Hypoxia is a condition in which the body or a region of the body is deprived of adequate oxygen supply at the tissue level. Hypoxia may be classified as either generalized, affecting the whole body, or local, affecting a region of the body. Although hypoxia is often a pathological condition, variations in arterial oxygen concentrations can be part of the normal physiology, for example, during hypoventilation training or strenuous physical exercise.
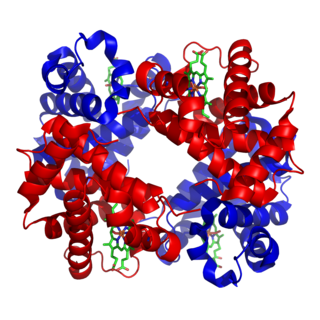
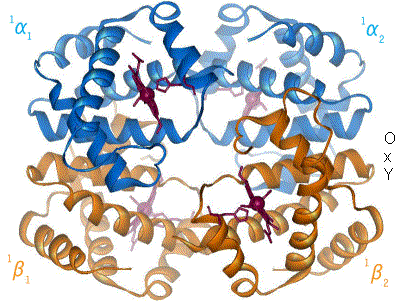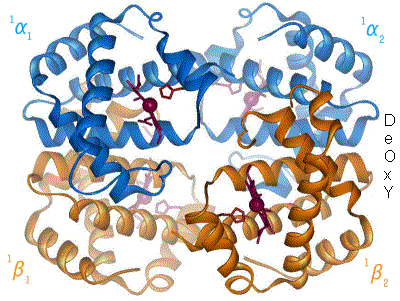
Hemoglobin, or haemoglobin, abbreviated Hb or Hgb, is the iron-containing oxygen-transport metalloprotein in the red blood cells (erythrocytes) of almost all vertebrates as well as the tissues of some invertebrates. Hemoglobin in blood carries oxygen from the lungs or gills to the rest of the body. There it releases the oxygen to permit aerobic respiration to provide energy to power the functions of the organism in the process called metabolism. A healthy individual has 12 to 20 grams of hemoglobin in every 100 mL of blood.
Red blood cells (RBCs), also referred to as red cells, red blood corpuscles (in humans or other animals not having nucleus in red blood cells), haematids, erythroid cells or erythrocytes (from Greek erythros for "red" and kytos for "hollow vessel", with -cyte translated as "cell" in modern usage), are the most common type of blood cell and the vertebrate's principal means of delivering oxygen (O2) to the body tissues—via blood flow through the circulatory system. RBCs take up oxygen in the lungs, or in fish the gills, and release it into tissues while squeezing through the body's capillaries.

Hemoglobinopathy is the medical term for a group of inherited blood disorders and diseases that primarily affect red blood cells. They are single-gene disorders and, in most cases, they are inherited as autosomal co-dominant traits.
Diffusing capacity of the lung (DL) measures the transfer of gas from air in the lung, to the red blood cells in lung blood vessels. It is part of a comprehensive series of pulmonary function tests to determine the overall ability of the lung to transport gas into and out of the blood. DL, especially DLCO, is reduced in certain diseases of the lung and heart. DLCO measurement has been standardized according to a position paper by a task force of the European Respiratory and American Thoracic Societies.

Christian Harald Lauritz Peter Emil Bohr (1855–1911) was a Danish physician, father of the physicist and Nobel laureate Niels Bohr, as well as the mathematician and football player Harald Bohr and grandfather of another physicist and Nobel laureate Aage Bohr. He married Ellen Adler in 1881.
An arterial blood gas (ABG) test measures the amounts of arterial gases, such as oxygen and carbon dioxide. An ABG test requires that a small volume of blood be drawn from the radial artery with a syringe and a thin needle, but sometimes the femoral artery in the groin or another site is used. The blood can also be drawn from an arterial catheter.
Fetal hemoglobin, or foetal haemoglobin is the main oxygen carrier protein in the human fetus. Hemoglobin F is found in fetal red blood cells, and is involved in transporting oxygen from the mother's bloodstream to organs and tissues in the fetus. It is produced at around 6 weeks of pregnancy and the levels remain high after birth until the baby is roughly 2–4 months old. Hemoglobin F has a different composition from the adult forms of hemoglobin, which allows it to bind oxygen more strongly. This way, the developing fetus is able to retrieve oxygen from the mother's bloodstream, which occurs through the placenta found in the mother's uterus.
Carboxyhemoglobin, or carboxyhaemoglobin, is a stable complex of carbon monoxide and hemoglobin (Hb) that forms in red blood cells upon contact with carbon monoxide. Carboxyhemoglobin is often mistaken for the compound formed by the combination of carbon dioxide (carboxyl) and hemoglobin, which is actually carbaminohemoglobin. Carboxyhemoglobin terminology emerged when carbon monoxide was known by its ancient name carbonic oxide; the preferred IUPAC nomenclature is carbonylhemoglobin.
Hypocapnia, also known as hypocarbia, sometimes incorrectly called acapnia, is a state of reduced carbon dioxide in the blood. Hypocapnia usually results from deep or rapid breathing, known as hyperventilation.
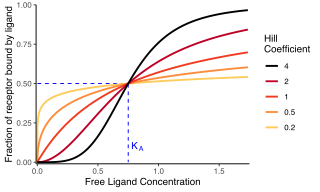
In biochemistry and pharmacology, the Hill equation refers to two closely related equations that reflect the binding of ligands to macromolecules, as a function of the ligand concentration. A ligand is "a substance that forms a complex with a biomolecule to serve a biological purpose", and a macromolecule is a very large molecule, such as a protein, with a complex structure of components. Protein-ligand binding typically changes the structure of the target protein, thereby changing its function in a cell.
The oxygen–hemoglobin dissociation curve, also called the oxyhemoglobin dissociation curve or oxygen dissociation curve (ODC), is a curve that plots the proportion of hemoglobin in its saturated (oxygen-laden) form on the vertical axis against the prevailing oxygen tension on the horizontal axis. This curve is an important tool for understanding how our blood carries and releases oxygen. Specifically, the oxyhemoglobin dissociation curve relates oxygen saturation (SO2) and partial pressure of oxygen in the blood (PO2), and is determined by what is called "hemoglobin affinity for oxygen"; that is, how readily hemoglobin acquires and releases oxygen molecules into the fluid that surrounds it.
Carbaminohemoglobin (or carbaminohaemoglobin, also known as carbhemoglobin and carbohemoglobin) is a compound of hemoglobin and carbon dioxide, and is one of the forms in which carbon dioxide exists in the blood. Twenty-three percent of carbon dioxide is carried in blood this way (70% is converted into bicarbonate by carbonic anhydrase and then carried in plasma, 7% carried as free CO2, dissolved in plasma).
The Haldane effect is a property of hemoglobin first described by John Scott Haldane, within which oxygenation of blood in the lungs displaces carbon dioxide from hemoglobin, increasing the removal of carbon dioxide. Consequently, oxygenated blood has a reduced affinity for carbon dioxide. Thus, the Haldane effect describes the ability of hemoglobin to carry increased amounts of carbon dioxide (CO2) in the deoxygenated state as opposed to the oxygenated state. A high concentration of CO2 facilitates dissociation of oxyhemoglobin.

2,3-Bisphosphoglyceric acid (2,3-BPG), also known as 2,3-diphosphoglyceric acid (2,3-DPG), is a three-carbon isomer of the glycolytic intermediate 1,3-bisphosphoglyceric acid (1,3-BPG). 2,3-BPG is present in human red blood cells at approximately 5 mmol/L. It binds with greater affinity to deoxygenated hemoglobin than it does to oxygenated hemoglobin due to conformational differences: 2,3-BPG fits in the deoxygenated hemoglobin conformation, but not as well in the oxygenated conformation. It interacts with deoxygenated hemoglobin beta subunits and so it decreases the affinity for oxygen and allosterically promotes the release of the remaining oxygen molecules bound to the hemoglobin; therefore it enhances the ability of RBCs to release oxygen near tissues that need it most. 2,3-BPG is thus an allosteric effector.
Chloride shift (also known as the Hamburger phenomenon or lineas phenomenon, named after Hartog Jakob Hamburger) is a process which occurs in a cardiovascular system and refers to the exchange of bicarbonate (HCO3−) and chloride (Cl−) across the membrane of red blood cells (RBCs).

Efaproxiral (INN) is an analogue of the cholesterol drug bezafibrate developed for the treatment of depression, traumatic brain injury, ischemia, stroke, myocardial infarction, diabetes, hypoxia, sickle cell disease, hypercholesterolemia and as a radio sensitiser.

Oxygen saturation is the fraction of oxygen-saturated hemoglobin relative to total hemoglobin in the blood. The human body requires and regulates a very precise and specific balance of oxygen in the blood. Normal arterial blood oxygen saturation levels in humans are 95–100 percent. If the level is below 90 percent, it is considered low and called hypoxemia. Arterial blood oxygen levels below 80 percent may compromise organ function, such as the brain and heart, and should be promptly addressed. Continued low oxygen levels may lead to respiratory or cardiac arrest. Oxygen therapy may be used to assist in raising blood oxygen levels. Oxygenation occurs when oxygen molecules enter the tissues of the body. For example, blood is oxygenated in the lungs, where oxygen molecules travel from the air and into the blood. Oxygenation is commonly used to refer to medical oxygen saturation.
The Root effect is a physiological phenomenon that occurs in fish hemoglobin, named after its discoverer R. W. Root. It is the phenomenon where an increased proton or carbon dioxide concentration (lower pH) lowers hemoglobin's affinity and carrying capacity for oxygen. The Root effect is to be distinguished from the Bohr effect where only the affinity to oxygen is reduced. Hemoglobins showing the Root effect show a loss of cooperativity at low pH. This results in the Hb-O2 dissociation curve being shifted downward and not just to the right. At low pH, hemoglobins showing the Root effect don't become fully oxygenated even at oxygen tensions up to 20kPa. This effect allows hemoglobin in fish with swim bladders to unload oxygen into the swim bladder against a high oxygen gradient. The effect is also noted in the choroid rete, the network of blood vessels which carries oxygen to the retina. In the absence of the Root effect, retia will result in the diffusion of some oxygen directly from the arterial blood to the venous blood, making such systems less effective for the concentration of oxygen. It has also been hypothesized that the loss of affinity is used to provide more oxygen to red muscle during acidotic stress.

The carbonic anhydrases form a family of enzymes that catalyze the interconversion between carbon dioxide and water and the dissociated ions of carbonic acid. The active site of most carbonic anhydrases contains a zinc ion. They are therefore classified as metalloenzymes. The enzyme maintains acid-base balance and helps transport carbon dioxide.



![The magnitude of the Bohr effect is given by
D
log
[?]
(
P
50
)
D
pH
{\textstyle {\scriptstyle \Delta \log(P_{50}) \over \Delta {\text{pH}}}}
, which is the slope on this graph. A steeper slope means a stronger Bohr effect. Bohr Effect Magnitude vs Body Size.png](http://upload.wikimedia.org/wikipedia/commons/thumb/3/33/Bohr_Effect_Magnitude_vs_Body_Size.png/330px-Bohr_Effect_Magnitude_vs_Body_Size.png)