
Rhenium is a chemical element with the symbol Re and atomic number 75. It is a silvery-gray, heavy, third-row transition metal in group 7 of the periodic table. With an estimated average concentration of 1 part per billion (ppb), rhenium is one of the rarest elements in the Earth's crust. Rhenium has the third-highest melting point and second-highest boiling point of any stable element at 5903 K. Rhenium resembles manganese and technetium chemically and is mainly obtained as a by-product of the extraction and refinement of molybdenum and copper ores. Rhenium shows in its compounds a wide variety of oxidation states ranging from −1 to +7.

Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively combined with other elements in chemical compounds. It was identified as a new element in 1781 and first isolated as a metal in 1783. Its important ores include scheelite, and wolframite, lending the element its alternate name.

Ceric ammonium nitrate (CAN) is the inorganic compound with the formula (NH4)2Ce(NO3)6. This orange-red, water-soluble cerium salt is a specialised oxidizing agent in organic synthesis and a standard oxidant in quantitative analysis.

Tungsten(VI) oxide, also known as tungsten trioxide or tungstic anhydride, WO3, is a chemical compound containing oxygen and the transition metal tungsten. It is obtained as an intermediate in the recovery of tungsten from its minerals. Tungsten ores are treated with alkalis to produce WO3. Further reaction with carbon or hydrogen gas reduces tungsten trioxide to the pure metal.
Acid salts are a class of salts that produce an acidic solution after being dissolved in a solvent. Its formation as a substance has a greater electrical conductivity than that of the pure solvent. An acidic solution formed by acid salt is made during partial neutralization of diprotic or polyprotic acids. A half-neutralization occurs due to the remaining of replaceable hydrogen atoms from the partial dissociation of weak acids that have not been reacted with hydroxide ions (OH−) to create water molecules. Acid salt is an ionic compound consisted of an anion, contributed from a weak parent acid, and a cation, contributed from a strong parent base.

In chemistry, a tungstate is a compound that contains an oxoanion of tungsten or is a mixed oxide containing tungsten. The simplest tungstate ion is WO2−
4, "orthotungstate". Many other tungstates belong to a large group of polyatomic ions that are termed polyoxometalates, ("POMs"), and specifically termed isopolyoxometalates as they contain, along with oxygen and maybe hydrogen, only one other element. Almost all useful tungsten ores are tungstates.

Sodium tungstate is the inorganic compound with the formula Na2WO4. This white, water-soluble solid is the sodium salt of tungstic acid. It is useful as a source of tungsten for chemical synthesis. It is an intermediate in the conversion of tungsten ores to the metal.
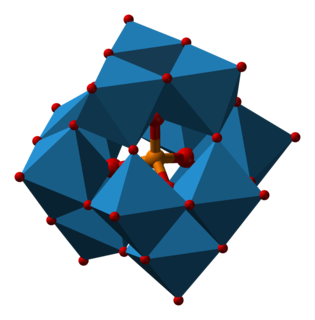
Phosphotungstic acid (PTA), tungstophosphoric acid (TPA), is a heteropoly acid with the chemical formula H3PW12O40. It is normally present as a hydrate. EPTA is the name of ethanolic phosphotungstic acid, its alcohol solution used in biology. It has the appearance of small, colorless-grayish or slightly yellow-green crystals, with melting point 89 °C (24 H2O hydrate). It is odorless and soluble in water (200 g/100 ml). It is not especially toxic, but is a mild acidic irritant. The compound is known by a variety of different names and acronyms (see 'other names' section of infobox).

In chemistry, a vanadate is an anionic coordination complex of vanadium. Often vanadate refers to oxoanions of vanadium, most of which exist in its highest oxidation state of +5. The complexes [V(CN)6]3- and [V2Cl9]3- are referred to as hexacyanovanadate and nonachlorodivanadate.

Metal nitrosyl complexes are complexes that contain nitric oxide, NO, bonded to a transition metal. Many kinds of nitrosyl complexes are known, which vary both in structure and coligand.

Ammonium tetrathiomolybdate is the chemical compound with the formula (NH4)2MoS4. This bright red ammonium salt is an important reagent in the chemistry of molybdenum and has been used as a building block in bioinorganic chemistry. The thiometallate anion has the distinctive property of undergoing oxidation at the sulfur centers concomitant with reduction of the metal from Mo(VI) to Mo(IV).

Ammonium paratungstate (or APT) is a white crystalline salt with the chemical formula (NH4)10(H2W12O42)·4H2O. It is described as "the most important raw material for all other tungsten products."

Hexamethyltungsten is the chemical compound W(CH3)6 also written WMe6. Classified as a transition metal alkyl complex, hexamethyltungsten is an air-sensitive, red, crystalline solid at room temperature; however, it is extremely volatile and sublimes at −30 °C. Owing to its six methyl groups it is extremely soluble in petroleum, aromatic hydrocarbons, ethers, carbon disulfide, and carbon tetrachloride.
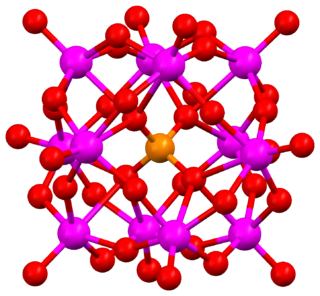
Keggin structure is the best known structural form for heteropoly acids. It is the structural form of α-Keggin anions, which have a general formula of [XM12O40]n−, where X is the heteroatom (most commonly are P5+, Si4+, or B3+), M is the addenda atom (most common are molybdenum and tungsten), and O represents oxygen. The structure self-assembles in acidic aqueous solution and is the most stable structure of polyoxometalate catalysts.
Thiophosphates are chemical compounds and anions with the general chemical formula PS
4−xO3−
x and related derivatives where organic groups are attached to one or more O or S. Thiophosphates feature tetrahedral phosphorus(V) centers.

Ammonium fluorosilicate (also known as ammonium hexafluorosilicate, ammonium fluosilicate or ammonium silicofluoride) has the formula (NH4)2SiF6. It is a toxic chemical, like all salts of fluorosilicic acid. It is made of white crystals, which have at least three polymorphs and appears in nature as rare minerals cryptohalite or bararite.
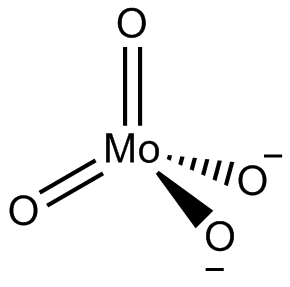
In chemistry a molybdate is a compound containing an oxoanion with molybdenum in its highest oxidation state of 6. Molybdenum can form a very large range of such oxoanions which can be discrete structures or polymeric extended structures, although the latter are only found in the solid state.The larger oxoanions are members of group of compounds termed polyoxometalates, and because they contain only one type of metal atom are often called isopolymetalates. The discrete molybdenum oxoanions range in size from the simplest MoO2−
4, found in potassium molybdate up to extremely large structures found in isopoly-molybdenum blues that contain for example 154 Mo atoms. The behaviour of molybdenum is different from the other elements in group 6. Chromium only forms the chromates, CrO2−
4, Cr
2O2−
7, Cr
3O2−
10 and Cr
4O2−
13 ions which are all based on tetrahedral chromium. Tungsten is similar to molybdenum and forms many tungstates containing 6 coordinate tungsten.

The cyanonickelates are a class of chemical compound containing anions consisting of nickel atoms, and cyanide groups. The most important of these are the tetracyanonickelates containing four cyanide groups per nickel. The tetracyanonickelates contain the [Ni(CN)4]2− anion. This can exist in solution or in solid salts. The ion has cyanide groups arranged in a square around the central nickel ion. The symmetry of the ion is D4h. The distance from the nickel atom to the carbon is 1.87 Å, and the carbon-nitrogen distance is 1.16 Å. Tetracyanonickelate(II) can be oxidised electrochemically in solution to yield tetracyanonickelate(III) [Ni(CN)4]−. [Ni(CN)4]− is unstable and Ni(III) oxidises the cyanide to cyanate OCN−. Tetracyanonickelate(III) can add two more cyanide groups to form hexacyanonickelate(III).

In chemistry, metatungstate refers to the anion with the formula [W12O40]8- and salts derived from this anion. The term also refers to protonated derivatives of this anion, including [H2W12O40]6-. The unprotonated anion [W12O40]12- has Td symmetry.

In chemistry, a transition metal chloride complex is a coordination complex that consists of a transition metal coordinated to one or more chloride ligand. The class of complexes is extensive.
![Structure of the paratungstate [H2W12O42] , highlighting its relatively low symmetry. EntryWithCollCode15237.png](http://upload.wikimedia.org/wikipedia/commons/thumb/7/7f/EntryWithCollCode15237.png/480px-EntryWithCollCode15237.png)

















