An international nonproprietary name (INN) is an official generic and nonproprietary name given to a pharmaceutical drug or an active ingredient. INNs are intended to make communication more precise by providing a unique standard name for each active ingredient, to avoid prescribing errors. The INN system has been coordinated by the World Health Organization (WHO) since 1953.
The nomenclature of monoclonal antibodies is a naming scheme for assigning generic, or nonproprietary, names to monoclonal antibodies. An antibody is a protein that is produced in B cells and used by the immune system of humans and other vertebrate animals to identify a specific foreign object like a bacterium or a virus. Monoclonal antibodies are those that were produced in identical cells, often artificially, and so share the same target object. They have a wide range of applications including medical uses.
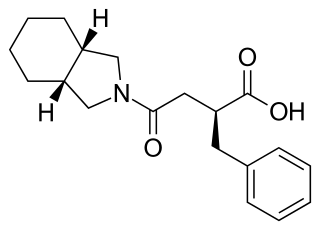
Mitiglinide is a drug for the treatment of type 2 diabetes.

Bradanicline is a drug which was being developed by Targacept that acts as a partial agonist at the α7 subtype of the neural nicotinic acetylcholine receptors. It showed cognitive enhancing effects in animal studies, and was being developed through a collaboration between Targacept and AstraZeneca as a potential treatment for schizophrenia and attention deficit disorder. Phase I clinical trials were completed successfully, and it was in phase II trials.
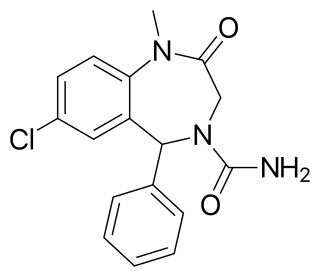
Carburazepam is a drug which is a benzodiazepine derivative.
Drug nomenclature is the systematic naming of drugs, especially pharmaceutical drugs. In the majority of circumstances, drugs have 3 types of names: chemical names, the most important of which is the IUPAC name; generic or nonproprietary names, the most important of which are international nonproprietary names (INNs); and trade names, which are brand names. Under the INN system, generic names for drugs are constructed out of affixes and stems that classify the drugs into useful categories while keeping related names distinguishable. A marketed drug might also have a company code or compound code.
Imgatuzumab (INN) is a humanized monoclonal antibody designed for the treatment of cancer. It is an anti-EGFR antibody that acts as an immunomodulator.
Parsatuzumab (INN) is a humanized monoclonal antibody designed for the treatment of cancer. It acts as an immunomodulator and binds to EGFL7.
Perakizumab (INN) is a humanized monoclonal antibody designed for the treatment of arthritis. It binds to IL17A and acts as an immunomodulator.

Pacritinib, sold under the brand name Vonjo, is an anti-cancer medication used to treat myelofibrosis. It is a macrocyclic protein kinase inhibitor. It mainly inhibits Janus kinase 2 (JAK2) and Fms-like tyrosine kinase 3\CD135 (FLT3).
Emibetuzumab (INN) (LY2875358) is a humanized monoclonal antibody designed for the treatment of cancer. It is in phase II trials for patients with NSCLC
Lulizumab pegol is a monoclonal antibody designed for the treatment of autoimmune diseases.
Sofituzumab vedotin is a monoclonal antibody designed for the treatment of ovarian cancer.
Ulocuplumab is a monoclonal antibody designed for the treatment of hematologic malignancies.
Seribantumab is a monoclonal antibody designed for the treatment of cancer.
Brontictuzumab is a humanized monoclonal antibody designed for the treatment of cancer.
Lumretuzumab is a humanized monoclonal antibody designed for the treatment of cancer.
Plozalizumab is a humanized monoclonal antibody designed for the treatment of diabetic nephropathy and arteriovenous graft patency.
Oleclumab is a human monoclonal antibody targeting the ectonucleotidase CD73 that was designed for the treatment of pancreatic and colorectal and other cancers.




