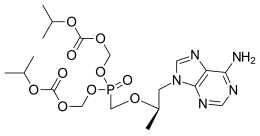
Reverse-transcriptase inhibitors (RTIs) are a class of antiretroviral drugs used to treat HIV infection or AIDS, and in some cases hepatitis B. RTIs inhibit activity of reverse transcriptase, a viral DNA polymerase that is required for replication of HIV and other retroviruses.

Lamivudine, commonly called 3TC, is an antiretroviral medication used to prevent and treat HIV/AIDS. It is also used to treat chronic hepatitis B when other options are not possible. It is effective against both HIV-1 and HIV-2. It is typically used in combination with other antiretrovirals such as zidovudine, dolutegravir, and abacavir. Lamivudine may be included as part of post-exposure prevention in those who have been potentially exposed to HIV. Lamivudine is taken by mouth as a liquid or tablet.

Emtricitabine, with trade name Emtriva, is a nucleoside reverse-transcriptase inhibitor (NRTI) for the prevention and treatment of HIV infection in adults and children. In 2019, it was the 494th most commonly prescribed medication in the United States, with more than 3 thousand prescriptions.

Emtricitabine/tenofovir, sold under the brand name Truvada among others, is a fixed-dose combination antiretroviral medication used to treat and prevent HIV/AIDS. It contains the antiretroviral medications emtricitabine and tenofovir disoproxil. For treatment, it must be used in combination with other antiretroviral medications. For prevention before exposure, in those who are at high risk, it is recommended along with safer sex practices. It does not cure HIV/AIDS. Emtricitabine/tenofovir is taken by mouth.

Pre-exposure prophylaxis (PrEP) is the use of medications to prevent the spread of disease in people who have not yet been exposed to a disease-causing agent, usually a virus. The term typically refers to the use of antiviral drugs as a strategy for the prevention of HIV/AIDS. PrEP is one of a number of HIV prevention strategies for people who are HIV negative but who have a higher risk of acquiring HIV, including sexually active adults at increased risk of contracting HIV, people who engage in intravenous drug use, and serodiscordant sexually active couples.

Efavirenz/emtricitabine/tenofovir, sold under the brand name Atripla among others, is a fixed-dose combination antiretroviral medication used to treat HIV/AIDS. It contains efavirenz, emtricitabine, and tenofovir disoproxil. It can be used by itself or together with other antiretroviral medications. It is taken by mouth.
Integrase inhibitors (INIs) are a class of antiretroviral drug designed to block the action of integrase, a viral enzyme that inserts the viral genome into the DNA of the host cell. Since integration is a vital step in retroviral replication, blocking it can halt further spread of the virus. Integrase inhibitors were initially developed for the treatment of HIV infection, but have been applied to other retroviruses. The class of integrase inhibitors called integrase strand transfer inhibitors (INSTIs) are in established medical use. Other classes, such as integrase binding inhibitors (INBIs), are still experimental.

Elvitegravir (EVG) is an integrase inhibitor used to treat HIV infection. It was developed by the pharmaceutical company Gilead Sciences, which licensed EVG from Japan Tobacco in March 2008. The drug gained approval by the U.S. Food and Drug Administration on August 27, 2012, for use in adult patients starting HIV treatment for the first time as part of the fixed dose combination known as Stribild. On September 24, 2014, the FDA approved Elvitegravir as a single pill formulation under the trade name Vitekta. On November 5, 2015, the FDA approved the drug for use in patients affected with HIV-1 as a part of a second fixed dose combination pill known as Genvoya.

Rilpivirine, sold under the brand names Edurant and Rekambys, is a medication, developed by Tibotec, used for the treatment of HIV/AIDS. It is a second-generation non-nucleoside reverse transcriptase inhibitor (NNRTI) with higher potency, longer half-life and reduced side-effect profile compared with older NNRTIs such as efavirenz.

Cobicistat, sold under the brand name Tybost, is a medication for use in the treatment of human immunodeficiency virus infection (HIV/AIDS). Its major mechanism of action is through the inhibition of human CYP3A proteins.
Discovery and development of nucleoside and nucleotide reverse-transcriptase inhibitors began in the 1980s when the AIDS epidemic hit Western societies. NRTIs inhibit the reverse transcriptase (RT), an enzyme that controls the replication of the genetic material of the human immunodeficiency virus (HIV). The first NRTI was zidovudine, approved by the U.S. Food and Drug Administration (FDA) in 1987, which was the first step towards treatment of HIV. Six NRTI agents and one NtRTI have followed. The NRTIs and the NtRTI are analogues of endogenous 2´-deoxy-nucleoside and nucleotide. Drug-resistant viruses are an inevitable consequence of prolonged exposure of HIV-1 to anti-HIV drugs.

iPrEx was a phase III clinical trial to determine whether the antiretroviral medication emtricitabine/tenofovir could safely and effectively prevent HIV acquisition through sex in men who have sex with men and transgender women. iPrEx was the first human study of an HIV prevention strategy known as pre-exposure prophylaxis, or PrEP.
Emtricitabine/rilpivirine/tenofovir is a fixed-dose combination of antiretroviral drugs for the treatment of HIV/AIDS. The drug was co-developed by Gilead Sciences and Johnson & Johnson's Tibotec division and was approved by the Food and Drug Administration in August 2011, and by the European Medicines Agency in November 2011, for patients who have not previously been treated for HIV. It is available as a once-a-day single tablet.
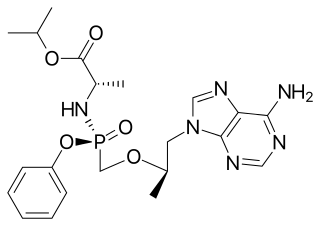
Tenofovir alafenamide, sold under the brand name Vemlidy, is an antiviral medication used against hepatitis B and HIV. It is used for the treatment of chronic hepatitis B virus (HBV) infection in adults with compensated liver disease and is given in combination with other medications for the prevention and treatment of HIV. It is taken by mouth.
Elvitegravir/cobicistat/emtricitabine/tenofovir, sold under the brand name Stribild, also known as the Quadpill, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. Elvitegravir, emtricitabine and tenofovir disoproxil directly suppress viral reproduction. Cobicistat increases the effectiveness of the combination by inhibiting the liver and gut wall enzymes that metabolize elvitegravir. It is taken by mouth.

Cabotegravir, sold under the brand name Vocabria among others, is a antiretroviral medication used for the treatment of HIV/AIDS. It is available in the form of tablets and as an intramuscular injection, as well as in an injectable combination with rilpivirine under the brand name Cabenuva.

Abacavir/dolutegravir/lamivudine, sold under the brand name Triumeq among others, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. It is a combination of three medications with different and complementary mechanisms of action: abacavir, dolutegravir and lamivudine.
Efavirenz/lamivudine/tenofovir (EFV/3TC/TDF), sold under the brand name Symfi among others, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. It combines efavirenz, lamivudine, and tenofovir disoproxil. As of 2019, it is listed by the World Health Organization as an alternative first line option to dolutegravir/lamivudine/tenofovir. It is taken by mouth.

Bictegravir/emtricitabine/tenofovir alafenamide, sold under the brand name Biktarvy, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. One tablet, taken orally once daily, contains 50 mg bictegravir, 200 mg emtricitabine, and 25 mg tenofovir alafenamide. It was approved for use in the United States in February 2018, and for use in the European Union in June 2018.

Lamivudine/tenofovir disoproxil, sold under the brand name Cimduo among others, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS in adults and children weighing more than 35 kilograms (77 lb). It contains lamivudine and tenofovir disoproxil. It is taken by mouth.