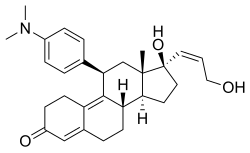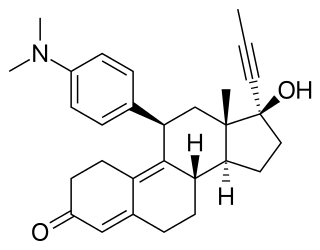
Mifepristone, also known as RU-486, is a medication typically used in combination with misoprostol to bring about a medical abortion during pregnancy and manage early miscarriage. This combination is 97% effective during the first 63 days of pregnancy. It is also effective in the second trimester of pregnancy. Effectiveness should be verified two weeks after use. It is taken by mouth.

A selective progesterone receptor modulator (SPRM) is an agent that acts on the progesterone receptor (PR), the biological target of progestogens like progesterone. A characteristic that distinguishes such substances from full receptor agonists and full antagonists is that their action differs in different tissues, i.e. agonist in some tissues while antagonist in others. This mixed profile of action leads to stimulation or inhibition in tissue-specific manner, which further raises the possibility of dissociating undesirable adverse effects from the development of synthetic PR-modulator drug candidates.

Norgestrienone, sold under the brand names Ogyline, Planor, and Miniplanor, is a progestin medication which has been used in birth control pills, sometimes in combination with ethinylestradiol. It was developed by Roussel Uclaf and has been registered for use only in France. Under the brand name Planor, it has been marketed in France as 2 mg norgestrienone and 50 μg ethinylestradiol tablets. It is taken by mouth.

Antiprogestogens, or antiprogestins, also known as progesterone antagonists or progesterone blockers, are a class of drugs which prevent progestogens like progesterone from mediating their biological effects in the body. They act by blocking the progesterone receptor (PR) and/or inhibiting or suppressing progestogen production. Antiprogestogens are one of three types of sex hormone antagonists, the others being antiestrogens and antiandrogens.

Chlormadinone acetate (CMA), sold under the brand names Belara, Gynorelle, Lutéran, and Prostal among others, is a progestin and antiandrogen medication which is used in birth control pills to prevent pregnancy, as a component of menopausal hormone therapy, in the treatment of gynecological disorders, and in the treatment of androgen-dependent conditions like enlarged prostate and prostate cancer in men and acne and hirsutism in women. It is available both at a low dose in combination with an estrogen in birth control pills and, in a few countries like France and Japan, at low, moderate, and high doses alone for various indications. It is taken by mouth.

Ulipristal acetate, sold under the brand name Ella among others, is a medication used for emergency contraception and uterine fibroids. As emergency contraception it should be used within 120 hours of vaginally penetrating intercourse. For fibroids it may be taken for up to six months. It is taken by mouth.

Demegestone, sold under the brand name Lutionex, is a progestin medication which was previously used to treat luteal insufficiency but is now no longer marketed. It is taken by mouth.

Aglepristone (INN) is a synthetic, steroidal antiprogestogen related to mifepristone which is marketed by Virbac in several European countries for use in veterinary medicine. It is specifically used as an abortifacient in pregnant animals. Aglepristone, similarly to mifepristone, also possesses some antiglucocorticoid activity.
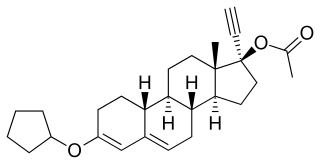
Quingestanol acetate, sold under the brand names Demovis and Pilomin among others, is a progestin medication which was used in birth control pills but is no longer marketed. It is taken by mouth.

Segesterone acetate (SGA), sold under the brand names Nestorone, Elcometrine, and Annovera, is a progestin medication which is used in birth control and in the treatment of endometriosis in the United States, Brazil, and other South American countries. It is available both alone and in combination with an estrogen. It is not effective by mouth and must be given by other routes, most typically as a vaginal ring or implant that is placed into fat.
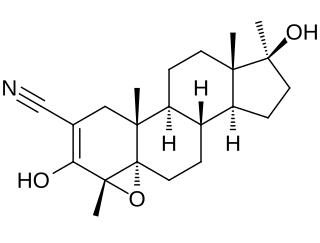
Epostane is an inhibitor of 3β-hydroxysteroid dehydrogenase (3β-HSD) that was developed as a contraceptive, abortifacient, and oxytocic drug but was never marketed. By inhibiting 3β-HSD, epostane blocks the biosynthesis of progesterone from pregnenolone, thereby functioning as an antiprogestogen and terminating pregnancy. The drug was trialed and in a study was found to be slightly more effective at inducing abortion relative to mifepristone.

Anordrin, also known as 2α,17α-diethynyl-A-nor-5α-androstane-2β,17β-diol dipropionate, is a synthetic, steroidal selective estrogen receptor modulator (SERM) which is used in China as an emergency contraceptive. It is the most commonly used emergency contraceptive in China. The drug is marketed in a combination formulation with mifepristone under the brand name Zi Yun. Anordrin has not been studied for use or marketed outside of China. It has been used in China since the 1970s.

Spirorenone (INN) is a steroidal antimineralocorticoid of the spirolactone group that was never marketed. Spirorenone possesses 5–8 times the antimineralocorticoid activity of spironolactone in animal studies. The initial discovery of spirorenone was deemed a great success, as no compound with greater antimineralocorticoid activity had been developed since spironolactone in 1957. Moreover, spirorenone itself has virtually no affinity for the androgen receptor while its progestogenic activity shows species differences, being somewhat greater than that of spironolactone in rabbits but absent in mice and rats. As such, it was characterized as a highly potent antimineralocorticoid with far fewer hormonal side effects relative to spironolactone.

A steroidal antiandrogen (SAA) is an antiandrogen with a steroidal chemical structure. They are typically antagonists of the androgen receptor (AR) and act both by blocking the effects of androgens like testosterone and dihydrotestosterone (DHT) and by suppressing gonadal androgen production. SAAs lower concentrations of testosterone through simulation of the negative feedback inhibition of the hypothalamus. SAAs are used in the treatment of androgen-dependent conditions in men and women, and are also used in veterinary medicine for the same purpose. They are the converse of nonsteroidal antiandrogens (NSAAs), which are antiandrogens that are not steroids and are structurally unrelated to testosterone.
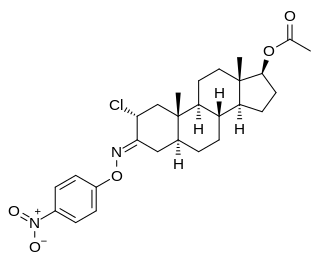
Nisterime acetate (USAN), also known as 2α-chloro-4,5α-dihydrotestosterone O-(p-nitrophenyl)oxime 17β-acetate or as 2α-chloro-5α-androstan-17β-ol-3-one O-(p-nitrophenyl)oxime 17β-acetate, is a synthetic, orally active anabolic-androgenic steroid (AAS) and a derivative of dihydrotestosterone (DHT) that was developed as a postcoital contraceptive but was never marketed. It is an androgen ester – specifically, the C17α acetate ester of nisterime. Unlike antiprogestogens like mifepristone, nisterime acetate does not prevent implantation and instead induces embryo resorption as well as interrupts the post-implantation stage of pregnancy.
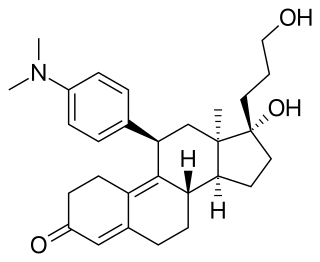
Onapristone (INN) is a synthetic and steroidal antiprogestogen with additional antiglucocorticoid activity which was developed by Schering and described in 1984 but was never marketed. It is a silent antagonist of the progesterone receptor (PR), in contrast to the related antiprogestogen mifepristone. Moreover, compared to mifepristone, onapristone has reduced antiglucocorticoid activity, shows little antiandrogenic activity, and has 10- to 30-fold greater potency as an antiprogestogen. The medication was under development for clinical use, for instance in the treatment of breast cancer and as an endometrial contraceptive, but was discontinued during phase III clinical trials in 1995 due to findings that liver function abnormalities developed in a majority patients.

Toripristone (INN) is a synthetic, steroidal antiglucocorticoid as well as antiprogestogen which was never marketed. It is reported as a potent and highly selective antagonist of the glucocorticoid receptor (GR), though it also acts as an antagonist of the progesterone receptor (PR). The pharmacological profile of toripristone is said to be very similar to that of mifepristone, except that toripristone does not bind to orosomucoid. The drug has been used to study the hypothalamic-pituitary-adrenal axis and has been used as a radiotracer for the GR. Its INN was given in 1990.

Asoprisnil ecamate (INN) is a synthetic, steroidal selective progesterone receptor modulator (SPRM) which was under development for the treatment of endometriosis, uterine fibroids, and menopausal symptoms but was discontinued. It is a potent and highly selective ligand of the progesterone receptor with mixed agonistic and antagonistic activity and much reduced antiglucocorticoid activity relative to mifepristone. The drug reached phase III clinical trials for the aforementioned indications prior to its discontinuation.
A sex-hormonal agent, also known as a sex-hormone receptor modulator, is a type of hormonal agent which specifically modulates the effects of sex hormones and of their biological targets, the sex hormone receptors. The sex hormones include androgens such as testosterone, estrogens such as estradiol, and progestogens such as progesterone. Sex-hormonal agents may be either steroidal or nonsteroidal in chemical structure and may serve to either enhance, inhibit, or have mixed effects on the function of the sex hormone systems.