In chemistry, pyran, or oxine, is a six-membered heterocyclic, non-aromatic ring, consisting of five carbon atoms and one oxygen atom and containing two double bonds. The molecular formula is C5H6O. There are two isomers of pyran that differ by the location of the double bonds. In 2H-pyran, the saturated carbon is at position 2, whereas, in 4H-pyran, the saturated carbon is at position 4.

In organic chemistry, nitro compounds are organic compounds that contain one or more nitro functional groups. The nitro group is one of the most common explosophores used globally. The nitro group is also strongly electron-withdrawing. Because of this property, C−H bonds alpha (adjacent) to the nitro group can be acidic. For similar reasons, the presence of nitro groups in aromatic compounds retards electrophilic aromatic substitution but facilitates nucleophilic aromatic substitution. Nitro groups are rarely found in nature. They are almost invariably produced by nitration reactions starting with nitric acid.

Tautomers are structural isomers of chemical compounds that readily interconvert. The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hydrogen atom within the compound. The phenomenon of tautomerization is called tautomerism, also called desmotropism. Tautomerism is for example relevant to the behavior of amino acids and nucleic acids, two of the fundamental building blocks of life.
In organic chemistry, the Ugi reaction is a multi-component reaction involving a ketone or aldehyde, an amine, an isocyanide and a carboxylic acid to form a bis-amide. The reaction is named after Ivar Karl Ugi, who first reported this reaction in 1959.

2-Pyridone is an organic compound with the formula C
5H
4NH(O). It is a colourless solid. It is well known to form hydrogen bonded dimers and it is also a classic case of a compound that exists as tautomers.
Pyrones or pyranones are a class of heterocyclic chemical compounds. They contain an unsaturated six-membered lactone ring, which has one oxygen atom and an adjacent ketone functional group. There are two isomers denoted as 2-pyrone and 4-pyrone. The 2-pyrone structure is found in nature as part of the coumarin ring system. 4-Pyrone is found in some natural chemical compounds such as chromone, maltol and kojic acid.
Pyrylium is a cation with formula C5H5O+, consisting of a six-membered ring of five carbon atoms, each with one hydrogen atom, and one positively charged oxygen atom. The bonds in the ring are conjugated as in benzene, giving it an aromatic character. In particular, because of the positive charge, the oxygen atom is trivalent. Pyrilium is a mono-cyclic and heterocyclic compound, one of the oxonium ions.
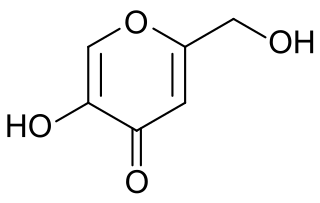
Kojic acid is an organic compound with the formula HOCH2C5H2O2OH. It is a derivative of 4-pyrone that functions in nature as a chelation agent produced by several species of fungi, especially Aspergillus oryzae, which has the Japanese common name koji. Kojic acid is a by-product in the fermentation process of malting rice, for use in the manufacturing of sake, the Japanese rice wine. It is a mild inhibitor of the formation of pigment in plant and animal tissues, and is used in food and cosmetics to preserve or change colors of substances. It forms a bright red complex with ferric ions.

A boronic acid is an organic compound related to boric acid in which one of the three hydroxyl groups is replaced by an alkyl or aryl group. As a compound containing a carbon–boron bond, members of this class thus belong to the larger class of organoboranes.

Dehydroacetic acid is an organic compound which has several industrial applications. The compound is classified as a pyrone derivative. It presents as an odorless, colorless to white crystalline powder, almost insoluble in water and moderately soluble in most organic solvents.

In organic chemistry, ethenone is the formal name for ketene, an organic compound with formula C2H2O or H2C=C=O. It is the simplest member of the ketene class. It is an important reagent for acetylations.
2-Pyrone (α-pyrone or pyran-2-one) is an unsaturated cyclic chemical compound with the molecular formula C5H4O2. It is isomeric with 4-pyrone.

Sir Jocelyn Field Thorpe FRS was a British chemist who made major contributions to organic chemistry, including the Thorpe-Ingold effect and three named reactions.

2-Mercaptopyridine is an organosulfur compound with the formula HSC5H4N. This yellow crystalline solid is a derivative of pyridine. The compound and its derivatives serve primarily as acylating agents. A few of 2-mercaptopyridine’s other uses include serving as a protecting group for amines and imides as well as forming a selective reducing agent. 2-Mercaptopyridine oxidizes to 2,2’-dipyridyl disulfide.
The retro-Diels–Alder reaction is the reverse of the Diels–Alder (DA) reaction, a [4+2] cycloelimination. It involves the formation of a diene and dienophile from a cyclohexene. It can be accomplished spontaneously with heat, or with acid or base mediation.
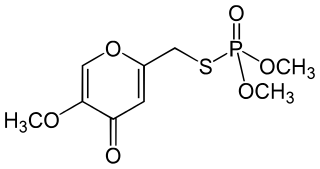
Endothion is an organic compound used as an insecticide and acaricides. It is part of the chemical class of organophosphorus compounds. It is generally described as white crystals with a slight odor. It is used as an insecticide, but not sold in the United States or Canada.

Chelidonic acid is a heterocyclic organic acid with a pyran skeleton.
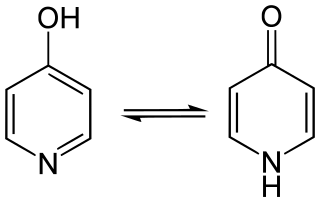
4-Pyridone is an organic compound with the formula C
5H
4NH(O). It is a colorless solid.

Tetronic acid is a chemical compound, classified as a γ-lactone, with the molecular formula C4H4O3.

6-Amyl-α-pyrone, also 6-pentyl-2-pyrone or 6PP, is an unsaturated lactone molecule. It contains two double bonds in the ring and a pentyl substituent at carbon adjacent to the ring oxygen. It is a colorless liquid which possesses characteristic coconut aroma, produced biologically by Trichoderma species. It is found in animal foods, peach, and heated beef.
















