Riboflavin, also known as vitamin B2, is a vitamin found in food and sold as a dietary supplement. It is essential to the formation of two major coenzymes, flavin mononucleotide and flavin adenine dinucleotide. These coenzymes are involved in energy metabolism, cellular respiration, and antibody production, as well as normal growth and development. The coenzymes are also required for the metabolism of niacin, vitamin B6, and folate. Riboflavin is prescribed to treat corneal thinning, and taken orally, may reduce the incidence of migraine headaches in adults.

Indigo dye is an organic compound with a distinctive blue color. Indigo is a natural dye obtained from the leaves of some plants of the Indigofera genus, in particular Indigofera tinctoria. Dye-bearing Indigofera plants were once common throughout the world. It is now produced via chemical routes. Blue colorants are rare. Since indigo is insoluble, it is also referred to as a pigment.

Jaundice, also known as icterus, is a yellowish or greenish pigmentation of the skin and sclera due to high bilirubin levels. Jaundice in adults is typically a sign indicating the presence of underlying diseases involving abnormal heme metabolism, liver dysfunction, or biliary-tract obstruction. The prevalence of jaundice in adults is rare, while jaundice in babies is common, with an estimated 80% affected during their first week of life. The most commonly associated symptoms of jaundice are itchiness, pale feces, and dark urine.

Bilirubin (BR) is a red-orange compound that occurs in the normal catabolic pathway that breaks down heme in vertebrates. This catabolism is a necessary process in the body's clearance of waste products that arise from the destruction of aged or abnormal red blood cells. In the first step of bilirubin synthesis, the heme molecule is stripped from the hemoglobin molecule. Heme then passes through various processes of porphyrin catabolism, which varies according to the region of the body in which the breakdown occurs. For example, the molecules excreted in the urine differ from those in the feces. The production of biliverdin from heme is the first major step in the catabolic pathway, after which the enzyme biliverdin reductase performs the second step, producing bilirubin from biliverdin.

Heme, or haem, is a ring-shaped iron-containing molecular component of hemoglobin, which is necessary to bind oxygen in the bloodstream. It is composed of four pyrrole rings with 2 vinyl and 2 propionic acid side chains. Heme is biosynthesized in both the bone marrow and the liver.

Excretion is elimination of metabolic waste, which is an essential process in all organisms. In vertebrates, this is primarily carried out by the lungs, kidneys, and skin. This is in contrast with secretion, where the substance may have specific tasks after leaving the cell. For example, placental mammals expel urine from the bladder through the urethra, which is part of the excretory system. Unicellular organisms discharge waste products directly through the surface of the cell.

ADME is the four-letter abbreviation (acronym) for absorption, distribution, metabolism, and excretion, and is mainly used in fields such as pharmacokinetics and pharmacology. The four letter stands for descriptors quantifying how a given drug interacts within body over time. The term ADME was first introduced in the 1960s, and has become a standard term widely used in scientific literature, teaching, drug regulations, and clinical practice.
Drug metabolism is the metabolic breakdown of drugs by living organisms, usually through specialized enzymatic systems. More generally, xenobiotic metabolism is the set of metabolic pathways that modify the chemical structure of xenobiotics, which are compounds foreign to an organism's normal biochemistry, such as any drug or poison. These pathways are a form of biotransformation present in all major groups of organisms and are considered to be of ancient origin. These reactions often act to detoxify poisonous compounds. The study of drug metabolism is the object of pharmacokinetics. Metabolism is one of the stages of the drug's transit through the body that involves the breakdown of the drug so that it can be excreted by the body.

Enterohepatic circulation is the circulation of biliary acids, bilirubin, drugs or other substances from the liver to the bile, followed by entry into the small intestine, absorption by the enterocyte and transport back to the liver. Enterohepatic circulation is an especially important concept in the field of toxicology as many lipophilic xenobiotics undergo this process causing repeated liver damage.

Hartnup disease is an autosomal recessive metabolic disorder affecting the absorption of nonpolar amino acids. Niacin is a precursor to nicotinamide, a necessary component of NAD+.

Urobilinogen is a yellow by-product of bilirubin reduction. It is formed in the intestines by the bacterial enzyme bilirubin reductase. About half of the urobilinogen formed is reabsorbed and taken up via the portal vein to the liver, enters circulation and is excreted by the kidney.

Uridine 5'-diphospho-glucuronosyltransferase is a microsomal glycosyltransferase that catalyzes the transfer of the glucuronic acid component of UDP-glucuronic acid to a small hydrophobic molecule. This is a glucuronidation reaction.
Metabolic wastes or excrements are substances left over from metabolic processes (such as cellular respiration) which cannot be used by the organism (they are surplus or toxic), and must therefore be excreted. This includes nitrogen compounds, water, CO2, phosphates, sulphates, etc. Animals treat these compounds as excretes. Plants have metabolic pathways which transforms some of them (primarily the oxygen compounds) into useful substances.

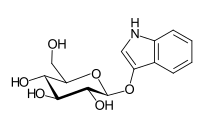
In organic chemistry, indoxyl is a nitrogenous substance with the chemical formula: C8H7NO. Indoxyl is isomeric with oxindol and is obtained as an oily liquid.

Glucobrassicin is a type of glucosinolate that can be found in almost all cruciferous plants, such as cabbages, broccoli, mustards, and woad. As for other glucosinolates, degradation by the enzyme myrosinase is expected to produce an isothiocyanate, indol-3-ylmethylisothiocyanate. However, this specific isothiocyanate is expected to be highly unstable, and has indeed never been detected. The observed hydrolysis products when isolated glucobrassicin is degraded by myrosinase are indole-3-carbinol and thiocyanate ion, which are envisioned to result from a rapid reaction of the unstable isothiocyanate with water. However, a large number of other reaction products are known, and indole-3-carbinol is not the dominant degradation product when glucosinolate degradation takes place in crushed plant tissue or in intact plants.

Henry Edward Schunck, also known as Edward von Schunck, was a British chemist who did much work with dyes.

Stercobilinogen is a chemical created by bacteria in the gut. It is made of broken-down hemoglobin. It is further processed to become the chemical that gives feces its brown color.

Phenylacetylglutamine is a product formed by the conjugation of phenylacetate and glutamine. It is a common metabolite that occurs naturally in human urine.

A urine test strip or dipstick is a basic diagnostic tool used to determine pathological changes in a patient's urine in standard urinalysis.
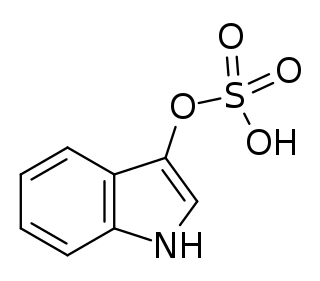
Indoxyl sulfate, also known as 3-indoxylsulfate and 3-indoxylsulfuric acid, is a metabolite of dietary L-tryptophan that acts as a cardiotoxin and uremic toxin. High concentrations of indoxyl sulfate in blood plasma are known to be associated with the development and progression of chronic kidney disease and vascular disease in humans. As a uremic toxin, it stimulates glomerular sclerosis and renal interstitial fibrosis.