
The cumene process is an industrial process for synthesizing phenol and acetone from benzene and propylene. The term stems from cumene, the intermediate material during the process. It was invented by R. Ūdris and P. Sergeyev in 1942 (USSR), and independently by Heinrich Hock in 1944.

Cyclopentanone is the organic compound with the formula (CH2)4CO. This cyclic ketone is a colorless volatile liquid.

Vinylacetylene is the organic compound with the formula C4H4. The colourless gas was once used in the polymer industry. It is composed of both alkyne and alkene groups and is the simplest enyne.

1,4-Benzoquinone, commonly known as para-quinone, is a chemical compound with the formula C6H4O2. In a pure state, it forms bright-yellow crystals with a characteristic irritating odor, resembling that of chlorine, bleach, and hot plastic or formaldehyde. This six-membered ring compound is the oxidized derivative of 1,4-hydroquinone. The molecule is multifunctional: it exhibits properties of a ketone, being able to form oximes; an oxidant, forming the dihydroxy derivative; and an alkene, undergoing addition reactions, especially those typical for α,β-unsaturated ketones. 1,4-Benzoquinone is sensitive toward both strong mineral acids and alkali, which cause condensation and decomposition of the compound.
A diamine is an amine with exactly two amino groups. Diamines are used as monomers to prepare polyamides, polyimides, and polyureas. The term diamine refers mostly to primary diamines, as those are the most reactive.

Crotonaldehyde is a chemical compound with the formula CH3CH=CHCHO. The compound is usually sold as a mixture of the E- and Z-isomers, which differ with respect to the relative position of the methyl and formyl groups. The E-isomer is more common (data given in Table is for the E-isomer). This lachrymatory liquid is moderately soluble in water and miscible in organic solvents. As an unsaturated aldehyde, crotonaldehyde is a versatile intermediate in organic synthesis. It occurs in a variety of foodstuffs, e.g. soybean oils.

4-Aminophenol (or para-aminophenol or p-aminophenol) is an organic compound with the formula H2NC6H4OH. Typically available as a white powder, it is commonly used as a developer for black-and-white film, marketed under the name Rodinal.
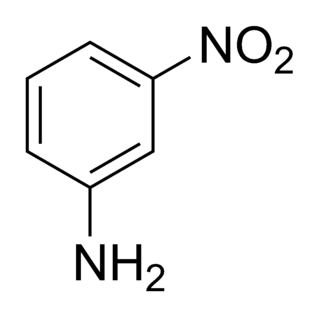
3-Nitroaniline is an organic compound with the formula H2NC6H4NO2. A yellow solid, it is a derivative of aniline, carrying a nitro functional group in position 3. It is an isomer of 2-nitroaniline and 4-nitroaniline. It is used as a precursor to dyes.
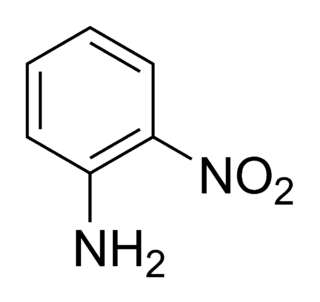
2-Nitroaniline is an organic compound with the formula H2NC6H4NO2. It is a derivative of aniline, carrying a nitro functional group in position 2. It is mainly used as a precursor to o-phenylenediamine.
Mononitrotoluene or nitrotoluene (MNT or NT), is any of three organic compounds with the formula C6H4(CH3)(NO2). They can be viewed as nitro derivatives of toluene or as methylated derivatives of nitrobenzene.
1,2-Dichloro-4-nitrobenzene is an organic compound with the formula 1,2-Cl2C6H3-4-NO2. This pale yellow solid is related to 1,2-dichlorobenzene by the replacement of one H atom with a nitro functional group. This compound is an intermediate in the synthesis of agrochemicals.
4-Nitrotoluene or para-nitrotoluene is an organic compound with the formula CH3C6H4NO2. It is a pale yellow solid. It is one of three isomers of nitrotoluene.

1,4-Naphthoquinone or para-naphthoquinone is a quinone derived from naphthalene. It forms volatile yellow triclinic crystals and has a sharp odor similar to benzoquinone. It is almost insoluble in cold water, slightly soluble in petroleum ether, and more soluble in polar organic solvents. In alkaline solutions it produces a reddish-brown color. Vitamin K is a derivative of 1,4-naphthoquinone. It is a planar molecule with one aromatic ring fused to a quinone subunit. It is an isomer of 1,2-naphthoquinone.
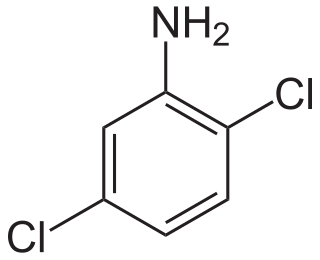
2,5-Dichloroaniline is an organic compound with the formula C6H3Cl2NH2. One of six isomers of dichloroaniline, it is a colorless solid that is insoluble in water. It is produced by hydrogenation of 1,4-dichloro-2-nitrobenzene. It is a precursor to dyes and pigments, e.g., Pigment Yellow 10.
2-Nitrobenzoic acid or o-nitrobenzoic acid is an organic compound with the formula C6H4(NO2)CO2H. It is prepared by oxidation of 2-nitrotoluene with nitric acid. In consists of a carboxylic acid group and a nitro group in the ortho configuration. Reduction of the nitro group into an amine produces anthranilic acid.
4-Chlorophenol is an organic compound with the formula C6H4ClOH. It is one of three monochlorophenol isomers. It is a colorless or white solid that melts easily and exhibits significant solubility in water. Its pKa is 9.14.

Diethylsuccinoylsuccinate is an organic compound with the formula [CH2C(OH)=C(CO2Et)]2 (Et = ethyl). A tetrasubstituted derivative of 1,4-cyclohexadiene, the compound is the enol tautomer of the corresponding cyclohexadione. It is produced by base-induced condensation of diethyl succinate:
In chemistry, the Halex process is used to convert aromatic chlorides to the corresponding aromatic fluorides. The process entails Halide exchange, hence the name. The reaction conditions call for hot (150-250 °C) solution of the aryl chloride and anhydrous potassium fluoride. Typical solvents are dimethylsulfoxide, dimethylformamide, and sulfolane. Potassium chloride is generated in the process. The reaction is mainly applied to nitro-substituted aryl chlorides. Sometimes more soluble fluorides, such as caesium fluoride and TBAF are used.

1,8-Naphthalic anhydride is an organic compound with the formula C10H6(C2O3). It is one of three isomers of naphthalic anhydride, the other two being the 1,2- and the 2,3-derivatives. The 1,8-isomer is prepared by aerobic oxidation of acenaphthene. 2,6-naphthalenedicarboxylic acid can be prepared from this anhydride. 1,8-Naphthalic anhydride is a precursor to the 4-chloro and 4,5-dichloro derivatives. These chloride groups are susceptible to displacement by amines and alkoxides, giving rise, ultimately, to a large family of naphthalimides, which are used as optical brighteners.
α-Halo carboxylic acids and esters are organic compounds with the respective formulas RCHXCO2H and RCHXCO2R' where R and R' are organic substituents. The X in these compounds is a halide, usually chloride and bromide. These compounds are often used as intermediates in the preparation of more elaborate derivatives. They are often potent alkylating agents. The mono halide derivatives are chiral.
















