Polybrominated diphenyl ethers or PBDEs, are a class of organobromine compounds that are used as flame retardants. Like other brominated flame retardants, PBDEs have been used in a wide array of products, including building materials, electronics, furnishings, motor vehicles, airplanes, plastics, polyurethane foams, and textiles. They are structurally akin to polychlorinated diphenyl ethers (PCDEs), polychlorinated biphenyls (PCBs) and other polyhalogenated compounds, consisting of two halogenated aromatic rings. PBDEs are classified according to the average number of bromine atoms in the molecule. The life-saving benefits of fire retardants led to their popularization. Standards for mass transit vehicles continues to increase as of 2021.

Flame retardants are a diverse group of chemicals that are added to manufactured materials, such as plastics and textiles, and surface finishes and coatings. Flame retardants are activated by the presence of an ignition source and prevent or slow the further development of flames by a variety of different physical and chemical mechanisms. They may be added as a copolymer during the polymerisation process, or later added to the polymer at a moulding or extrusion process or applied as a topical finish. Mineral flame retardants are typically additive, while organohalogen and organophosphorus compounds can be either reactive or additive.
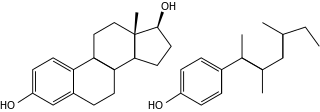
Endocrine disruptors, sometimes also referred to as hormonally active agents, endocrine disrupting chemicals, or endocrine disrupting compounds are chemicals that can interfere with endocrine systems. These disruptions can cause numerous adverse human health outcomes, including alterations in sperm quality and fertility; abnormalities in sex organs‚ endometriosis‚ early puberty‚ altered nervous system or immune function; certain cancers; respiratory problems; metabolic issues; diabetes, obesity, or cardiovascular problems; growth, neurological and learning disabilities, and more. Found in many household and industrial products, endocrine disruptors "interfere with the synthesis, secretion, transport, binding, action, or elimination of natural hormones in the body that are responsible for development, behavior, fertility, and maintenance of homeostasis ."
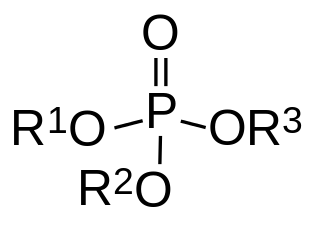
In organic chemistry, organophosphates are a class of organophosphorus compounds with the general structure O=P(OR)3, a central phosphate molecule with alkyl or aromatic substituents. They can be considered as esters of phosphoric acid. Organophosphates are best known for their use as pesticides.
Bisphenol A (BPA) is a chemical compound primarily used in the manufacturing of various plastics. It is a colourless solid which is soluble in most common organic solvents, but has very poor solubility in water. BPA is produced on an industrial scale by the condensation reaction of phenol and acetone. Global production in 2022 was estimated to be in the region of 10 million tonnes.
Tributyl phosphate, known commonly as TBP, is an organophosphorus compound with the chemical formula (CH3CH2CH2CH2O)3PO. This colourless, odorless liquid finds some applications as an extractant and a plasticizer. It is an ester of phosphoric acid with n-butanol.
Brominated flame retardants (BFRs) are organobromine compounds that have an inhibitory effect on combustion chemistry and tend to reduce the flammability of products containing them. The brominated variety of commercialized chemical flame retardants comprise approximately 19.7% of the market. They are effective in plastics and textile applications like electronics, clothes, and furniture.

A fire retardant is a substance that is used to slow down or stop the spread of fire or reduce its intensity. This is commonly accomplished by chemical reactions that reduce the flammability of fuels or delay their combustion. Fire retardants may also cool the fuel through physical action or endothermic chemical reactions. Fire retardants are available as powder, to be mixed with water, as fire-fighting foams and fire-retardant gels. Fire retardants are also available as coatings or sprays to be applied to an object.

Tricresyl phosphate (TCP), is a mixture of three isomeric organophosphate compounds most notably used as a flame retardant. Other uses include as a plasticizer in manufacturing for lacquers and varnishes and vinyl plastics and as an antiwear additive in lubricants. Pure tricresyl phosphate is a colourless, viscous liquid, although commercial samples are typically yellow. It is virtually insoluble in water, but easily soluble in organic solvents like toluene, hexane, and diethyl ether among others. It was synthesized by Alexander Williamson in 1854 upon reacting phosphorus pentachloride with cresol, though today's manufacturers can prepare TCP by mixing cresol with phosphorus oxychloride or phosphoric acid as well. TCP, especially the all-ortho isomer, is the causative agent in a number of acute poisonings. Its chronic toxicity is also of concern. The ortho-isomer is rarely used on its own outside of laboratory studies that require isomeric purity, due to its extremely toxic nature, and is generally excluded from commercial products where TCP is involved.

Decabromodiphenyl ether is a brominated flame retardant which belongs to the group of polybrominated diphenyl ethers (PBDEs). It was commercialised in the 1970s and was initially thought to be safe, but is now recognised as a hazardous and persistent pollutant. It was added to Annex A of the Stockholm Convention on Persistent Organic Pollutants in 2017, which means that treaty members must take measures to eliminate its production and use. The plastics industry started switching to decabromodiphenyl ethane as an alternative in the 1990s, but this is now also coming under regulatory pressure due to concerns over human health.
Pentabromodiphenyl ether is a brominated flame retardant which belongs to the group of polybrominated diphenyl ethers (PBDEs). Because of their toxicity and persistence, their industrial production is to be eliminated under the Stockholm Convention, a treaty to control and phase out major persistent organic pollutants (POP).
Octabromodiphenyl ether is a brominated flame retardant which belongs to the group of polybrominated diphenyl ethers (PBDEs).

Triphenyl phosphate (TPhP) is the chemical compound with the formula OP(OC6H5)3. It is the simplest aromatic organophosphate. This colourless solid is the ester (triester) of phosphoric acid and phenol. It is used as a plasticizer and a fire retardant in a wide variety of settings and products.
Aryldialkylphosphatase is a metalloenzyme that hydrolyzes the triester linkage found in organophosphate insecticides:

Tris(1,3-dichloroisopropyl)phosphate (TDCPP) is a chlorinated organophosphate. Organophosphate chemicals have a wide variety of applications and are used as flame retardants, pesticides, plasticizers, and nerve gases. TDCPP is structurally similar to several other organophosphate flame retardants, such as tris(2-chloroethyl) phosphate (TCEP) and tris(chloropropyl)phosphate (TCPP). TDCPP and these other chlorinated organophosphate flame retardants are all sometimes referred to as "chlorinated tris".

Susan D. Shaw was an American environmental health scientist, marine toxicologist, explorer, ocean conservationist, and author. A Doctor of Public Health, she was a professor in the Department of Environmental Health Sciences at the School of Public Health at the State University of New York at Albany, and Founder/President of the Shaw Institute, a nonprofit scientific institution with a mission to improve human and ecological health through innovative science and strategic partnerships. Shaw is globally recognized for pioneering high-impact environmental research on ocean pollution, climate change, oil spills, and plastics that has fueled public policy over three decades. In 1983, with landscape photographer Ansel Adams, she published Overexposure, the first book to document the health hazards of photographic chemicals. Shaw is credited as the first scientist to show that brominated flame retardant chemicals used in consumer products have contaminated marine mammals and commercially important fish stocks in the northwest Atlantic Ocean. She became the first scientist to dive into the Gulf of Mexico oil slick following the 2010 BP Deepwater Horizon oil rig explosion to investigate the impacts of chemical dispersants used in response to the spill.
Bicyclic phosphate is a class of organophosphate compounds that are used as flame retardants, stabilizers and antioxidants. They are also used in spectroscopic studies.
The Shaw Institute, formerly the Marine & Environmental Research Institute, is a 501(c)(3) nonprofit scientific research organization based in Blue Hill, Maine and New York City. The institute conducts research into ocean pollution, flame retardants, microplastics and plastic pollution, sentinel species and climate change.

Bis(2-ethylhexyl)tetrabromophthalate (or TBPH), is a brominated phthalate derivative with the formula C24H34Br4O4 commonly used as a brominated flame retardant (BFR).

Tris(2-ethylhexyl)phosphate (TEHP) is an organic chemical compound in the organophosphate group. It is a triakylphosphate.














