
Boric acid, more specifically orthoboric acid, is a compound of boron, oxygen, and hydrogen with formula B(OH)3. It may also be called hydrogen orthoborate, trihydroxidoboron or boracic acid. It is usually encountered as colorless crystals or a white powder, that dissolves in water, and occurs in nature as the mineral sassolite. It is a weak acid that yields various borate anions and salts, and can react with alcohols to form borate esters.
A borate is any of a range of boron oxyanions, anions containing boron and oxygen, such as orthoborate BO3−3, metaborate BO−2, or tetraborate B4O2−7; or any salt of such anions, such as sodium metaborate, Na+[BO2]− and borax (Na+)2[B4O7]2−. The name also refers to esters of such anions, such as trimethyl borate B(OCH3)3.
Molybdenum trioxide describes a family of inorganic compounds with the formula MoO3(H2O)n where n = 0, 1, 2. These compounds are produced on the largest scale of any molybdenum compound. The anhydrous oxide is a precursor to molybdenum metal, an important alloying agent. It is also an important industrial catalyst. It is a yellow solid, although impure samples can appear blue or green.

Boron trioxide or diboron trioxide is the oxide of boron with the formula B2O3. It is a colorless transparent solid, almost always glassy (amorphous), which can be crystallized only with great difficulty. It is also called boric oxide or boria. It has many important industrial applications, chiefly in ceramics as a flux for glazes and enamels and in the production of glasses.

Phosphorus pentoxide is a chemical compound with molecular formula P4O10 (with its common name derived from its empirical formula, P2O5). This white crystalline solid is the anhydride of phosphoric acid. It is a powerful desiccant and dehydrating agent.

Telluric acid, or more accurately Orthotelluric acid, is a chemical compound with the formula Te(OH)6, often written as H6TeO6. It is a white crystalline solid made up of octahedral Te(OH)6 molecules which persist in aqueous solution. In the solid state, there are two forms, rhombohedral and monoclinic, and both contain octahedral Te(OH)6 molecules, containing one hexavalent tellurium (Te) atom in the +6 oxidation state, attached to six hydroxyl (–OH) groups, thus, it can be called tellurium(VI) hydroxide. Telluric acid is a weak acid which is dibasic, forming tellurate salts with strong bases and hydrogen tellurate salts with weaker bases or upon hydrolysis of tellurates in water. It is used as tellurium-source in the synthesis of oxidation catalysts.
Sodium perborate is chemical compound whose chemical formula may be written NaH2BO4, Na2H4B2O8, or, more properly, [Na+]2[B2O4(OH)4]2−. Its name is sometimes abbreviated as PBS.
Lithium metaborate is a chemical compound of lithium, boron, and oxygen with elemental formula LiBO2. It is often encountered as a hydrate, LiBO2·nH2O, where n is usually 2 or 4. However, these formulas do not describe the actual structure of the solids.

Phenylboronic acid or benzeneboronic acid, abbreviated as PhB(OH)2 where Ph is the phenyl group C6H5-, is a boronic acid containing a phenyl substituent and two hydroxyl groups attached to boron. Phenylboronic acid is a white powder and is commonly used in organic synthesis. Boronic acids are mild Lewis acids which are generally stable and easy to handle, making them important to organic synthesis.
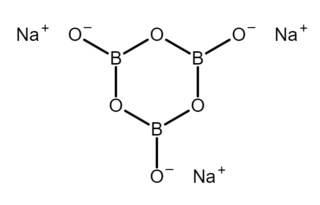
Sodium metaborate is a chemical compound of sodium, boron, and oxygen with formula NaBO2. However, the metaborate ion is trimeric in the anhydrous solid, therefore a more correct formula is Na3B3O6 or (Na+)3[B3O6]3−. The formula can be written also as Na2O·B2O3 to highlight the relation to the main oxides of sodium and boron. The name is also applied to several hydrates whose formulas can be written NaBO2·nH2O for various values of n.
Barium borate is an inorganic compound, a borate of barium with a chemical formula BaB2O4 or Ba(BO2)2. It is available as a hydrate or dehydrated form, as white powder or colorless crystals. The crystals exist in the high-temperature α phase and low-temperature β phase, abbreviated as BBO; both phases are birefringent, and BBO is a common nonlinear optical material.
Boron phosphate is an inorganic compound with the chemical formula BPO4. The simplest way of producing it is the reaction of phosphoric acid and boric acid. It is a white infusible solid that evaporates above 1450 °C.

A metaborate is a borate anion consisting of boron and oxygen, with empirical formula BO−2. Metaborate also refers to any salt or ester of such anion. Metaborate is one of the boron's oxyanions. Metaborates can be monomeric, oligomeric or polymeric.
Fluorooxoborate is one of a series of anions or salts that contain boron linked to both oxygen and fluorine. Several structures are possible, rings, or chains. They contain [BOxF4−x](x+1)− units BOF32− BO2F23−, or BO3F14−. In addition there can be borate BO3 triangles and BO4 tetrahedrons. These can then be linked by sharing oxygen atoms, and when they do that, the negative charge is reduced. They are distinct from the fluoroborates in which fluorine is bonded to the metals rather than the boron atoms. For example, KBBF, KBe2BO3F2 is a fluoroborate and has more fluorine and oxygen than can be accommodated by the boron atom.
Tetraboric acid or pyroboric acid is a chemical compound with empirical formula H2B4O7. It is a colourless water-soluble solid formed by the dehydration or polymerization of boric acid.
Hexahydroborite is a mineral composed of calcium, boron, oxygen, and hydrogen, with formula CaB2H12O6, more precisely [Ca2+]([B(OH4)]−)2·2H2O.
Disodium enneaborate is the traditional name for a salt of sodium, boron, oxygen, and hydrogen, with elemental formula Na2B9H22O20 or Na2B9O9·11H2O. It is the sodium borate with the highest boron/sodium ratio.
Trisodium borate is a chemical compound of sodium, boron, and oxygen, with formula Na3BO3, or (Na+)3[BO3]3−. It is a sodium salt of the orthoboric acid B(OH)3.

Sodium tetrahydroxyborate is a salt of with chemical formula NaH4BO4 or Na+[B(OH)4]−. It is one of several sodium borates. At room temperature it is a colorless crystalline solid.
Protactinium compounds are compounds containing the element protactinium. These compounds usually have protactinium in the +5 oxidation state, although these compounds can also exist in the +2, +3 and +4 oxidation states.











