Research

Her early research was on the cattle disease, bovine viral diarrhoea. Nuttall found that the bovine viral diarrhoea virus was often present as a contaminant in foetal bovine serum, a commonly used laboratory reagent, a result that was published in Nature in 1977. [4] [8] She then worked with ornithologist Chris Perrins trying to identify the virus responsible for puffinosis, a disease that affects the Manx shearwater (Puffinus puffinus) sea bird. [4] [9] During this research, Nuttall visited sea-bird colonies and became interested in ticks, arthropods that often infest sea birds as well as other vertebrates, and can act as vectors for disease. [4]
Nonsystemic transmission
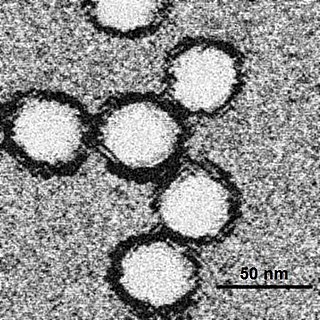
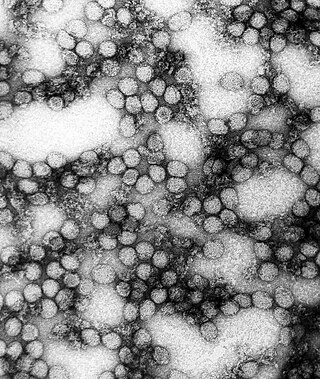
In the early 1980s, Nuttall started to study viruses transmitted by ticks, initially focusing on orbiviruses, nairoviruses and thogotoviruses. [1] While performing experiments designed to explore whether Thogoto virus, an influenza-like virus, can be transmitted between ticks during mating, Nuttall and her colleagues unexpectedly found that control ticks became infected. [4] They discovered that the virus can be transmitted between infected and uninfected ticks when they feed simultaneously on apparently uninfected guinea pigs, without the virus being present at a detectable level in the blood. [10] [11] [12] With Milan Labuda, Nuttall subsequently demonstrated the same phenomenon with tick-borne encephalitis virus, a better-understood arthropod-transmitted virus. [13]
These results challenge the then-accepted idea that arthropod-transmitted viruses can only be transmitted when the arthropod vector feeds on an infected host in which the virus is replicating and circulating in the blood above a threshold level. [4] [11] [12] [14] Sarah Randolph comments:
Suddenly the conventional wisdom that systemic infections above a certain threshold level were necessary for transmission, and could therefore be used to assay host competence, was over-turned. Viraemia and even bacteraemia are now seen as an inconsistent, species-specific consequence of infection but not a necessary condition for transmission. [12]
The phenomenon of nonviraemic or nonsystemic transmission turns out to be widespread – it has been subsequently observed with many other viruses transmitted by ticks, black flies and mosquitos, including major human pathogens such as West Nile virus, as well as other pathogens, including the spirochaete Borrelia burgdorferi , which causes Lyme disease. [11] [15] [16] – and important in natural transmission. [14] Nuttall's group has continued to study the phenomenon, and have shown that immunity to tick-borne encephalitis virus does not prevent nonsystemic transmission. [17]
Tick saliva

Nuttall's recent work has focused on discovering the function of tick saliva's many constituents, as well as the mechanisms by which tick-transmitted pathogens use them to enhance their transmission. [3] Nuttall and colleagues were the first to show, in 1989, that tick saliva promotes the transmission of viruses, a phenomenon which they have called "saliva-assisted transmission"; this has been suggested as a mechanism for nonsystemic transmission. [4] [12] [18] [19] In 1998, she and her colleagues characterised three immunoglobulin-binding proteins, which were among the first proteins from tick saliva to be sequenced. [12] [20] [21] With Norbert Fuchsberger, Valeria Hajnicka and others, Nuttall has shown that tick saliva suppresses the host's antiviral immune responses, including natural killer cell activity and the induction of type I interferon and cytokines involved in inflammation. [4] Her group has also shown that the proteins in saliva differ between individual ticks of the same species and also change over the course of feeding. [20]
Drugs and vaccines
Some components of tick saliva have potential as drugs to treat a range of conditions unrelated to tick disease. [3] [22] Nuttall comments that "These molecules have been refined by millions of years of evolution. There are no toxicity problems, they work on a range of animals, they aren't fragile — and there are an awful lot of them." [22] For example, with Guido Paesen, Nuttall has characterised histamine-binding proteins that can suppress inflammation in humans by binding directly to histamine, rather than blocking its access to cell receptors, of which at least four are known. [4] [22] [23] One of these proteins, rEV131, was investigated by NERC spin-off company Evolutec for the treatment of hay fever and for use in recovery from cataract surgery. [4] [22] Another example is the anticoagulant variegin, discovered by Nuttall and Maria Kazimirova in the tropical bont tick (Amblyomma variegatum), which represents a novel class of thrombin inhibitor; it has been shown to prevent venous thrombosis in a zebrafish model. [24] [25]
Tick saliva products are also possible targets for vaccines to control tick infestation and, potentially, to prevent the diseases they carry. [3] [20] One vaccine candidate explored by Nuttall's group is 64TRP, a 15 kDa Rhipicephalus appendiculatus protein from the cement cone that glues the tick's mouthparts to the host. Vaccination with 64TRP can protect against tick-borne encephalitis virus carried by a different type of tick, the castor bean tick (Ixodes ricinus), in a mouse model. [20] [26]