
Lipoxygenases are a family of (non-heme) iron-containing enzymes most of which catalyze the dioxygenation of polyunsaturated fatty acids in lipids containing a cis,cis-1,4- pentadiene into cell signaling agents that serve diverse roles as autocrine signals that regulate the function of their parent cells, paracrine signals that regulate the function of nearby cells, and endocrine signals that regulate the function of distant cells.

ALOX15 is, like other lipoxygenases, a seminal enzyme in the metabolism of polyunsaturated fatty acids to a wide range of physiologically and pathologically important products. ▼ Gene Function
In enzymology, a linoleate 11-lipoxygenase (EC 1.13.11.45) is an enzyme that catalyzes the chemical reaction
In enzymology, a linoleate diol synthase (EC 1.13.11.44) is an enzyme that catalyzes the chemical reaction

Cyclooxygenase 1 (COX-1), also known as prostaglandin G/H synthase 1, prostaglandin-endoperoxide synthase 1 or prostaglandin H2 synthase 1, is an enzyme that in humans is encoded by the PTGS1 gene. In humans it is one of two cyclooxygenases.

Arachidonate 15-lipoxygenase type II is an enzyme that in humans is encoded by the ALOX15B gene. ALOX15B, also known as 15-lipoxygenase-2, is distinguished from its related oxygenase, ALOX15 or 15-lipoxygenase-1.

Arachidonate 12-lipoxygenase, 12R type, also known as ALOX12B, 12R-LOX, and arachidonate lipoxygenase 3, is a lipoxygenase-type enzyme composed of 701 amino acids and encoded by the ALOX12B gene. The gene is located on chromosome 17 at position 13.1 where it forms a cluster with two other lipoxygenases, ALOXE3 and ALOX15B. Among the human lipoxygenases, ALOX12B is most closely related in amino acid sequence to ALOXE3

Epidermis-type lipoxygenase 3 is a member of the lipoxygenase family of enzymes; in humans, it is encoded by the ALOXE3 gene. This gene is located on chromosome 17 at position 13.1 where it forms a cluster with two other lipoxygenases, ALOX12B and ALOX15B. Among the human lipoxygenases, ALOXE3 is most closely related in amino acid sequence to ALOX12B. ALOXE3, ALOX12B, and ALOX15B are often classified as epidermal lipoxygenases, in distinction to the other three human lipoxygenases, because they were initially defined as being highly or even exclusively expressed and functioning in skin. The epidermis-type lipoxygenases are now regarded as a distinct subclass within the multigene family of mammalian lipoxygenases with mouse Aloxe3 being the ortholog to human ALOXE3, mouse Alox12b being the ortholog to human ALOX12B, and mouse Alox8 being the ortholog to human ALOX15B [supplied by OMIM]. ALOX12B and ALOXE3 in humans, Alox12b and Aloxe3 in mice, and comparable orthologs in other in other species are proposed to act sequentially in a multistep metabolic pathway that forms products that are structurally critical for creating and maintaining the skin's water barrier function.
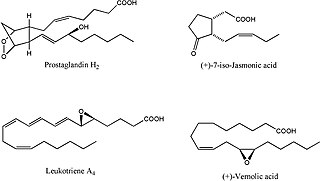
Oxylipins constitute a family of oxygenated natural products which are formed from fatty acids by pathways involving at least one step of dioxygen-dependent oxidation. Oxylipins are derived from polyunsaturated fatty acids (PUFAs) by COX enzymes (cyclooxygenases), by LOX enzymes (lipoxygenases), or by cytochrome P450 epoxygenase.
Apocarotenoids are organic compounds which occur widely in living organisms. They are derived from carotenoids by oxidative cleavage, catalyzed by carotenoid oxygenases. Examples include the vitamin A retinoids retinal, retinoic acid, and retinol; and the plant hormone abscisic acid.
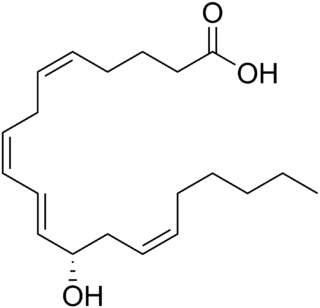
12-Hydroxyeicosatetraenoic acid (12-HETE) is a derivative of the 20 carbon polyunsaturated fatty acid, arachidonic acid, containing a hydroxyl residue at carbon 12 and a 5Z,8Z,10E,14Z Cis–trans isomerism configuration (Z=cis, E=trans) in its four double bonds. It was first found as a product of arachidonic acid metabolism made by human and bovine platelets through their 12S-lipoxygenase (i.e. ALOX12) enzyme(s). However, the term 12-HETE is ambiguous in that it has been used to indicate not only the initially detected "S" stereoisomer, 12S-hydroxy-5Z,8Z,10E,14Z-eicosatetraenoic acid (12(S)-HETE or 12S-HETE), made by platelets, but also the later detected "R" stereoisomer, 12(R)-hydroxy-5Z,8Z,10E,14Z-eicosatetraenoic acid (also termed 12(R)-HETE or 12R-HETE) made by other tissues through their 12R-lipoxygenase enzyme, ALOX12B. The two isomers, either directly or after being further metabolized, have been suggested to be involved in a variety of human physiological and pathological reactions. Unlike hormones which are secreted by cells, travel in the circulation to alter the behavior of distant cells, and thereby act as Endocrine signalling agents, these arachidonic acid metabolites act locally as Autocrine signalling and/or Paracrine signaling agents to regulate the behavior of their cells of origin or of nearby cells, respectively. In these roles, they may amplify or dampen, expand or contract cellular and tissue responses to disturbances.
Linoleate 9S-lipoxygenase (EC 1.13.11.58, 9-lipoxygenase, 9S-lipoxygenase, linoleate 9-lipoxygenase, LOX1 (gene), 9S-LOX) is an enzyme with systematic name linoleate:oxygen 9S-oxidoreductase. This enzyme catalyses the following chemical reaction
Linoleate 8R-lipoxygenase (EC 1.13.11.60, linoleic acid 8R-dioxygenase, 5,8-LDS (bifunctional enzyme), 7,8-LDS (bifunctional enzyme), 5,8-linoleate diol synthase (bifunctional enzyme), 7,8-linoleate diol synthase (bifunctional enzyme), PpoA) is an enzyme with systematic name linoleate:oxygen (8R)-oxidoreductase. This enzyme catalyses the following chemical reaction
Linoleate 10R-lipoxygenase (EC 1.13.11.62, 10R-DOX, (10R)-dioxygenase, 10R-dioxygenase) is an enzyme with systematic name linoleate:oxygen (10R)-oxidoreductase. This enzyme catalyses the following chemical reaction

Geosmin synthase or germacradienol-geosmin synthase designates a class of bifunctional enzymes that catalyze the conversion of farnesyl diphosphate (FPP) to geosmin, a volatile organic compound known for its earthy smell. The N-terminal half of the protein catalyzes the conversion of farnesyl diphosphate to germacradienol and germacrene D, followed by the C-terminal-mediated conversion of germacradienol to geosmin. The conversion of FPP to geosmin was previously thought to involve multiple enzymes in a biosynthetic pathway.
Colneleate synthase (EC 4.2.1.121, 9-divinyl ether synthase, 9-DES, CYP74D, CYP74D1, CYP74 cytochrome P-450, DES1) is an enzyme with systematic name (8E)-9-((1E,3E)-nona-1,3-dien-1-yloxy)non-8-enoate synthase. This enzyme catalyses the following chemical reaction
5-epi-α-Selinene synthase is an enzyme with systematic name (2Z,6E)-farnesyl-diphosphate diphosphate-lyase . This enzyme catalyses the following chemical reaction
9,12-octadecadienoate 8-hydroperoxide 8R-isomerase is an enzyme with systematic name (8R,9Z,12Z)-8-hydroperoxyoctadeca-9,12-dienoate hydroxymutase ( -5,8-dihydroxyoctadeca-9,12-dienoate-forming). This enzyme catalyses the following chemical reaction
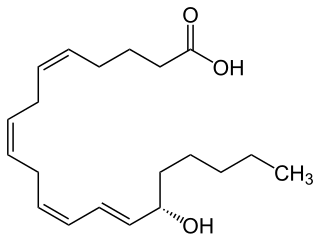
15-Hydroxyeicosatetraenoic acid (also termed 15-HETE, 15(S)-HETE, and 15S-HETE) is an eicosanoid, i.e. a metabolite of arachidonic acid. Various cell types metabolize arachidonic acid to 15(S)-hydroperoxyeicosatetraenoic acid (15(S)-HpETE). This initial hydroperoxide product is extremely short-lived in cells: if not otherwise metabolized, it is rapidly reduced to 15(S)-HETE. Both of these metabolites, depending on the cell type which forms them, can be further metabolized to 15-oxo-eicosatetraenoic acid (15-oxo-ETE), 5S,15S-dihydroxy-eicosatetraenoic acid (5(S),15(S)-diHETE), 5-oxo-15(S)-hydroxyeicosatetraenoic acid (5-oxo-15(S)-HETE, a subset of specialized pro-resolving mediators viz., the lipoxins, a class of pro-inflammatory mediators, the eoxins, and other products that have less well-defined activities and functions. Thus, 15(S)-HETE and 15(S)-HpETE, in addition to having intrinsic biological activities, are key precursors to numerous biologically active derivatives.
Eoxins are proposed to be a family of proinflammatory eicosanoids. They are produced by human eosinophils, mast cells, the L1236 Reed–Sternberg cell line derived from Hodgkin's lymphoma, and certain other tissues. These cells produce the eoxins by initially metabolizing arachidonic acid, an omega-6 (ω-6) fatty acid, via any enzyme possessing 15-lipoxygenase activity. The product of this initial metabolic step, 15(S)-hydroperoxyeicosatetraenoic acid, is then converted to a series of eoxins by the same enzymes that metabolize the 5-lipoxygenase product of arachidonic acid metabolism, i.e. 5-Hydroperoxy-eicosatetraenoic acid to a series of leukotrienes. That is, the eoxins are 14,15-disubstituted analogs of the 5,6-disubstituted leukotrienes.








