
A pigment is a powder used to add color or change visual appearance. Pigments are completely or nearly insoluble and chemically unreactive in water or another medium; in contrast, dyes are colored substances which are soluble or go into solution at some stage in their use. Dyes are often organic compounds whereas pigments are often inorganic. Pigments of prehistoric and historic value include ochre, charcoal, and lapis lazuli.

Nils Gabriel Sefström was a Swedish chemist. Sefström was a student of Berzelius and, when studying the brittleness of steel in 1830, he rediscovered a new chemical element, to which he gave the name vanadium.

Wilhelm Carl Werner Otto Fritz Franz Wien was a German physicist who, in 1893, used theories about heat and electromagnetism to deduce Wien's displacement law, which calculates the emission of a blackbody at any temperature from the emission at any one reference temperature.

Rudolf Julius Emanuel Clausius was a German physicist and mathematician and is considered one of the central founding fathers of the science of thermodynamics. By his restatement of Sadi Carnot's principle known as the Carnot cycle, he gave the theory of heat a truer and sounder basis. His most important paper, "On the Moving Force of Heat", published in 1850, first stated the basic ideas of the second law of thermodynamics. In 1865 he introduced the concept of entropy. In 1870 he introduced the virial theorem, which applied to heat.

Thermionic emission is the liberation of electrons from an electrode by virtue of its temperature. This occurs because the thermal energy given to the charge carrier overcomes the work function of the material. The charge carriers can be electrons or ions, and in older literature are sometimes referred to as thermions. After emission, a charge that is equal in magnitude and opposite in sign to the total charge emitted is initially left behind in the emitting region. But if the emitter is connected to a battery, the charge left behind is neutralized by charge supplied by the battery as the emitted charge carriers move away from the emitter, and finally the emitter will be in the same state as it was before emission.

Johann Wolfgang Döbereiner was a German chemist who is known best for work that was suggestive of the periodic law for the chemical elements, and for inventing the first lighter, which was known as the Döbereiner's lamp. He became a professor of chemistry and pharmacy for the University of Jena.

Louis Carl Heinrich Friedrich Paschen, was a German physicist, known for his work on electrical discharges. He is also known for the Paschen series, a series of hydrogen spectral lines in the infrared region that he first observed in 1908. He established the now widely used Paschen curve in his article "Über die zum Funkenübergang in Luft, Wasserstoff und Kohlensäure bei verschiedenen Drücken erforderliche Potentialdifferenz". He is known for the Paschen-Back effect, which is the Zeeman effect's becoming non-linear at high magnetic field. He helped explain the hollow cathode effect in 1916.

Richard Adolf Zsigmondy was an Austrian-born chemist. He was known for his research in colloids, for which he was awarded the Nobel Prize in chemistry in 1925, as well as for co-inventing the slit-ultramicroscope, and different membrane filters. The crater Zsigmondy on the Moon is named in his honour.

Friedlieb Ferdinand Runge was a German analytical chemist. Runge identified the mydriatic effects of belladonna extract, identified caffeine, and discovered the first coal tar dye.

Oil paint is a type of slow-drying paint that consists of particles of pigment suspended in a drying oil, commonly linseed oil. The viscosity of the paint may be modified by the addition of a solvent such as turpentine or white spirit, and varnish may be added to increase the glossiness of the dried oil paint film. The addition of oil or alkyd medium can also be used to modify the viscosity and drying time of oil paint. Oil paints were first used in Asia as early as the 7th century AD and can be seen in examples of Buddhist paintings in Afghanistan. Oil-based paints made their way to Europe by the 12th century and were used for simple decoration, but oil painting did not begin to be adopted as an artistic medium there until the early 15th century. Common modern applications of oil paint are in finishing and protection of wood in buildings and exposed metal structures such as ships and bridges. Its hard-wearing properties and luminous colors make it desirable for both interior and exterior use on wood and metal. Due to its slow-drying properties, it has recently been used in paint-on-glass animation. The thickness of the coat has considerable bearing on the time required for drying: thin coats of oil paint dry relatively quickly.

Annalen der Physik is one of the oldest scientific journals on physics; it has been published since 1799. The journal publishes original, peer-reviewed papers on experimental, theoretical, applied, and mathematical physics and related areas. The editor-in-chief is Stefan Hildebrandt. Prior to 2008, its ISO 4 abbreviation was Ann. Phys. (Leipzig), after 2008 it became Ann. Phys. (Berl.).

The Zöllner illusion is an optical illusion named after its discoverer, German astrophysicist Johann Karl Friedrich Zöllner. In 1860, Zöllner sent his discovery in a letter to physicist and scholar Johann Christian Poggendorff, editor of Annalen der Physik und Chemie, who subsequently discovered the related Poggendorff illusion in Zöllner's original drawing.

Pollucite is a zeolite mineral with the formula (Cs,Na)2Al2Si4O12·2H2O with iron, calcium, rubidium and potassium as common substituting elements. It is important as a significant ore of caesium and sometimes rubidium. It forms a solid solution series with analcime. It crystallizes in the isometric - hexoctahedral crystal system as colorless, white, gray, or rarely pink and blue masses. Well formed crystals are rare. It has a Mohs hardness of 6.5 and a specific gravity of 2.9. It has a brittle fracture and no cleavage.

Alfred Heinrich Bucherer was a German physicist, who is known for his experiments on relativistic mass. He also was the first who used the phrase "theory of relativity" for Einstein's theory of special relativity.

Mellitic anhydride, the anhydride of mellitic acid, is an organic compound with the formula C12O9.
In physics, a quantum is the minimum amount of any physical entity involved in an interaction.
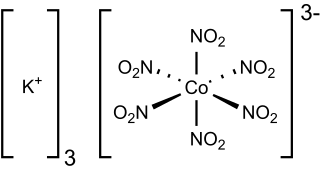
Potassium hexanitritocobaltate(III) is a salt with the formula K3[Co(NO2)6]. It is a yellow solid that is poorly soluble in water. The compound finds some use as a yellow pigment under the name Indian Yellow.
Wasium was the suggested name of a chemical element found by J. F. Bahr. The name was derived from the House of Vasa the Royal House of Sweden.

Thiocarbonic acid is an inorganic acid with the chemical formula H2CS3. It is an analog of carbonic acid H2CO3, in which all oxygen atoms are replaced with sulfur atoms. It is an unstable hydrophobic red oily liquid.
















