with aldehydes and acetals
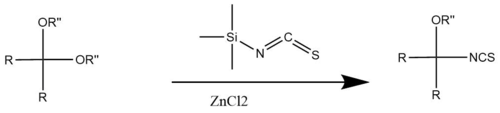
As a nitrogen nucleophile, TMSNCS adds across the carbonyl group of aldehydes and substitutes isothiocyanate for one of the ether groups on acetals via acid-catalyzed processes. [8]
-

-
Mercapto-1,2,4-triazoles
A one-step method to prepare mercapto-1,2,4-tiazoles is favored on the industrial scale due to its high efficiency (88% yield) and non-constraining conditions: not requiring anhydrous solvent, inert gas temperature, or chromatographic purification. The method can be summarized as follows: after the equimolar mixture of benzohydrazide and TMSNCS reflux in the presence of EtOH for 5 hours, NaOH is added to the reaction mixture and the solution is refluxed for 4 h. Acetic acid is then used to cool and neutralize, ultimately yielding the pure white solid 3-phenyl-5-mercapto-4H-1,2,4-triazole at 88% yield. [6]
-

2-Amino-1,3,4-oxadiazoles
TMSNCS used for the synthesis of 2-amino-1,3,4-oxadiazoles. The TMSNCS reagent assists in the production of the thiosemicarbazide and the subsequent reaction (cyclodesulfurization of thiosemicarbazides under basic conditions in the presence of I2/KI) results in 2-amino-1,3,4-oxadiazoles in high yields (79–94%). [9] The 2-amino-1,3,4-oxadiazoles resulting from this reaction are: 2-Amino-5-phenyl-1,3,4-oxadiazole, 2-Amino-5-(p-methylphenyl)-1,3,4-oxadiazole, 2-Amino-5-(p-chlorophenyl)-1,3,4-oxadiazole, 2-Amino-5-(p-methoxyphenyl)- 1,3,4-oxadiazole, 2-Amino-5-(p-nitrophenyl) -1,3,4-oxadiazole, 2-Amino-5-(o-methylphenyl)-1,3,4-oxadiazole, 2-Amino-5-(o-chlorophenyl)-1,3,4-oxadiazole, ect. [10]
-

This page is based on this
Wikipedia article Text is available under the
CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.





 [4]
[4] 



