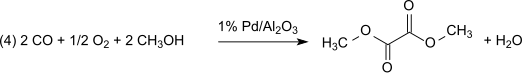| |
 | |
| Names | |
|---|---|
| Preferred IUPAC name Dimethyl oxalate | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.008.231 |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C4H6O4 | |
| Molar mass | 118.088 g·mol−1 |
| Appearance | White crystals |
| Melting point | 53 to 55 °C (127 to 131 °F; 326 to 328 K) [1] |
| Boiling point | 166 to 167 °C (331 to 333 °F; 439 to 440 K) [1] |
| −55.7·10−6 cm3/mol | |
| Related compounds | |
Related compounds | Diethyl oxalate, Diphenyl oxalate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
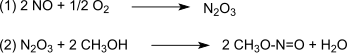
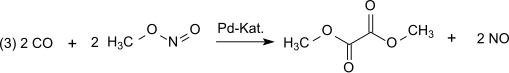
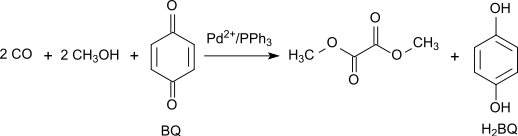
Dimethyl oxalate is an organic compound with the formula (CO2CH3)2 or (CH3)2C2O4. It is the dimethyl ester of oxalic acid. Dimethyl oxalate is a colorless or white solid that is soluble in water.