Lipid-lowering agents, also sometimes referred to as hypolipidemic agents, cholesterol-lowering drugs, or antihyperlipidemic agents are a diverse group of pharmaceuticals that are used to lower the level of lipids and lipoproteins such as cholesterol, in the blood (hyperlipidemia). The American Heart Association recommends the descriptor 'lipid lowering agent' be used for this class of drugs rather than the term 'hypolipidemic'.

Rosiglitazone is an antidiabetic drug in the thiazolidinedione class. It works as an insulin sensitizer, by binding to the PPAR in fat cells and making the cells more responsive to insulin. It is marketed by the pharmaceutical company GlaxoSmithKline (GSK) as a stand-alone drug or for use in combination with metformin or with glimepiride. First released in 1999, annual sales peaked at approximately $2.5-billion in 2006; however, following a meta-analysis in 2007 that linked the drug's use to an increased risk of heart attack, sales plummeted to just $9.5-million in 2012. The drug's patent expired in 2012.

Flurazepam is a drug which is a benzodiazepine derivative. It possesses anxiolytic, anticonvulsant, hypnotic, sedative and skeletal muscle relaxant properties. It produces a metabolite with a long half-life, which may stay in the bloodstream for days. Flurazepam was patented in 1968 and came into medical use the same year. Flurazepam, developed by Roche Pharmaceuticals was one of the first benzo hypnotics to be marketed.
Pharmacovigilance, also known as drug safety, is the pharmaceutical science relating to the "collection, detection, assessment, monitoring, and prevention" of adverse effects with pharmaceutical products. The etymological roots for the word "pharmacovigilance" are: pharmakon and vigilare. As such, pharmacovigilance heavily focuses on adverse drug reactions (ADR), which are defined as any response to a drug which is noxious and unintended, including lack of efficacy. Medication errors such as overdose, and misuse and abuse of a drug as well as drug exposure during pregnancy and breastfeeding, are also of interest, even without an adverse event, because they may result in an adverse drug reaction.

3-Aminopyridine-2-carboxaldehyde thiosemicarbazone is a substance that is being studied in the treatment of cancer.

Eszopiclone, sold under the brand name Lunesta among others, is a medication used in the treatment of insomnia. Evidence supports slight to moderate benefit up to six months. It is taken by mouth.
In the United States, a boxed warning is a type of warning that appears near the beginning of the package insert for certain prescription drugs, so called because the U.S. Food and Drug Administration specifies that it is formatted with a 'box' or border around the text to emphasize it is of utmost importance. The FDA can require a pharmaceutical company to place a boxed warning. It is the strongest warning that the FDA requires, and signifies that medical studies indicate that the drug carries a significant risk of preventable, serious or even life-threatening adverse effects.
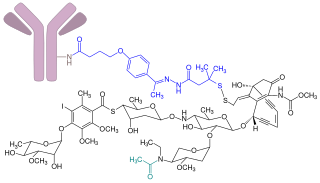
Gemtuzumab ozogamicin, sold under the brand name Mylotarg, is an antibody-drug conjugate that is used to treat acute myeloid leukemia.
Lintuzumab (SGN-33) is a humanized monoclonal antibody used in the treatment of cancer. The drug had been developed by Seattle Genetics as a treatment for acute myeloid leukemia (AML), a condition which results in the deaths of 9,000 people a year in the United States. Lintuzumab targets the CD33 protein, which is expressed in AML and other myeloproliferative diseases, but does not appear in abundance on normal cells.

Lestaurtinib is a tyrosine kinase inhibitor structurally related to staurosporine. This semisynthetic derivative of the indolocarbazole K252a was investigated by Cephalon as a treatment for various types of cancer. It is an inhibitor of the kinases fms-like tyrosine kinase 3 (FLT3), Janus kinase 2 (JAK2), tropomyosin receptor kinase (trk) A (TrkA), TrkB and TrkC.

Sapacitabine is a chemotherapeutic drug developed by US biotechnology firm Cyclacel currently undergoing clinical trials against leukemia.

Histamine dihydrochloride is a salt of histamine that is used as a drug for the prevention of relapse in patients diagnosed with acute myeloid leukemia (AML).

Midostaurin, sold under the brand name Rydapt & Tauritmo both by Novartis, is a multi-targeted protein kinase inhibitor that has been investigated for the treatment of acute myeloid leukemia (AML), myelodysplastic syndrome (MDS) and advanced systemic mastocytosis. It is a semi-synthetic derivative of staurosporine, an alkaloid from the bacterium Streptomyces staurosporeus.
Brilacidin, an investigational new drug, is a polymer-based antibiotic currently in human clinical trials, and represents a new class of antibiotics called host defense protein mimetics, or HDP-mimetics, which are non-peptide synthetic small molecules modeled after host defense peptides (HDPs). HDPs, also called antimicrobial peptides, some of which are defensins, are part of the innate immune response and are common to most higher forms of life. As brilacidin is modeled after a defensin, it is also called a defensin mimetic.

Vadastuximab talirine is an antibody-drug conjugate (ADC) directed to CD33 (siglec-3) which is a transmembrane receptor expressed on cells of myeloid lineage. The experimental drug, being developed by Seattle Genetics, was in clinical trials for the treatment of acute myeloid leukemia (AML).
Seagen Inc. is an American biotechnology company focused on developing and commercializing innovative, empowered monoclonal antibody-based therapies for the treatment of cancer. The company, headquartered in Bothell, Washington, is the industry leader in antibody-drug conjugates or ADCs, a technology designed to harness the targeting ability of monoclonal antibodies to deliver cell-killing agents directly to cancer cells. Antibody-drug conjugates are intended to spare non-targeted cells and thus reduce many of the toxic effects of traditional chemotherapy, while potentially enhancing antitumor activity.
FLAG is a chemotherapy regimen used for relapsed and refractory acute myeloid leukemia (AML). The acronym incorporates the three primary ingredients of the regimen:
- Fludarabine: an antimetabolite that, while not active toward AML, increases formation of an active cytarabine metabolite, ara-CTP, in AML cells;
- Arabinofuranosyl cytidine : an antimetabolite that has been proven to be the most active toward AML among various cytotoxic drugs in single-drug trials; and
- Granulocyte colony-stimulating factor (G-CSF): a glycoprotein that shortens the duration and severity of neutropenia.

Venetoclax, sold under the brand names Venclexta and Venclyxto, is a medication used to treat adults with chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), or acute myeloid leukemia (AML).

Decitabine/cedazuridine, sold under the brand name Inqovi among others, is a fixed-dose combination anticancer medication used for the treatment of adults with myelodysplastic syndromes and chronic myelomonocytic leukemia (CMML). It is a combination of decitabine, a nucleoside metabolic inhibitor, and cedazuridine, a cytidine deaminase inhibitor.
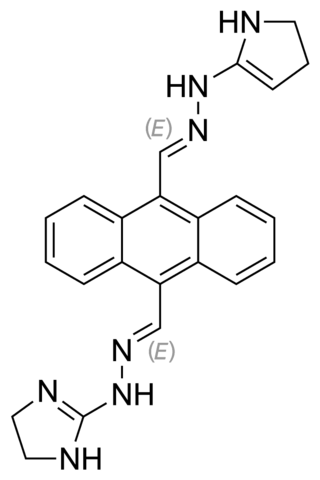
Bisantrene, trademarked as Zantrène, is an anthracenyl bishydrazone with anthracycline-like antineoplastic activity and an antimetabolite. Bisantrene intercalates with and disrupts the configuration of DNA, resulting in DNA single-strand breaks, DNA-protein crosslinking, and inhibition of DNA replication. This agent is similar to doxorubicin in activity, but unlike anthracyclines like doxorubicin, exhibits little cardiotoxicity.













