
Dess–Martin periodinane (DMP) is a chemical reagent used in the Dess–Martin oxidation, oxidizing primary alcohols to aldehydes and secondary alcohols to ketones. This periodinane has several advantages over chromium- and DMSO-based oxidants that include milder conditions, shorter reaction times, higher yields, simplified workups, high chemoselectivity, tolerance of sensitive functional groups, and a long shelf life. However, use on an industrial scale is made difficult by its cost and its potentially explosive nature. It is named after the American chemists Daniel Benjamin Dess and James Cullen Martin who developed the reagent in 1983. It is based on IBX, but due to the acetate groups attached to the central iodine atom, DMP is much more reactive than IBX and is much more soluble in organic solvents.

Periodinanes also known as λ5-iodanes are organoiodine compounds with iodine in the +5 oxidation state. These compounds are described as hypervalent because the iodine center has more than 8 valence electrons.
The Griess test is an analytical chemistry test which detects the presence of nitrite ion in solution. One of its most important uses is the determination of nitrite in drinking water. The Griess diazotization reaction, on which the Griess reagent relies, was first described in 1858 by Peter Griess. The test has also been widely used for the detection of nitrates, which are a common component of explosives, as they can be reduced to nitrites and detected with the Griess test.
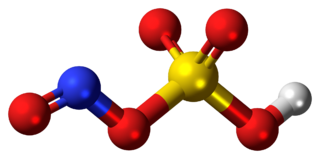
Nitrosylsulfuric acid is the chemical compound with the formula HSO4NO. It is a colourless solid that is used industrially in the production of caprolactam, and was formerly part of the lead chamber process for producing sulfuric acid. The compound is the mixed anhydride of sulfuric acid and nitrous acid.

Methyl red (2-(N,N-dimethyl-4-aminophenyl) azobenzenecarboxylic acid), also called C.I. Acid Red 2, is an indicator dye that turns red in acidic solutions. It is an azo dye, and is a dark red crystalline powder. Methyl red is a pH indicator; it is red in pH under 4.4, yellow in pH over 6.2, and orange in between, with a pKa of 5.1. Murexide and methyl red are investigated as promising enhancers of sonochemical destruction of chlorinated hydrocarbon pollutants. Methyl red is classed by the IARC in group 3 - unclassified as to carcinogenic potential in humans.
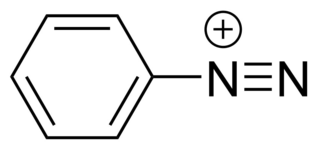
Diazonium compounds or diazonium salts are a group of organic compounds sharing a common functional group [R−N+≡N]X− where R can be any organic group, such as an alkyl or an aryl, and X is an inorganic or organic anion, such as a halide. The parent compound where R is hydrogen, is diazenylium.
The Niementowski quinoline synthesis is the chemical reaction of anthranilic acids and ketones to form γ-hydroxyquinoline derivatives.

Iodobenzene is an aryl iodide and the simplest of the iodobenzenes, consisting of a benzene ring substituted with one iodine atom. Its chemical formula is C6H5I. It is useful as a synthetic intermediate in organic chemistry. It is a volatile colorless liquid, although aged samples appear yellowish.

Iodine monochloride is an interhalogen compound with the formula ICl. It is a red-brown chemical compound that melts near room temperature. Because of the difference in the electronegativity of iodine and chlorine, this molecule is highly polar and behaves as a source of I+. Discovered in 1814 by Gay-Lussac, iodine monochloride is the first interhalogen compound discovered.

Anthranilic acid is an aromatic acid with the formula C6H4(NH2)(CO2H) and has a sweetish taste. The molecule consists of a benzene ring, ortho-substituted with a carboxylic acid and an amine. As a result of containing both acidic and basic functional groups, the compound is amphoteric. Anthranilic acid is a white solid when pure, although commercial samples may appear yellow. The anion [C6H4(NH2)(CO2)]−, obtained by the deprotonation of anthranilic acid, is called anthranilate. Anthranilic acid was once thought to be a vitamin and was referred to as vitamin L1 in that context, but it is now known to be non-essential in human nutrition.

2-Naphthylamine is one of two isomeric aminonaphthalenes, compounds with the formula C10H7NH2. It is a colorless solid, but samples take on a reddish color in air because of oxidation. It was formerly used to make azo dyes, but it is a known carcinogen and has largely been replaced by less toxic compounds.
The Balz–Schiemann reaction is a chemical reaction in which a primary aromatic amine is transformed to an aryl fluoride via a diazonium tetrafluoroborate intermediate. This reaction is a traditional route to fluorobenzene and some related derivatives, including 4-fluorobenzoic acid.

Methyl anthranilate, also known as MA, methyl 2-aminobenzoate, or carbomethoxyaniline, is an ester of anthranilic acid. Its chemical formula is C8H9NO2. It has a strong and fruity grape smell, and one of its key uses is as a flavoring agent.
In enzymology, an anthranilate 1,2-dioxygenase (deaminating, decarboxylating) (EC 1.14.12.1) is an enzyme that catalyzes the chemical reaction
In enzymology, an anthranilate 3-monooxygenase (EC 1.14.16.3) is an enzyme that catalyzes the chemical reaction

Benzenediazonium tetrafluoroborate is an organic compound with the formula [C6H5N2]BF4. It is a salt of a diazonium cation and tetrafluoroborate. It exists as a colourless solid that is soluble in polar solvents. It is the parent member of the aryldiazonium compounds, which are widely used in organic chemistry.
Thiosalicylic acid is an organosulfur compound containing carboxyl and sulfhydryl functional groups. Its molecular formula is ortho-C6H4(−SH)(−C −OH). It is a yellow solid that is slightly soluble in water, ethanol and diethyl ether, and alkanes, but more soluble in DMSO.
The Pschorr cyclization is a name reaction in organic chemistry, which was named after its discoverer, the German chemist Robert Pschorr (1868-1930). It describes the intramolecular substitution of aromatic compounds via aryldiazonium salts as intermediates and is catalyzed by copper. The reaction is a variant of the Gomberg-Bachmann reaction. The following reaction scheme shows the Pschorr cyclization for the example of phenanthrene:

Isatoic anhydride is an organic compound derived from anthranilic acid. A white solid, it is prepared by reaction of anthranilic acid with phosgene.

Diphenic acid, also known as Dibenzoic acid, is an organic compound with the formula (C6H4CO2H)2. It is the most studied of several isomeric dicarboxylic acids of biphenyl. It is a white solid that can be prepared in the laboratory from anthranilic acid via the diazonium salt. It is the product of the microbial action on phenanthrene.













