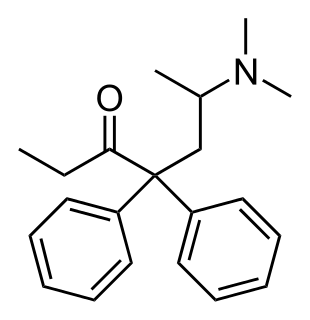
Methadone, sold under the brand names Dolophine and Methadose among others, is a synthetic opioid agonist used for opioid maintenance therapy in opioid dependence and for chronic pain management. It is most commonly used in the treatment of addiction to heroin. Detoxification using methadone can be accomplished in less than a month, or it may be done gradually over as long as six months. While a single dose has a rapid effect, maximum effect can take up to five days of use. The pain-relieving effects last about six hours after a single dose. After long-term use, in people with normal liver function, effects last 8 to 36 hours. Methadone is usually taken by mouth and rarely by injection into a muscle or vein.

Dizocilpine (INN), also known as MK-801, is a uncompetitive antagonist of the N-Methyl-D-aspartate (NMDA) receptor, a glutamate receptor, discovered by a team at Merck in 1982. Glutamate is the brain's primary excitatory neurotransmitter. The channel is normally blocked with a magnesium ion and requires depolarization of the neuron to remove the magnesium and allow the glutamate to open the channel, causing an influx of calcium, which then leads to subsequent depolarization. Dizocilpine binds inside the ion channel of the receptor at several of PCP's binding sites thus preventing the flow of ions, including calcium (Ca2+), through the channel. Dizocilpine blocks NMDA receptors in a use- and voltage-dependent manner, since the channel must open for the drug to bind inside it. The drug acts as a potent anti-convulsant and probably has dissociative anesthetic properties, but it is not used clinically for this purpose because of the discovery of brain lesions, called Olney's lesions (see below), in laboratory rats. Dizocilpine is also associated with a number of negative side effects, including cognitive disruption and psychotic-spectrum reactions. It inhibits the induction of long term potentiation and has been found to impair the acquisition of difficult, but not easy, learning tasks in rats and primates. Because of these effects of dizocilpine, the NMDA receptor pore-blocker ketamine is used instead as a dissociative anesthetic in human medical procedures. While ketamine may also trigger temporary psychosis in certain individuals, its short half-life and lower potency make it a much safer clinical option. However, dizocilpine is the most frequently used uncompetitive NMDA receptor antagonist in animal models to mimic psychosis for experimental purposes.
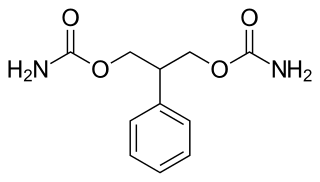
Felbamate is an anticonvulsant used in the treatment of epilepsy. It is used to treat partial seizures in adults and partial and generalized seizures associated with Lennox–Gastaut syndrome in children. However, an increased risk of potentially fatal aplastic anemia and/or liver failure limit the drug's usage to severe refractory epilepsy.

NMDA receptor antagonists are a class of drugs that work to antagonize, or inhibit the action of, the N-Methyl-D-aspartate receptor (NMDAR). They are commonly used as anesthetics for animals and humans; the state of anesthesia they induce is referred to as dissociative anesthesia.

Tenocyclidine (TCP) was discovered by a team at Parke-Davis in the late 1950s. It is a dissociative anesthetic drug with psychostimulant and hallucinogenic effects. It is similar in effects to phencyclidine (PCP) but is considerably more potent. TCP has slightly different binding properties to PCP, with more affinity for the NMDA receptors, but less affinity for the sigma receptors. Because of its high affinity for the PCP site of the NMDA receptor complex, the 3H radiolabelled form of TCP is widely used in research into NMDA receptors.

Glutamate [NMDA] receptor subunit epsilon-2, also known as N-methyl D-aspartate receptor subtype 2B, is a protein that in humans is encoded by the GRIN2B gene.

Glutamate [NMDA] receptor subunit zeta-1 is a protein that in humans is encoded by the GRIN1 gene.

Selfotel (CGS-19755) is a drug which acts as a competitive NMDA antagonist, directly competing with glutamate for binding to the receptor. Initial studies showed it to have anticonvulsant, anxiolytic, analgesic and neuroprotective effects, and it was originally researched for the treatment of stroke, but subsequent animal and human studies showed phencyclidine-like effects, as well as limited efficacy and evidence for possible neurotoxicity under some conditions, and so clinical development was ultimately discontinued.

Neramexane is a drug related to memantine, which acts as an NMDA antagonist and has neuroprotective effects. It is being developed for various possible applications, including treatment of tinnitus, Alzheimer's disease, drug addiction and as an analgesic. Animal studies have also suggested antidepressant and nootropic actions, so there are a wide range of potential applications this drug may be used for. It also acts as a nicotinic acetylcholine receptor antagonist.

Perzinfotel (EAA-090) is a drug which acts as a potent NMDA antagonist. It has neuroprotective effects and has been investigated for the treatment of stroke, but lacks analgesic effects. Nevertheless, it shows a good safety profile compared to older drugs, although further development of this drug has been discontinued.

2-Methyl-6-(phenylethynyl)pyridine (MPEP) is a research drug which was one of the first compounds found to act as a selective antagonist for the metabotropic glutamate receptor subtype mGluR5. After being originally patented as a liquid crystal for LCDs, it was developed by the pharmaceutical company Novartis in the late 1990s. It was found to produce neuroprotective effects following acute brain injury in animal studies, although it was unclear whether these results were purely from mGluR5 blockade as it also acts as a weak NMDA antagonist, and as a positive allosteric modulator of another subtype mGlu4, and there is also evidence for a functional interaction between mGluR5 and NMDA receptors in the same populations of neurons. It was also shown to produce antidepressant and anxiolytic effects in animals, and to reduce the effects of morphine withdrawal, most likely due to direct interaction between mGluR5 and the μ-opioid receptor.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.

Indantadol is a drug which was formerly being investigated as an anticonvulsant and neuroprotective and is now under development for the treatment of neuropathic pain and chronic cough in Europe by Vernalis and Chiesi. It acts as a competitive, reversible, and non-selective monoamine oxidase inhibitor, and as a low affinity, non-competitive NMDA receptor antagonist. A pilot study of indantadol for chronic cough was initiated in October 2009 and in April 2010 it failed to achieve significant efficacy in neuropathic pain in phase IIb clinical trials.

Traxoprodil is a drug developed by Pfizer which acts as an NMDA antagonist, selective for the NR2B subunit. It has neuroprotective, analgesic, and anti-Parkinsonian effects in animal studies. Traxoprodil has been researched in humans as a potential treatment to lessen the damage to the brain after stroke, but results from clinical trials showed only modest benefit. The drug was found to cause EKG abnormalities and its clinical development was stopped. More recent animal studies have suggested traxoprodil may exhibit rapid-acting antidepressant effects similar to those of ketamine, although there is some evidence for similar psychoactive side effects and abuse potential at higher doses, which might limit clinical acceptance of traxoprodil for this application.

Levomethadone, sold under the brand name L-Polamidon among others, is a synthetic opioid analgesic and antitussive which is marketed in Europe and is used for pain management and in opioid maintenance therapy. In addition to being used as a pharmaceutical drug itself, levomethadone is the main therapeutic component of methadone.

HA-966 or (±) 3-Amino-1-hydroxy-pyrrolidin-2-one is a molecule used in scientific research as a glycine receptor and NMDA receptor antagonist / low efficacy partial agonist. It has neuroprotective and anticonvulsant, anxiolytic, antinociceptive and sedative / hypnotic effects in animal models. Pilot human clinical trials in the early 1960s showed that HA-966 appeared to benefit patients with tremors of extrapyramidal origin.

Rapastinel (INN) (former developmental code names GLYX-13, BV-102) is a novel antidepressant that is under development by Allergan (previously Naurex) as an adjunctive therapy for the treatment of treatment-resistant major depressive disorder. It is a centrally active, intravenously administered (non-orally active) amidated tetrapeptide (Thr-Pro-Pro-Thr-NH2) that acts as a selective, weak partial agonist (mixed antagonist/agonist) of an allosteric site of the glycine site of the NMDA receptor complex (Emax ≈ 25%). The drug is a rapid-acting and long-lasting antidepressant as well as robust cognitive enhancer by virtue of its ability to both inhibit and enhance NMDA receptor-mediated signal transduction.
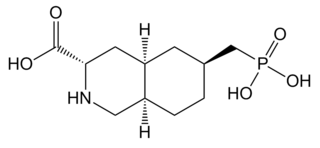
LY-235959 is a competitive antagonist at the NMDA receptor. It has analgesic and neuroprotective effects and causes hypothermia in animal models, as well as reducing the development of tolerance to morphine and altering the reinforcing effects of cocaine.

Pregnenolone succinate is a synthetic pregnane steroid and an ester of pregnenolone which is described as a glucocorticoid and anti-inflammatory drug and has been patented and marketed as a topical medication in the form of a cream for the treatment of allergic, pruritic, and inflammatory dermatitis. It has also been described as a non-hormonal sterol, having neurosteroid activity, and forming a progesterone analogue via dehydrogenation.

PD-137889 (N-methylhexahydrofluorenamine) is a chemical compound that is active as an NMDA receptor antagonist in the central nervous system at roughly 30 times the potency of the "flagship" of its class, ketamine, and substitutes for phencyclidine in animal studies. Ki [3H]TCP binding = 27 nM versus ketamine's Ki = 860 nM.




















